Part C A 53.5 mL sample of an 9.0 % (m/v) KBr solution is diluted with water so that the final volume is 231.0 mL. Express your answer to two significant figures and include the appropriate units. HẢ ? Value Units Submit Request Answer Part D A9.0 mL sample of a 42.0 % (m/v) acetic acid (HC,H3O2) solution is added to water to give a final volume of 48 mL. Express your answer to two significant figures and include the appropriate units. HẢ ? Val
Part C A 53.5 mL sample of an 9.0 % (m/v) KBr solution is diluted with water so that the final volume is 231.0 mL. Express your answer to two significant figures and include the appropriate units. HẢ ? Value Units Submit Request Answer Part D A9.0 mL sample of a 42.0 % (m/v) acetic acid (HC,H3O2) solution is added to water to give a final volume of 48 mL. Express your answer to two significant figures and include the appropriate units. HẢ ? Val
Chapter20: Applications Of Oxidation/reduction Titrations
Section: Chapter Questions
Problem 20.25QAP
Related questions
Question
Transcribed Image Text:Course Home
Courses
Ô https://openvellum.ecollege.com/course.html?courseld=16516363&OpenVellumHMAC=6607995b61ef1ebc798fc86a6680d4f.
Syllabus
I Review | Constants | Periodic Table
Scores
Calculate the final concentration of each of the
Part C
following.
eТеxt
You may want to reference
(Pages 306 - 308) Section 9.5 while completing this
problem.
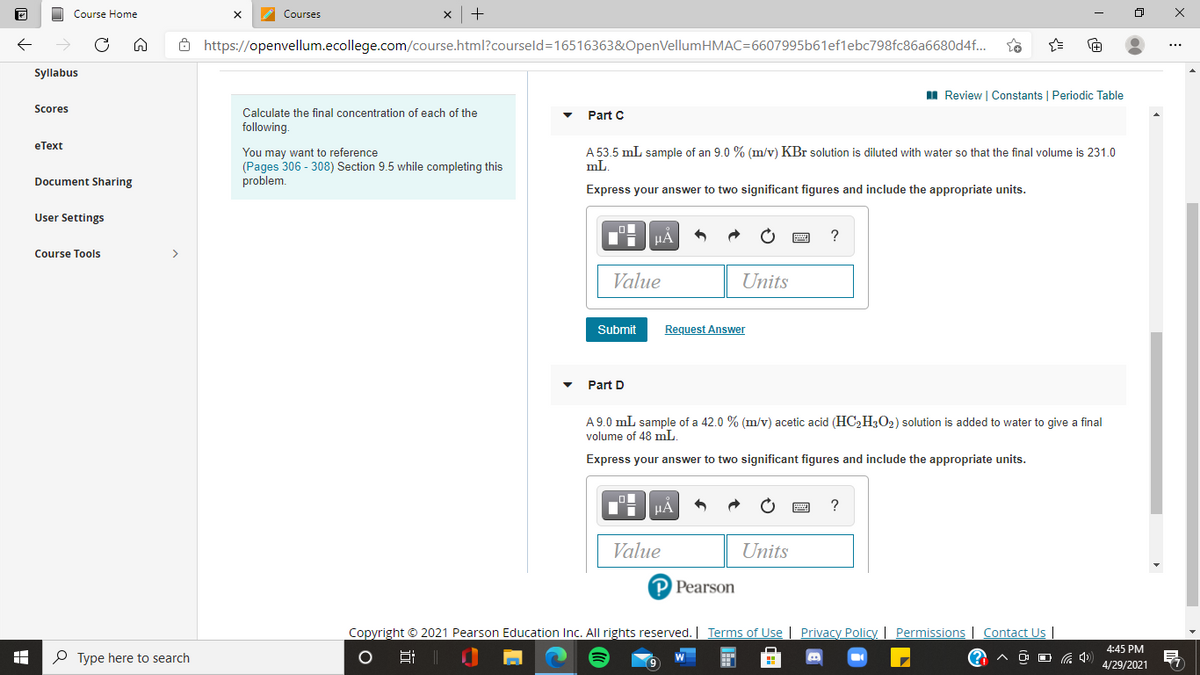
A 53.5 mL sample of an 9.0 % (m/v) KBr solution is diluted with water so that the final volume is 231.0
mL.
