My Questions | bartleby sw5_25473_1jpg (700x686) b Digital Resources for Chemistry Smartwork5 X X X sw5.wwnorton.com/problemsets/filemanager/userfiles/ctcox@stanford.edu/sw5_25473_1jpg Time 0s Sample A Time 5s Time 0 s Time 5 s Sample B 9:46 AM Type here to search 10/24/2019
Q: What is ΔG0 (in units of J) for the following reaction at 298K? O2(g) + 4H+(aq) +4Fe2+(aq) →…
A: The given reaction:
Q: Consider the following processes: 2A → (1/2)B - C AH¡ = 5 kJ/mol (3/2)B + 4C - · 2A + C + 3D AH,…
A: Given data :-
Q: [References] Carbon dioxide from the atmosphere "weathers," or dissolves, limestone (CACO3) by the…
A:
Q: The energy for the system to reach 20.5 °C is supplied by a chunk of hot metal. What would be the…
A: The heat of a reaction is the amount of energy absorbed or released during the course of a reaction.
Q: Calculate the ΔG° (kJ/mol) for the following reaction at 152 °C, using the following data. Eneter…
A: Answer: -2085.472485 kJ/mole is the value of ∆G°rxn at 152°C
Q: 19.8 (Pages 871-877) ; Watch WE 19.6. Consider the following reaction: 285 K CACO, (s) - CaO (s)+CO,…
A:
Q: Consider the following chemical reaction and calculate the ΔHrxn (you do not need to write out the…
A: Interpretation - To determine the enthalpy of the reaction for the given reaction M(g) + 2 N(s) →…
Q: What is the change in the overall standard entropy (S°) for the following chemical reaction. 31 (aq)…
A:
Q: For the reaction2CO2(g)+ 5H2(g)C2H2(g) + 4H2O(g)H° = 5 kJ and S° = -125 J/KAt standard conditions,…
A: Given that delta S is -125J/K and the value of delta H is 5kJ. Thus, the value of delta G in…
Q: For reactions carried out under standard-state conditions, the equation AG=AH– TAS becomes AG° = H°…
A: The given data is as follows: N2O4(g) ⇌ 2NO2(g) ∆H°=58.0 kJ/mol=58000 J/mol T1=25 °C=25+273=298 K…
Q: ΔHo reaction = -112.2kJ ΔS = -15.26JK-1mol-1 ΔG = -18.81kJ Discuss the meaning of each term/value…
A: ΔHo reaction = -112.2kJ ΔH means change in enthalpy , that is amount of heat absorbed or evolved…
Q: Enter your answer in the provided box. Calculate AG for the process: BaF2(s) = Ba*(aq) + 2F (aq) The…
A: Given data : 1.Ksp = 1.70×10-6 2. Temperature T = 25°C = (25+273)K T= 298K
Q: Styrene is produced by catalytic dehydrogenation of ethylbenzene at high temperature in the presence…
A:
Q: Find the q dissolution given the following: Mass of Water= 200g Initial Temperature of Water=…
A: Given : Mass of water = 200 g Initial temperature of water = 20.0 °C Final temperature of water =…
Q: 2. Calculate reaction AH°rxn and reaction AU°rxn for the total oxidation of benzene. Use the stable…
A: The complete oxidation of benzene gives Carbon dioxide gas and liquid water as - C6H6(l) + 7.5O2(g)…
Q: 2. For a particular hypothetical reaction, A+ B → 2C , the value of AG° is -125 kJ/mol. What is the…
A:
Q: CO(g)+2H2(g)⇌CH3OH(g) K = 2.18×102 at 340 K Calculate ΔrG for the reaction at 340 K in STANDARD…
A: Given: Equilibrium constant, K = 2.18 × 102 Temperature = 340 K.
