Percent weight of material A 100 75 50 25 Melting temperature of material A Liquid Liquid and solid Melting temperature of material B Solid 25 50 75 100 Percent weight of material B FIGURE P2.31 Temperature
Percent weight of material A 100 75 50 25 Melting temperature of material A Liquid Liquid and solid Melting temperature of material B Solid 25 50 75 100 Percent weight of material B FIGURE P2.31 Temperature
Chapter2: Loads On Structures
Section: Chapter Questions
Problem 1P
Related questions
Question
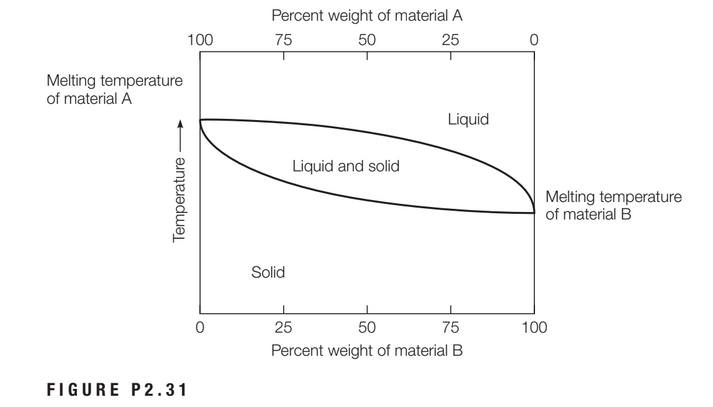
Considering an alloy of the two soluble components A and B described by a phase diagram Figure P2.31, determine the masses of the alloy that are in the liquid and solid phases at a given temperature if the total mass of the alloy is 100 g, component B represents 60% of the alloy, 25% of the liquid is component B, and 70% of solid is component B.
Transcribed Image Text:Percent weight of material A
100
75
50
25
Melting temperature
of material A
Liquid
Liquid and solid
Melting temperature
of material B
Solid
25
50
75
100
Percent weight of material B
FIGURE P2.31
Temperature
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, civil-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you


Structural Analysis (10th Edition)
Civil Engineering
ISBN:
9780134610672
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Principles of Foundation Engineering (MindTap Cou…
Civil Engineering
ISBN:
9781337705028
Author:
Braja M. Das, Nagaratnam Sivakugan
Publisher:
Cengage Learning


Structural Analysis (10th Edition)
Civil Engineering
ISBN:
9780134610672
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Principles of Foundation Engineering (MindTap Cou…
Civil Engineering
ISBN:
9781337705028
Author:
Braja M. Das, Nagaratnam Sivakugan
Publisher:
Cengage Learning

Fundamentals of Structural Analysis
Civil Engineering
ISBN:
9780073398006
Author:
Kenneth M. Leet Emeritus, Chia-Ming Uang, Joel Lanning
Publisher:
McGraw-Hill Education


Traffic and Highway Engineering
Civil Engineering
ISBN:
9781305156241
Author:
Garber, Nicholas J.
Publisher:
Cengage Learning