Q: What is the pH of a solution that results from mixing together equal volumes of a 0.020 M solution ...
A: Initial moles of NH3 = V x 0.0200 = 0.0200V molMoles of HCl added = V x 0.010 = 0.010V mol NH3 + HCl...
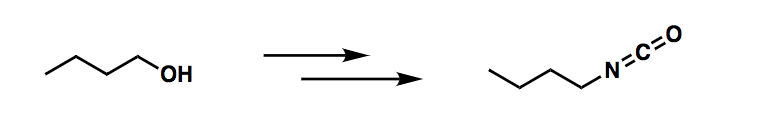
Q: Describe a synthesis for the following compound, using the given starting material and any necessary...
A: Please find below the synthesis reaction.
Q: The concentration of total phosphorous (HPO4^2- and H2PO4^-) found in a blood sample is determined b...
A: Henderson - hasselbach equation enables the calculation of pH of a buffer solution made by mixing kn...
Q: Characterize EACH of the three given statements as being TRUE or FALSE and then indicate the collect...
A: A solution always reflects a homogeneous mixture and suspension always reflects the heterogeneous mi...
Q: Explain why 1-bromo-2,2-dimethylpropane has difficulty undergoing both SN2 and SN1 reactions. . Can ...
A: During SN1 reaction, carbocation intermediate is formed. During SN2 reaction involves the attack of ...
Q: Biological catalysts are called enzymes. The concentrations of enzymes in cells are usually quite sm...
A: Enzymes are biological catalysts that influence the rate of a biochemical reaction. These biocataly...
Q: The tetraethyl lead Pb(C2H5)4 in a 25.00 mL sample of aviation gasoline was shaken with 15.00 mL of ...
A: To determine: Find the amount of tetraethyl lead present. The chemical reaction between thiosulphate...
Q: MasteringChemistry: CHE154 S X session.masteringchemistry.co This Mac can't connect to iCloud I a pr...
A: Concentration of OH- formed during the dissolution of NaF in water is determined as follows,
Q: Given that the solubility of BaC2O4 is s = 0.29 g/L, calculate the solubility product constant (Ksp)...
A: The solubility product constant (Ksp) represents the solubility of products at equilibrium for solid...
Q: Liquid butane (C4H10) is stored in cylinders to be used as afuel. The normal boiling point of butane...
A: According to Gay-Lussac’s Law, for a closed system, at constant volume (isochoric process), pressure...
Q: According to the second law of thermodynamics, the change in the entropy of the universe (ΔSuniv) du...
A: The second law of thermodynamics states that the total entropy of the universe always increases for ...
Q: The indicator phenolphthalein was added to the phosphoric acid solution prior to the titration. A g...
A: Given that the titration of phosphoric acid occurs. Phosphoric acid can be considered a relatively w...
Q: In which solvent—ethanol or diethyl ether—would the equilibrium for the following SN2 reaction lie f...
A: The equilibrium reaction provided is shown below.
Q: Compounds like CCl2F2 are known as chlorofluorocarbons,or CFCs. These compounds were once widely use...
A: Given The heat of vaporization of CCl2F2 is 289J/g. The mass of water is 200g. The heat of fusion of...
Q: A second-order reaction (2 A --> B) with a rate constant of 0.350 M-1 s-1 is found to have a half...
A: Since for the above 2nd order reaction, the relationship between the concentration and time is as sh...
Q: Most of the pKa values given in this text were determined in water. How would the pKa values of carb...
A: The solubility and dissociation of compound depend upon the type of solvent used for dissolution for...
Q: You have a gas mixture of O2 and N2 that has a total pressure of 2.2 atm. If the mole fraction of O...
A:
Q: The following figures represent distributions of two types of gas molecules between two containers c...
A: Since the entropy of system is maximum when the gas molecules occupies the complete volume in a symm...
Q: The balanced reaction between aqueous nitric acid and aqueous strontium hydroxide is __________.
A: The reaction is between acid and base Hence the product will be salt and water Hence HNO3 (aq)+ Sr...
Q: The reaction of an alkyl chloride with potassium iodide is generally carried out in acetone to maxim...
A: Since potassium iodide is soluble in water while potassium chloride is not, the alkyl chloride will ...
Q: can you show the mechanism?
A: The solvent TsOH is known as tosylic acid. It is categorized as a solid that remains soluble in H2O,...
Q: Four solutions of an acid dissolved in water are sketched below, as if under a microscope so powerfu...
A: A strong acid is the one which completely dissociates in water to give its conjugate base and hydron...
Q: The molecules have the same molecular formula (C3H8O) but differentchemical structures. (a) Which mo...
A: Propanol shown hydrogen bonding whereas, ethyl methyl ether does not show hydrogen bonding because n...
Q: What volume of 5.00 M NaOH stock solution is needed to prepare 100.0 mL of 0.250 M NaOH solution? ...
A: Since the moles of NaOH in final solution should be same as the moles of NaOH we are taking Hence as...
Q: Consider these compounds: A. BaF2 B. Ca(OH)2 C. AgI D. NiCO3 Complete the following statements b...
A: Given Data:
Q: What stereoisomers does the following reaction form?