Document Sharing
Express your answer to two significant figures and include the appropriate units.
User Settings
HẢ
?
Course Tools
Value
Units
Submit
Request Answer
Part D
A 9.0 mL sample of a 42.0 % (m/v) acetic acid (HC2H3O2) solution is added to water to give a final
volume of 48 mL.
Express your answer to two significant figures and include the appropriate units.
HẢ
?
Value
Units
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy. | Permissions | Contact Us |
4:45 PM
P Type here to search
(?
4/29/2021
Transcribed Image Text:Course Home
Courses
Ô https://openvellum.ecollege.com/course.html?courseld=16516363&OpenVellumHMAC=6607995b61ef1ebc798fc86a6680d4f.
Syllabus
I Review | Constants | Periodic Table
Scores
Calculate the final concentration of each of the
following.
Part A
eТеxt
You may want to reference
(Pages 306 - 308) Section 9.5 while completing this
problem.
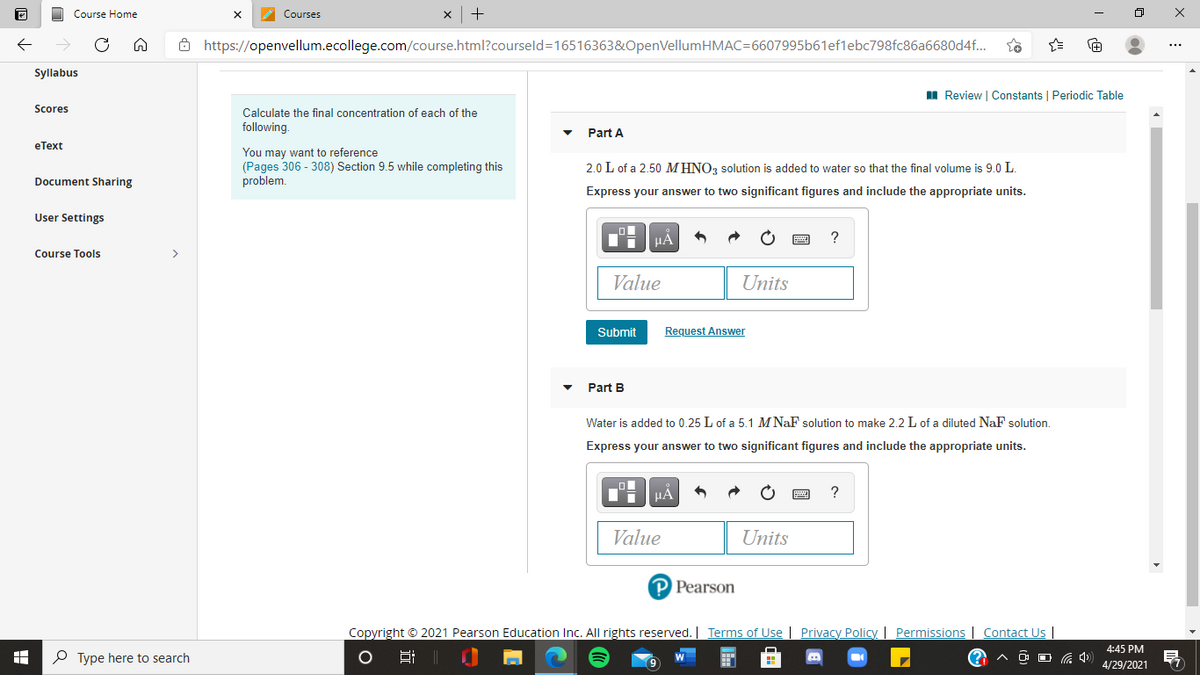
2.0 L of a 2.50 MHNO, solution is added to water so that the final volume is 9.0 L.
Document Sharing
Express your answer to two significant figures and include the appropriate units.
User Settings
HA
?
Course Tools
Value
Units
Submit
Request Answer
Part B
Water is added to 0.25 L of a 5.1 M NaF solution to make 2.2L of a diluted NaF solution.
Express your answer to two significant figures and include the appropriate units.
T'i HẢ
?
Value
Units
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy. | Permissions | Contact Us |
4:45 PM
P Type here to search
(?
4/29/2021
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning