Q: The energyE of the electron in a hydrogen atom can be calculated from the Bohr formula: R, E = In th...
A:
Q: B. How much zinc chloride is formed when 32.18 g of zinc reacts with excess hydrochloric acid, Use t...
A: Gg
Q: How much carbohydrates calories in one food product serving, if it contains 23 of carb? 46 4 92 207
A:
Q: 3. Can you synthesis the following compound from ethyne? You can choose any other material you need....
A: Ethyne has triple bond and is acidic In nature and the acidic proton can be abstract by base
Q: The volume of 13.7 M H2SO4 solution required to prepare 285 mL of 1.17 M H2SO4 is _________mL.
A: Here we have to calculate volume of H2SO4 needed to prepare 285 mL, 1.17M H2SO4 solution-
Q: identify the systematic name
A:
Q: Arrange the compounds in order of increasing solubility in water (least soluble first).
A:
Q: 2. DECOLORIZATION Dissolve 1g of brown sugar in 15 ml. dist. Water in a small beaker and divide the ...
A: Since you have asked multiple questions, we will answer only first question for you. In order to get...
Q: Weigh out around 0.3 g of cobalt chloride hexahydrate, CoCl,-6H,O, and place it in a clean, dry beak...
A: Equlibrium reaction of cobalt chloride hexahydrate with ethanol is given below.
Q: -393.5 kJ mol AH°rxn = -285.8 kJ mol AH°rxn = - 3119.6 kJ mol" AH°rxn C(graphite) + O:(g)→CO2(g) H2(...
A:
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: R. E=- n In...
A: Given, Energy of electron in hydrogen atom , E=-Ryn2 where Ry is rydberg constant , Ry=1.097×107 m-1
Q: H
A:
Q: QUESTION 28 This is a process in which the precipitate is heated for an hour or more in the solution...
A: Formed precipitation heating is called as ?
Q: 1. An unknown carbohydrate gave a red precipitate when tested with Benedict's reagent and quickly ga...
A: Here we have to draw a conclusion about a carbohydrate which reduce Benedict's reagent and Barfoed's...
Q: Calculate the number of milliliters of 0.650 M NaOH required to precipitate all of the Zn2+ ions in ...
A: Given : Zn(NO3)2(aq) + 2NaOH(aq) Zn(OH)2(s) + 2NaNO3(aq) Molarity of NaOH solution = 0.650 M or 0....
Q: What minimum mass of H2SO4 would you need? Express your answer in grams.
A:
Q: Draw dot-and-cross diagrams for the ions in the following ionic compounds. Show only the outer elect...
A:
Q: The normal boiling point of a certain liquid X is 121.00 °C, but when 0.12 kg of benzamide (C,H,NO) ...
A:
Q: Gold is alloyed (mixed) with other metals to increase its hardness in making jewellery. Consider a p...
A: Given: Total weight of jewellery = 12 g Volume of jewellery = 0.675 cm3 Density of gold = 19.3 g/cm3...
Q: CH3 H3CH2C CH,CH2CH 1. CH3 C=C H CH3
A: IUPAC naming of the given compounds.
Q: Can someone explain how to get the following: h(1)-0(2)-c(3) bond angle? also, determine the hybri...
A: Given:
Q: What is the major product that results from the following reaction? TSCI NaOEt HO pyridine
A: The reaction is an example of substitution- elimination reaction
Q: what is ΔHcond for this substance
A:
Q: Test A. Instruction: Write the word equation and the balanced chemical equation of the given chemica...
A: The balanced chemical equations of given reactions are given below.
Q: QUESTION 4 Colloidal suspensions can often be coagulated by I. heating II. stirring III. adding a no...
A:
Q: Give all possible and complete reaction for the following. Properly name the organic compound. 1. Ox...
A:
Q: A metal radiator is made from 13.96 kg of iron. The specific heat of iron is 0.449 J/g °C. How much ...
A: Given;
Q: Give the IUPAC names of the following:
A:
Q: Chlorine is a(n) (CIRCLE one) ortho-para/meta director. Explain your answer. In your explanation, yo...
A: If any group, that pushes electron density towards ortho and para positions, that group is ortho par...
Q: Balance the following equations. Classify the type of chemical reaction. (NH),Cr,0, > N, + Cr,0, + H...
A:
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom or molecule: -B...
A: 1) Ground state is lowest energy state which is E Answer: E 2) D,C, B are all excited state So, t...
Q: CH, CH Mg co2 H;O*/H,0 CH,CH,Br diethyl ether KMno4 H3O* /H;O -CH,CH3 KOH / H,0 KCr,07 IV -CH2OH H;S...
A:
Q: Which of the following body fluids is least acidic? Gastric juice = pH 1.2 Urine = pH 5.5 Saliva = p...
A:
Q: Draw the skeletal structure of: cyclohexane-1,2-dicarbaldehyde
A: Draw the skeletal structure of: cyclohexane-1,2-dicarbaldehyde
Q: Why different molecular bonds have different IR spectroscopic peaks or bands? Please shortly write a...
