Part B: Conductivity of pure water, solids and solutions. Substance Conductivity Substance Conductivity Pure water, H20 (1) Pure water NaCl (s) C12H22O1 (s) NaCl (aq) C12H2201 (aq) Diluted NaCl (aq) Diluted C12H22011 (aq) Most Diluted NaCl (aq) Most Diluted Cı2H22O11 (aq) Substance Conductivity Substance Conductivity Pure water Pure water KNO3 (s) Ca(C2H3O2)2 (s) KNO: (aq) Ca(C2H3O2)2 (aq) Diluted KNO3 (aq) Diluted Ca(C2H,O2)2 (aq) Most Diluted KNO3 (aq) Most Diluted Ca(C2H3O2)2 (aq)
Part B: Conductivity of pure water, solids and solutions. Substance Conductivity Substance Conductivity Pure water, H20 (1) Pure water NaCl (s) C12H22O1 (s) NaCl (aq) C12H2201 (aq) Diluted NaCl (aq) Diluted C12H22011 (aq) Most Diluted NaCl (aq) Most Diluted Cı2H22O11 (aq) Substance Conductivity Substance Conductivity Pure water Pure water KNO3 (s) Ca(C2H3O2)2 (s) KNO: (aq) Ca(C2H3O2)2 (aq) Diluted KNO3 (aq) Diluted Ca(C2H,O2)2 (aq) Most Diluted KNO3 (aq) Most Diluted Ca(C2H3O2)2 (aq)
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter13: The Chemistry Of Solutes And Solutions
Section: Chapter Questions
Problem 115QRT
Related questions
Question
https://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4
I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this lab
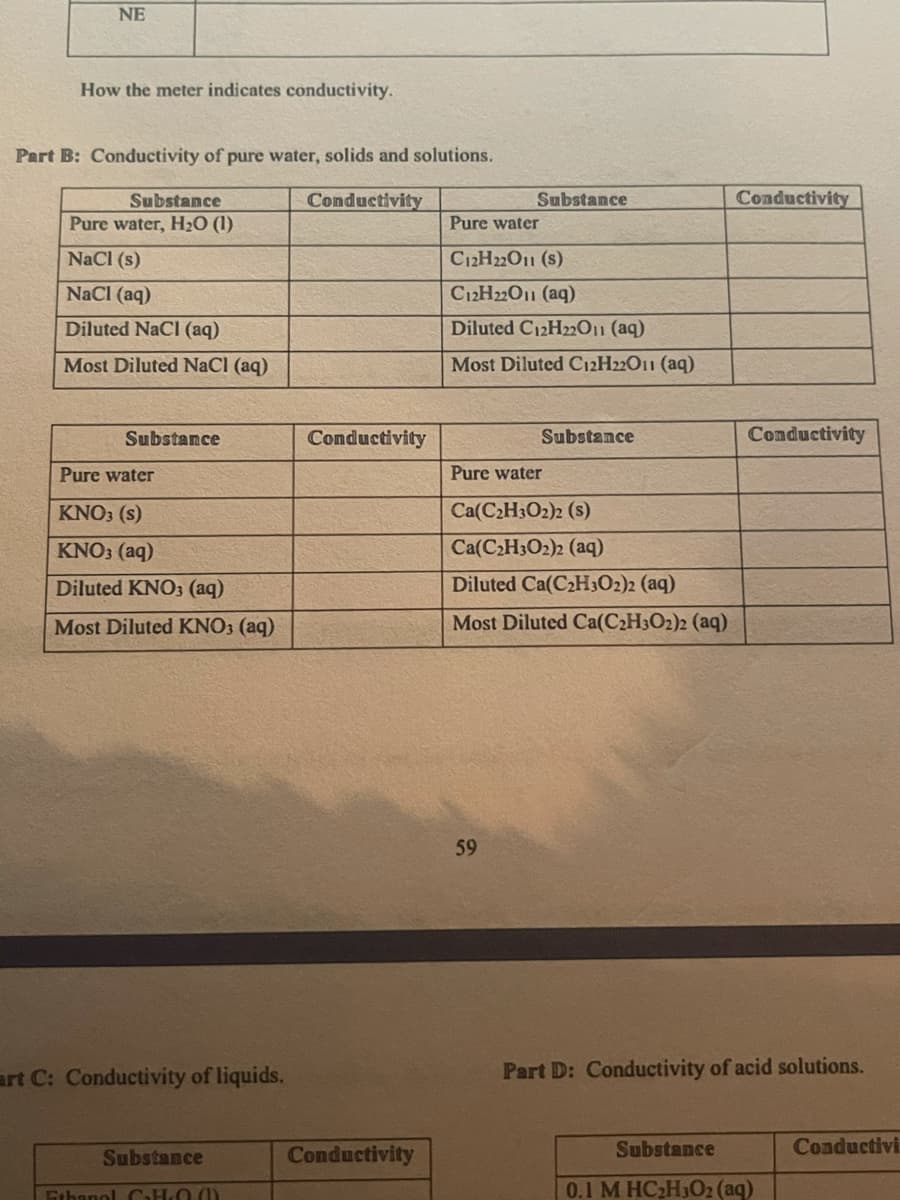
Transcribed Image Text:NE
How the meter indicates conductivity.
Part B: Conductivity of pure water, solids and solutions.
Substance
Conductivity
Substance
Conductivity
Pure water, H2O (1)
Pure water
NaCl (s)
C12H22O1 (s)
NaCl (aq)
C12H22011 (aq)
Diluted NaCl (aq)
Diluted C12H22011 (aq)
Most Diluted NaCl (aq)
Most Diluted C12H22O11 (aq)
Substance
Conductivity
Substance
Conductivity
Pure water
Pure water
KNO3 (s)
Ca(C2H3O2)2 (s)
KNO3 (aq)
Ca(C2H3O2)2 (aq)
Diluted KNO3 (aq)
Diluted Ca(C2H3O2)2 (aq)
Most Diluted KNO3 (aq)
Most Diluted Ca(C2H3O2)2 (aq)
59
art C: Conductivity of liquids.
Part D: Conductivity of acid solutions.
Substance
Conductivity
Substance
Conductivi
Ethanol CHO ()
0.1 M HC2H302 (aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning