O KINETICS AND EQUILIBRIUM Using the van't Hoff equation to predict K at a different tempera... -5 for a certain reaction. At -8.86 °C the concentration equilibrium constant K 4.4 x 10 с Here are some facts about the reaction: The net change in moles of gases is 2. O The initial rate of the reaction is 14. mol L S If the reaction is run at constant pressure, 106. kJ/mol of heat are absorbed. O Yes. x10 Using these facts, can you calculate K at 4.5 °C? с X No. If you said yes, then enter your answer at right. Round it to 2 significant digits. Explanation Check O 2019 McGra Lenovo II Yes. x10 Using these facts, can you calculate K at 4.5 °C? C ONo. If you said yes, then enter your answer at right. Round it to 2 significant digits. Yes, and K will be bigger If you said no, can you at least decide whether K at Yes, and K will be 4.5 °C will be bigger or smaller than K at -8.86 °C? smaller C No. Explanation Check X
O KINETICS AND EQUILIBRIUM Using the van't Hoff equation to predict K at a different tempera... -5 for a certain reaction. At -8.86 °C the concentration equilibrium constant K 4.4 x 10 с Here are some facts about the reaction: The net change in moles of gases is 2. O The initial rate of the reaction is 14. mol L S If the reaction is run at constant pressure, 106. kJ/mol of heat are absorbed. O Yes. x10 Using these facts, can you calculate K at 4.5 °C? с X No. If you said yes, then enter your answer at right. Round it to 2 significant digits. Explanation Check O 2019 McGra Lenovo II Yes. x10 Using these facts, can you calculate K at 4.5 °C? C ONo. If you said yes, then enter your answer at right. Round it to 2 significant digits. Yes, and K will be bigger If you said no, can you at least decide whether K at Yes, and K will be 4.5 °C will be bigger or smaller than K at -8.86 °C? smaller C No. Explanation Check X
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.45PAE: The following equilibrium is established in a closed container: C(s)+O2(g)CO2(g)H=393kJmol1 How does...
Related questions
Question
Using the information can you calculate Kc at 4.5 C. If you said yes enter the answer in second row. If no can you decide whether KC at 4.5 will be bigger or smaller than Kc at - 8.86
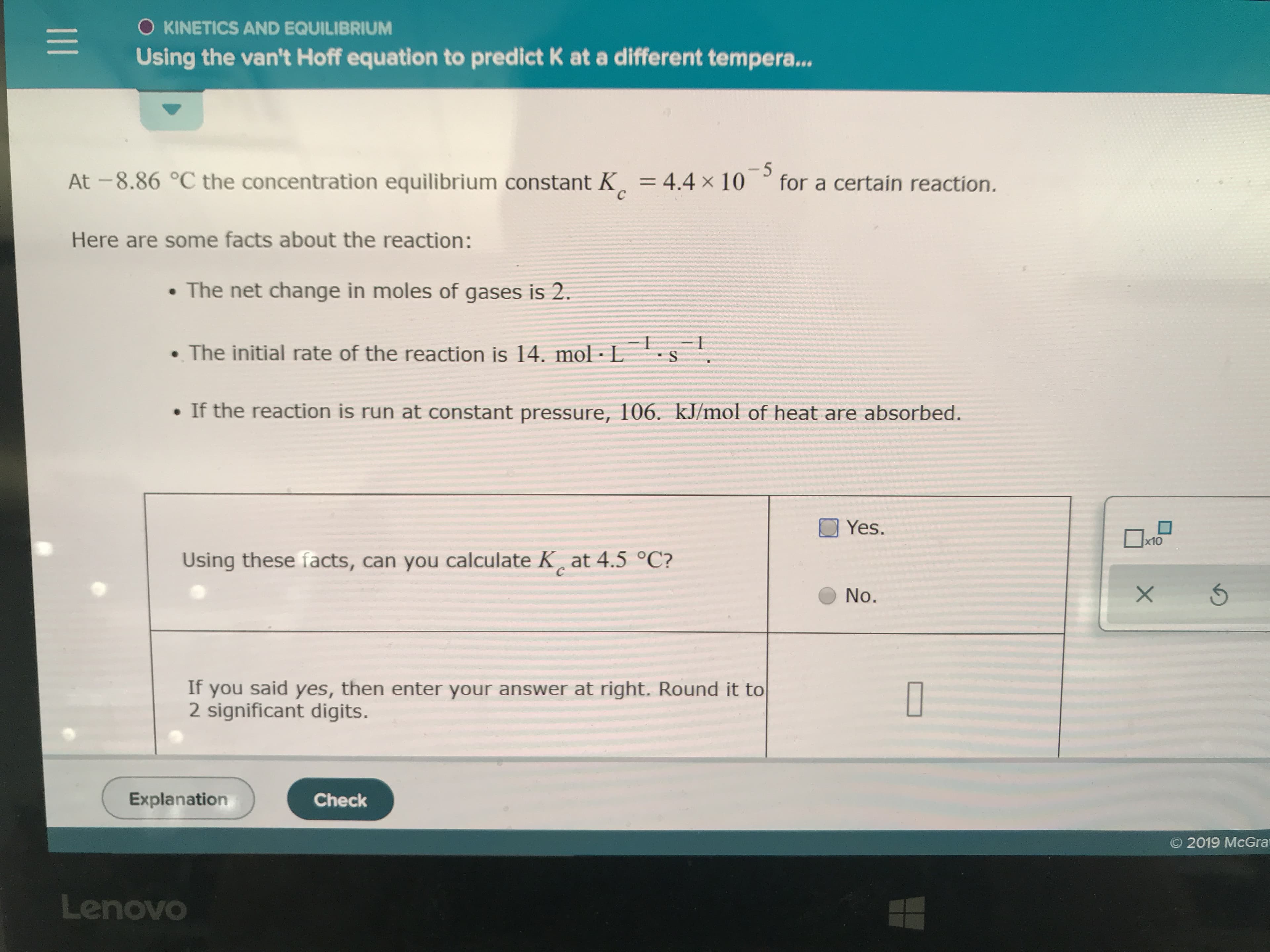
Transcribed Image Text:O KINETICS AND EQUILIBRIUM
Using the van't Hoff equation to predict K at a different tempera...
-5
for a certain reaction.
At -8.86 °C the concentration equilibrium constant K
4.4 x 10
с
Here are some facts about the reaction:
The net change in moles of gases is 2.
O
The initial rate of the reaction is 14. mol L
S
If the reaction is run at constant pressure, 106. kJ/mol of heat are absorbed.
O
Yes.
x10
Using these facts, can you calculate K at 4.5 °C?
с
X
No.
If
you said yes, then enter your answer at right. Round it to
2 significant digits.
Explanation
Check
O 2019 McGra
Lenovo
II

Transcribed Image Text:Yes.
x10
Using these facts, can you calculate K at 4.5 °C?
C
ONo.
If you said yes, then enter your answer at right. Round it to
2 significant digits.
Yes, and K will be
bigger
If you said no, can you at least decide whether K at
Yes, and K will be
4.5 °C will be bigger or smaller than K at -8.86 °C?
smaller
C
No.
Explanation
Check
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning