Nickel carbonyl decomposes to form nickel and carbon monoxide, like this: Ni(co),(9)→ Nis)+4 CO(9) At a certain temperature, a chemist finds that a 2.5 L reaction vessel containing a mixture of nickel carbonyl, nickel, and carbon monoxide at equilibrium has the following composition: compound amount 0.103 g Ni(CO), 61.3 g Ni 1.42 g co Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. K = 0
Nickel carbonyl decomposes to form nickel and carbon monoxide, like this: Ni(co),(9)→ Nis)+4 CO(9) At a certain temperature, a chemist finds that a 2.5 L reaction vessel containing a mixture of nickel carbonyl, nickel, and carbon monoxide at equilibrium has the following composition: compound amount 0.103 g Ni(CO), 61.3 g Ni 1.42 g co Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. K = 0
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 60QRT
Related questions
Question
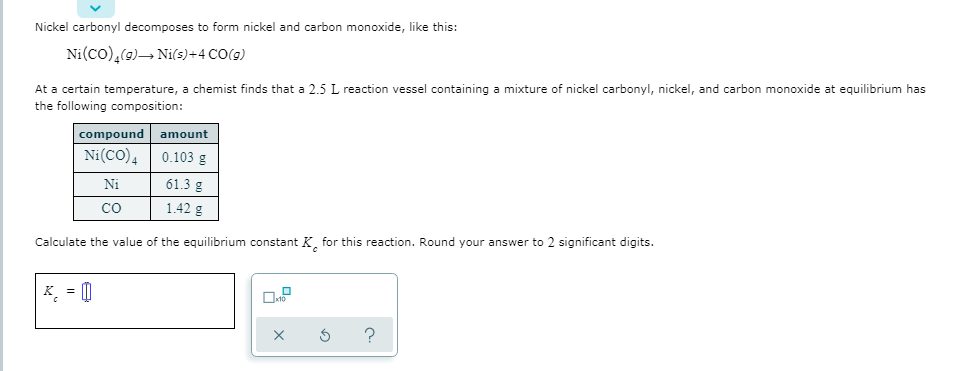
Transcribed Image Text:Nickel carbonyl decomposes to form nickel and carbon monoxide, like this:
Ni(co),(9)→ Nis)+4 CO(9)
At a certain temperature, a chemist finds that a 2.5 L reaction vessel containing a mixture of nickel carbonyl, nickel, and carbon monoxide at equilibrium has
the following composition:
compound amount
0.103 g
Ni(CO),
61.3 g
Ni
1.42 g
co
Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits.
K = 0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning