১ PIEL SANA Con ALOE VERA LIMPIEZA DE MANOS SIN AGUA GEL HYGIENQUE POUR MAINS GEL HIGIENICO PARA MAOS HYGIENIC HANDS GEL BY Me BY BYzy Fe Birg Al Sp AtClo ,BrzAeBxz Br
১ PIEL SANA Con ALOE VERA LIMPIEZA DE MANOS SIN AGUA GEL HYGIENQUE POUR MAINS GEL HIGIENICO PARA MAOS HYGIENIC HANDS GEL BY Me BY BYzy Fe Birg Al Sp AtClo ,BrzAeBxz Br
Chapter9: Energy For Today
Section: Chapter Questions
Problem 9E
Related questions
Question
Is my proposed synthesis correct in the following picture?
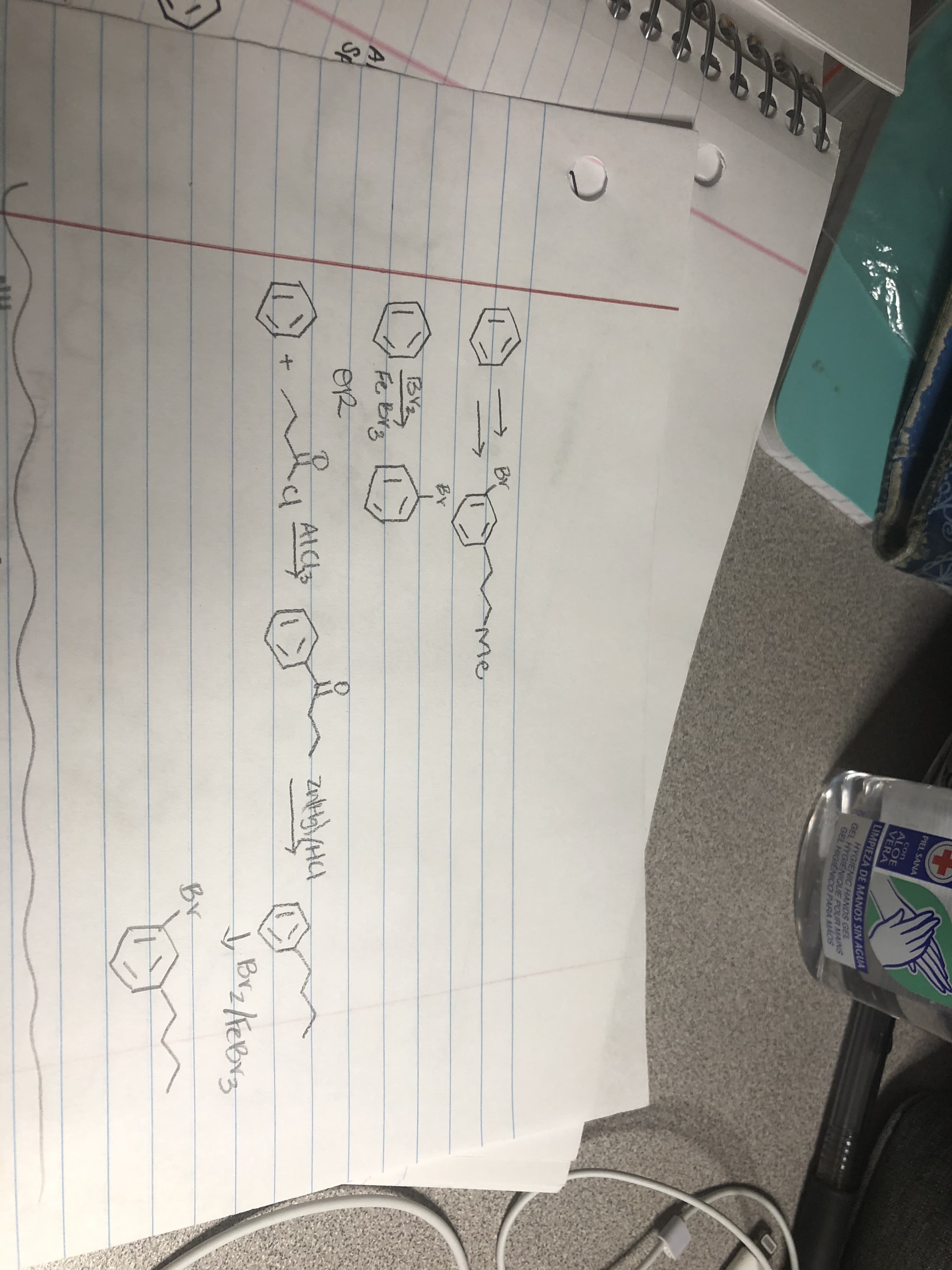
Transcribed Image Text:১
PIEL SANA
Con
ALOE
VERA
LIMPIEZA DE MANOS SIN AGUA
GEL HYGIENQUE POUR MAINS
GEL HIGIENICO PARA MAOS
HYGIENIC HANDS GEL
BY
Me
BY
BYzy
Fe Birg
Al
Sp
AtClo
,BrzAeBxz
Br
Expert Solution
Step 1
In the given reaction, reduction occurs at the last step in mechanism. The correct sequence the given reaction is as follows:
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

