Our molecular model of matter describes a fluid as consisting of lots of little particles (atoms or molecules) moving around very fast. Collisions between the molecules makes the particles of the fluid change directions and speeds often and randomly. We've talked about two different phenomena that depend on these random molecular interactions: viscosity and diffusion. Viscosity is the way collisions with other parts of the fluid slow down faster-moving bits of fluid, and diffusion is the way collisions spread out concentrations of molecules. Let's consider how the viscosity and diffusion coefficients might depend on the properties of the fluid by dimensional analysis ▼ Part A ▼ What are the units of viscosity, in base SI units (kg, m, s)? kg m.s Submit ✓ Correct Part B Previous Answers What are the units of the diffusion coefficient, in base I units (kg, m, s)? Submit ✓ Correct Previous Answers V
Our molecular model of matter describes a fluid as consisting of lots of little particles (atoms or molecules) moving around very fast. Collisions between the molecules makes the particles of the fluid change directions and speeds often and randomly. We've talked about two different phenomena that depend on these random molecular interactions: viscosity and diffusion. Viscosity is the way collisions with other parts of the fluid slow down faster-moving bits of fluid, and diffusion is the way collisions spread out concentrations of molecules. Let's consider how the viscosity and diffusion coefficients might depend on the properties of the fluid by dimensional analysis ▼ Part A ▼ What are the units of viscosity, in base SI units (kg, m, s)? kg m.s Submit ✓ Correct Part B Previous Answers What are the units of the diffusion coefficient, in base I units (kg, m, s)? Submit ✓ Correct Previous Answers V
Physics for Scientists and Engineers: Foundations and Connections
1st Edition
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Katz, Debora M.
Chapter15: Fluids
Section: Chapter Questions
Problem 77PQ
Related questions
Question
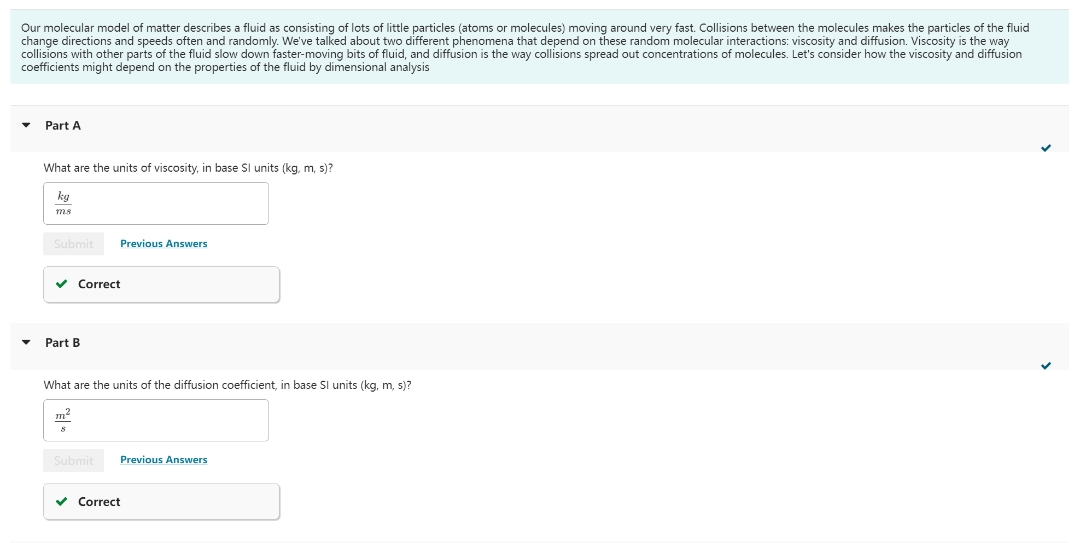
Transcribed Image Text:Our molecular model of matter describes a fluid as consisting of lots of little particles (atoms or molecules) moving around very fast. Collisions between the molecules makes the particles of the fluid
change directions and speeds often and randomly. We've talked about two different phenomena that depend on these random molecular interactions: viscosity and diffusion. Viscosity is the way
collisions with other parts of the fluid slow down faster-moving bits of fluid, and diffusion is the way collisions spread out concentrations of molecules. Let's consider the viscosity and diffusion
coefficients might depend on the properties of the fluid by dimensional analysis
▼
Part A
What are the units of viscosity, in base SI units (kg, m, s)?
kg
ms
Submit
✓ Correct
Part B
Previous Answers
What are the units of the diffusion coefficient, in base SI units (kg, m, s)?
m²
8
Submit
Previous Answers
✓ Correct
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University