Q: 12. The following process occurs naturally: C (diamond) C (graphite) AG° = -2.90 kJ/mol (a) Is this…
A: Graphite and diamond allotrops of carbon. They have different type of bonding and molecular…
Q: Find AGrn for the reaction: 3 C(s) + 4 Hz(g) → CaH(g) Use the following reactions with known values…
A:
Q: Calculate the change in internal energy of the following reaction at 1 atm and 25oC. 4NH3(g) +…
A: We have to calculate the change in internal energy of the following reaction at 1 atm and 25oC.…
Q: The pressure equilibrium constant for the dissociation reaction 2 H2S (g) = 2 H2 (g) + S2 (g) is…
A: Given data: Kp1 = 0.0118 Temperature T1 = 1065o C ∆Hodissociation = 117.4 kJ Temperature T2 = 1200o…
Q: Calculate the overall AG°ƒfor the chemical reaction at 100°C in units of kJ/mole to 2 decimal…
A:
Q: For a reaction 2A+B2-2AB AG=-35.3 kJ-mol-1 and AG = -28.5 kJ.mol-1 at a temperature of 419 K.…
A:
Q: Determine ΔSsurr for the reaction (in kJ/mol K) Group of answer choices
A: The reaction
Q: Carbon dioxide from the atmosphere "weathers," or dissolves, limestone (CaCO3) by the reac CACO3 (s)…
A:
Q: Use the standard molar entropies to calculate ΔS°rxn for the following reactions: Ag+ (aq)+Cl-…
A: Answer :- a) ∆S° = -207 (J/K) Ag+(aq) +Cl-(aq) → AgCl(s) , ∆S° = -207 (J/K)…
Q: 4. a. Consider the data below for the reaction H2Ou) = H2O() - Plot a graph and determine AH and AS…
A: When a system receives an amount of energy q at a constant temperature, T, the entropy increase DS…
Q: Dissolution of Ammonium Nitrate mass of water: 200 g initial temperature of water: 20°C final…
A: Dissolution of ammonium nitrate in water is endothermic reaction. It absorbs heat from the system
Q: c56b6cb03#10302 I Review I Constants I Periodic neat capacity of an object indicates how much gy…
A: The questions are based on the concept of chemical thermodynamics. we have to calculate mass of the…
Q: For the reaction Cr(C6H6)2 (s) --> Cr(s) + 2 C6H6 (g), the reaction internal energy change at 1 atm…
A: The internal energy change can be calculated by the following formula. ∆H=∆U-∆n(g)RT∆U=change in…
Q: What is the value of AG (kJ mol-1 ) for this reaction at 201.4 °C? The data refer to 25 °C: P2 (g) +…
A:
Q: Find AGpen for the reaction: 3 C(s) + 4 Hk(g) CaHs(g) Use the following reactions with known values…
A: The value of ∆G depends on the Stoichiometry of the chemical equation If a chemical equation is…
Q: ETHYLENE WHICH IS A GAS CAN BE HYDROGENATED TO PRODUCE ETHANE WHICH IS ALSO A GAS ACCORDING TO THE…
A: Here we are required to find the molar quantity of the reactant
Q: In the reactions at 298.15K (1) C(diamond)+O2(g)-CO2(g) 4,H,=-395.4kJ.mol. (2)…
A: According to Hess's law change of enthalpy in a chemical reaction is same regardless of whether the…