A: In the following reaction alcohol acts as a nucleophile and it extracts the proton from HBr to cover...
Q: What is the IUPAC name for hydromorphone? Also, What are the functional groups are present in hydrom...
A: The IUPAC name for hydromorphone is 4,5-α-Epoxy-3-hydroxy-17-methyl morphinan-6-one.
Q: Phosgene (COCl2) is a potent chemical warfare agent that is now outlawed by international agreement....
A: Since initial [COCl2] = moles initial / volume = 0.01 / 10 = 0.001 M Assuming that y concentration ...
Q: A sugar solution contains 750 mg of sucrose in a volume of 1.5 L. What is the concentration of sucr...
A: The expression that is used to calculate concentration in mass by volume is shown below in which m i...
Q: If 35.0 ml of an aqueous H2SO4 solution requires 19.7 ml of a 0.71 M NaOH to reach the endpoint, wha...
A: Balanced equation is given by, H2SO4 (aq)+ 2 NaOH (aq)→ Na2SO4(aq) + 2 H2O(l)
Q: Using your knowledge of temperature and reaction rate, explain why food spoils at a slower rate when...
A: Any certain reaction's rate remains directly related to the temperature. If the temperature of any s...
Q: A small amount of another organic product is formed in a Williamson ether synthesis. What is this pr...
A: Please find below the reaction taking place
Q: A tank is first evacuated, and then connected to a 75.0 L cylinder of compressed nitrogen gas. The g...
A: Boyle's law explains that the pressure of the gas is inversely proportional to the volume of the gas...
Q: session.masteringchemistry.co This Mac can a problem wit Open iCloud pre <CHE154 S20 Ch17 Sec7-9 Lat...
A: Since the Ca(OH)2 salt is not completely soluble in water hence we need to take Ksp into account Sin...
Q: 7.) A student dissolved 5.00 g of Co(NO3)2 in enough water to make 100. mL of stocksolution. They to...
A: Molar mass of Co(NO3)2 is 183 g/mol Hence moles of Co(NO3)2 taken = mass / molar mass = 5 / 183 = 0....
Q: Find the solubility of AgI in 3.3 M NH3. (Ksp of AgI = 8.3 10-17 and Kf of Ag(NH3)2+ = 1.7 107)
A: Given, KSP for AgI = 8.3 × 10-17 KSP for Ag(NH3)2+ = 1.7 × 107 Concentration of NH3 = 3.3 M Let S ...
Q: Identify the substitution products that form when 2-bromo-2-methylpropane is dissolved in a mixture ...
A: On the basis of mechanism, chemical reactions can be classified as substitution reactions, addition ...
Q: Assume that, at 20oC, the vapor pressure of pure ethanol is 85.0 mmHg. What would the vapor pressure...
A: Given: Vapor pressure of pure ethanol = 85.0 mmHg. Mass of glycerin = 53.6 g Mass of ethanol = 133.7...
Q: I need help with quesion 2. Thanks.
A: The expression of entropy according to the statistical thermodynamics is, This statistical expressi...
Q: Which group 1 cation(s) can dissolve in a hot water bath?
A: According to qualitative analysis, group one cations are silver ion Ag+, lead ion Pb2+, and mercury ...
Q: A compound with formula C3H10O gives two signals only, both singlets, in the 'H NMR spectrum. Which ...
A: Given that there are only two signals for the compound and those signals are both singlets. Single...
Q: unlv.instructure.com Question 3 Using Hess' law, determine AH for the following reaction: CS2(1) + 3...
A: In accordance with Hess law, enthalpy change for any specific reaction following multiple steps is s...
Q: From the equation O2 + 2C = 2CO. If 2.73 moles of C are reacted with an excess of O2, How many moles...
A: The balanced equation is given below:
Q: estion 18 Write down the complete and net ionic equation for the following reaction SrBrz (aq) + K2S...
A: SrBr2 (aq) + K2SO4(aq) ----> SrSO4 (s) + 2KBr (aq) Above is the balanced reaction Since while w...
Q: Draw the substitution products for each of the following SN2 reactions. If the products can exist as...
A:
Q: Two patterns of packing two different circles of the samesize are shown here. For each structure (a)...
A:
Q: cis-1-Bromo-4-tert-butylcyclohexane and trans-1-bromo-4-tert-butylcyclohexane both react with sodium...
A: When the groups attached are either at 1,2-Cis or 1,4-Cis then the most stable product formed will b...
Q: Ascorbic acid (Vitamin C) is a diprotic acid.( H2C6H6O6). It ionizes as follows H2C6H6O6 (aq) &l...
A: Weak acid undergoes moderate dissociation and produce ions when it gets dissolved in aqueous solutio...
Q: Determine the standard free energy change at 25 °C for the reaction 2 H2(g) + O2(g) ---> 2 H2O(g)...
A: The standard thermodynamic data for the given reactants and products at given temperature is,
Q: Circle which of the following ions will be good leaving groups in sn1 and sn2 reactions?
A: Leaving group is the one that accepts a lone pair when the bond between it and its neighbouring grou...
how would I synthesize the product? Create a chloride compound?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images