A: when a infrared light goes through a organic compound , the compound absorbed some of the frequenci...
Q: 1. Please name the following compounds ÇI ÇH3 H,CHC-c-c=c-CH3 1) H ÇI CH3 2) H3CHC=ċ-ċ-CECH H ÇI CH3...
A: Write IUPAC name of the given--
Q: 8. (E)-1,2-bis(2,4,6-trinitrophenyl)ethene 9. 3-nitro-5-(trichloromethyl)phenyl benzoate 10. 4-ethyl...
A:
Q: When treated with an acid catalyst, 4-hydroxybutanoic acid forms a cyclic ester (a lactone). Draw th...
A: We have to predict structure formula of lactone as well as name.
Q: Draw a structural formula for the product formed by treating each compound with warm chromic acid, H...
A: Chromic acid is a strong oxidizing agent
Q: hat volume of H2 gas, measured at 9 °C and 722 torr, can be obtained by reacting 8.40 g of zinc meta...
A: Given, Temperature = 9oC = 273 + 9 = 282 K Pressure = 722 torr = 0.95 atm Volume of HCl = 185 ml = 0...
Q: How is the geometry of a molecule defined and why is the study of molecular geometry important?
A: The geometry of the molecule and it's important is given below.
Q: Problem: A gas cylinder has an internal volume of 2,600L. If the sun heats the gas cylinder from a t...
A:
Q: Write a summary for pre paration of p.Mitroaniline experiment pertaining to'three step syn' thesis, ...
A: Here we have to write summary of preparation of p-nitro aniline including 3 steps acetylation, nitra...
Q: Determine the CBOD and NBOD of 125 mg/L of CH;COOH solution. Assume the following reaction! CH3COOH ...
A: Solution -
Q: A metal has an atomic radius of 0.1385 nm . The volume of its cubic unit cell is 0115 x10-23 cm3. Wh...
A:
Q: AGNO3/NH3 3) H3C= Cu(NH3)* 4) H3C=
A: Reactions of alkynes:
Q: B .Find the molecular formula of the gas in the bacterial fermentation of grain to produce ethanol. ...
A: Given, The percentage of Carbon = 27.29 % The percentage of Oxygen = 72.71 %
Q: A- Draw each of the following compounds. 1. 1-Ethoxy-2-bromopropane 2. 3-Ethyl-4-methylcyclohexaneth...
A:
Q: The decomposition of a radioactive isotope follows first order rate law kinetics. Calculate the conc...
A: Radioactive decay follows the first-order kinetics. The half-life of the first-order reaction does n...
Q: The correct statement is…. Crystal field strength at [PtCl4]2- > [Pt(NH3)4]2+ Crystal field strength...
A: Crystal field strength of 5th period transition metal is more than 4rth period transition metal and ...
Give the IUPAC name
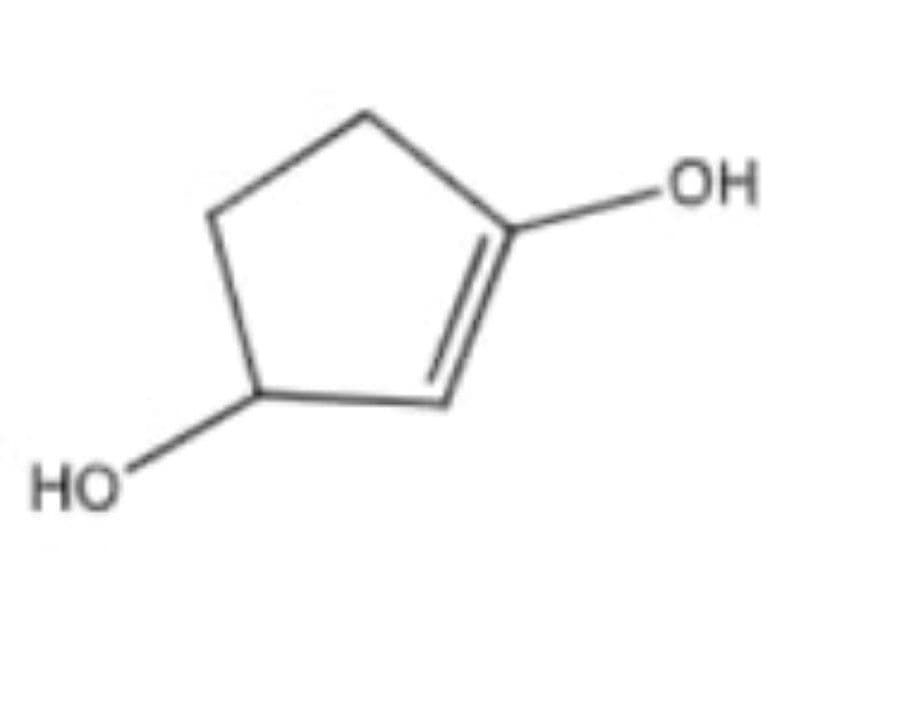
Step by step
Solved in 3 steps with 1 images