Q: Using the data, which DG vs T plot most accurately reflects this reaction? CI -OH OH AH ran -110.6…
A: Given: ΔH = -110.6 KJ/mol = -110600 J/mol. (Since 1 KJ = 1000 J) And ΔS =…
Q: Reaction 1: FeO(s) +CO(g)-→ Fe(1) + CO,(g) AGn >0 C(s) + CO2(g) 2 CO(g) AG <0 Reaction 2: Overall…
A: Particle diagrams are used to represent the composition of the elements or compounds present in a…
Q: For a reaction 2A+B2-2AB AG= 34.8 kJ-mol-1 and AG = 42.4 kJ.mol-1 at a temperature of 143 K.…
A: Given data : 2A + B2 ---->2AB Standard Gibbs free energy = 42.4KJmol-1 Gibbs free energy =…
Q: For the following reaction A,H° and AS° are -58.03 kJ mol and -176.6 J mol K-. respectively. 2…
A: The question is based on the concept of chemical equilibrium. we have to calculate Gibbs free energy…
Q: For the combustion of carbon monoxide; CO + 1/2 02→ CO, find the ratio of the equilibrium constants…
A:
Q: The equilibrium constant Kc for the reaction fructose-1,6-diphosphate = glyceraldehyde-3-phosphate +…
A: We have to find out the standard Gibbs free energy and change in Gibbs free energy for the given…
Q: Calculate the values of E°, A,G°, and K for the following reactions at 25.00°C. (Assume R =…
A: We would use following formulae to calculate these values : 1.) E° = E°r ( cathode ) - E°r ( anode )…
Q: When pure (18M) sulfuric acid dissolves in water, a significant increase in the temperature of the…
A: The spontaneous nature of the solvation is determined by calculating the change of free energy of…
Q: 3. Use the data oven i the table to calculate AHrxn, ASrxn, AGrxn, and Keq for the following…
A:
Q: For the reaction S(s,rhombic) + 2co(g)----> so2(g) +2c(s, graphite) Delta H =75kj and delta s=…
A: The question is based on the concept of chemical thermodynamics. we have to calculate equilibrium…
Q: Given the partial pressures of H20, CO, and H2, what is the reaction quotient Q for the following…
A:
Q: CO(g)+2H2(g)⇌CH3OH(g) K=2.18×10^2 at 340 K Calculate ΔrG for the reaction at 340 K at EQUILIBRIUM…
A: Given: Equilibrium constant, K = 2.18 × 102 And temperature = 340 K
Q: What is the value of AG (kJ mol-1) for this reaction at 57.4 °C? The data refer to 25 °C: P2 (g) +…
A:
Q: What is the value of AG (kJ mol-1) for this reaction at 349.4 °C? The data refer to 25 °C: P2 (g) +…
A:
Q: 4.00 LL reaction vessel at 1100 KK: C(s)+H2O(g)⇌CO(g)+H2(g)C(s)+H2O(g)⇌CO(g)+H2(g) If during the…
A:
Q: AH AS AG0 -TAS alt- NE AG-AH-TAS Effect of Temperature on Spontaneity AG oinen Reaction…
A: ∆G = ∆H - T∆S When ∆G > 0 => reaction is non spontaneous When ∆G < 0…
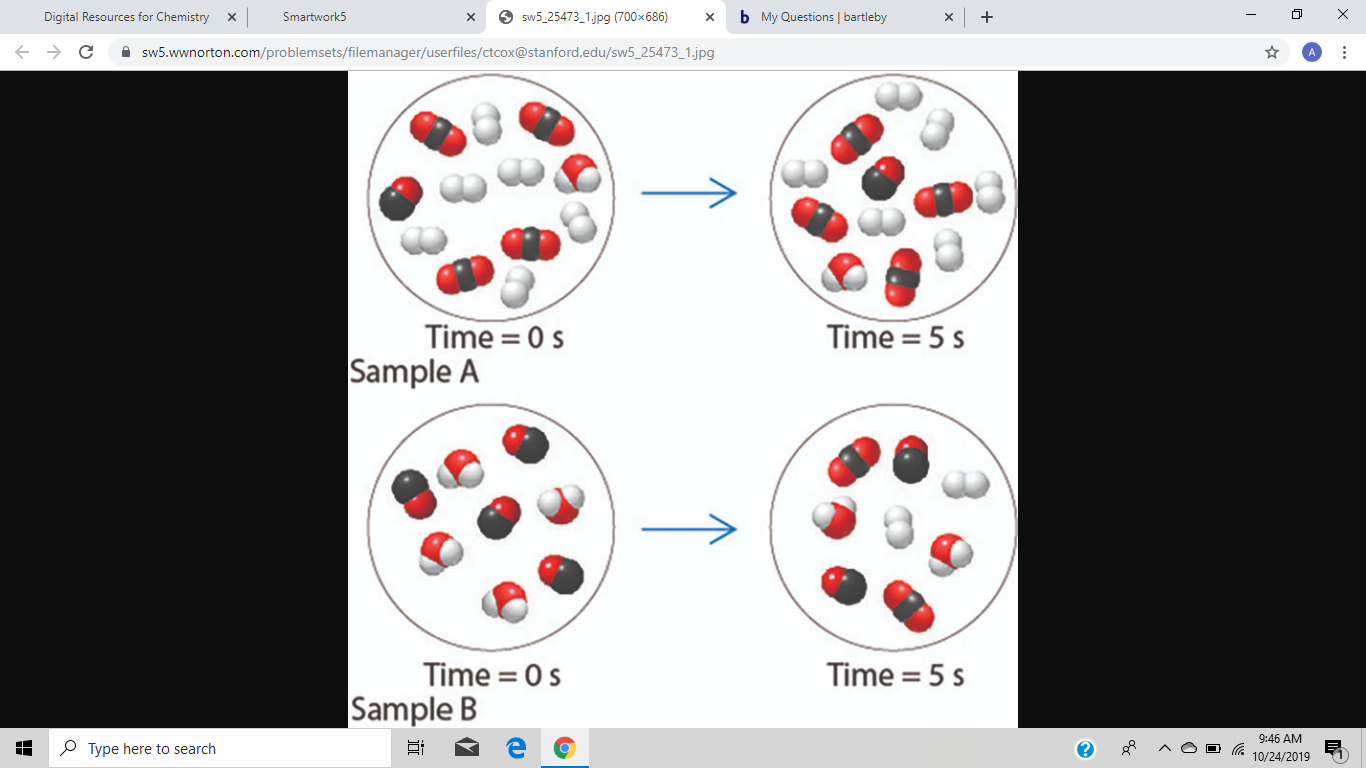
Two
Calculate the reaction quotient Q, for Sample B at t = 5 s.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Chemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.A. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…
- There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…Convert kPa/sec to uL/min for the following values. -0.0019 kPa/sec -0.0029 kPa/sec -0.0028 kPa/sec -0.0021 kPa/sec -0.0026 kPa/sec -0.0034 kPa/secChemistry Fraction no. Elution Vol. (mL) Abs (λ = 410) Fraction no. Elution Vol. (mL) Abs (λ = 410) 1 (PB pH 6.0) 1 0.0050 11 11 0.0041 2 2 0.0032 12 12 0.0060 3 3 0.0040 13 13 0.0064 4 4 0.0064 14 14 0.0063 5 5 0.0067 15 15 0.1109 6 6 0.0067 16 16 2.8902 7 7 0.0050 17 17 0.8500 8 (PB pH 8.0) 8 0.0049 18 18 0.2019 9 9 0.0052 19 19 0.0378 10 10 0.0050 20 20 0.0073 Use the data above to plot an elution profile, in which the absorbance is on Y-axis, and elution volume (volume = fraction number) is on X-axis. The elution profile must indicate at (1) which point the pH change occurred, (2) the fraction(s) which contain the highest myoglobin content, and (3) the maximum absorbance detected.
- Pls answer the question linked below: https://pasteboard.co/0afMgF1vi1jX.pngy= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)I would need help with these questions. The method referenced above was followed by a student and she got the following results: Caffeine Std. Conc. (ppm) Absorbance 100 1.806 50 0.899 40 0.724 30 0.545 20 0.365 10 0.183 Further, she analyzed an unknown sample and she got the following results: Sample # Absorbance 1 0.398 1. Graphically find the concentration in ppm and then calculate in mol/L of Caffeine in Sample #1? 2. Which solvent was used to extract Caffeine and why? Why is it necessary to do extraction three times? 3. Cuvettes used in this experiment were made from which material and why? 4. At what wavelength the Absorbance was measured?
- Hello, I'm having difficulties to answer the chemistry problem. Can you please help me with all the parts? I really need help. Also, I need to be prepared for the exam. Please I need help with all the parts a,b,c,d. 7. A 4.82g sample of iron is heated to 84.3 C. A 1.25 g sample of gold is heated to 95.1 C. Both of these metals are added to 9.47 g of cold water that is initally at 14.5 C. a) From the first law of thermodynamics, what is the equation relating the sum of heats of two components? b) Algebraically substitute in the specific heat capacites for the metal and water, the masses of the samples, and the temperature change (see problem 7.) c) Solve the algebraic problem (b) for the Tp the final temperature of water. d) Determine the final temperature of the metals in water. Hint: it's the same for all three components.1. 500.0 mL 0.2 M KI2. 500.0 mL 0.2 M KCl3. 500.0 mL 0.1 M K2S2O84. 500.0 mL 0.1 M K2SO45. 500.0 mL 4.0 mM Na2S2O3 (from Na2S2O3∙5H2O)pls compelete the 2nd table given the data4. Ksp=4.1x10-36 for Pb3(AsO4)2Calculate Eofor Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO43-Pb2+ +2e- Pb(s) Eo= -0.126 V

