Q: Q2- How mainy mls of Atropine sulphate present in lab. As a stock sol. Of 1: 20 conc. should be used…
A: 1:20 concentration indicate that 20 mL of stock solution contains 1 g of atropine sulphate.
Q: 1. Calculate the mass, in grams, of sodium hydroxide needed to prepare 500 mL of approximately 0.1 M…
A: Mole = Molarity × Volume in litre Molar mass NaoH = 39.997g/mol.
Q: e) How long (in seconds) will it take for a 30% NH,NO, solution to become a 19% NH,NO2 solution…
A: Solution - According to the question - We need two times: and Calculating using equation above:…
Q: ater? nk in water?
A: 1) heptane is seven membered alkane. Condensed structure formula is CH3CH2CH2CH2CH2CH2CH3 2) As…
Q: | Complete the data table below. Concentration of HA pH [H]= X from an ice Percent Ionization table…
A: Solution : I- Initial concentration C - Change E -Equilibrium
Q: Calculate the heat of hydration for silver nitrate. ΑΣφ ? AHnydration k II
A: Total Energy needed for completion of the reaction is known as enthalpy of the reaction
Q: In an experiment, Milabel were tasked to prepare 200.00 mL of 3.5 M solution from a concentrated…
A:
Q: Number of moles of sodium sulfate dissolved in 2 L of a 0.12 M sulfate, Na2SO4solution
A: Given :- molarity of Na2SO4 solution = 0.12 M Volume of solution = 2 L To calculate…
Q: A concentrated phosphoric acid solution is 85.5%H 3 PO 4 by mass and has a density of 1.69g / m * L…
A:
Q: What volume of 9 M HCl must be used to prepare 1.5 L of 0.25 M HCl?
A: The volume of 9 M HCl that must be used to prepare 1.5 L of 0.25 M HCl is to be determined.
Q: Calculate the amount of conc. HCl
A:
Q: 11) How much 0.05 M HCl solution can be made by diluting 250 mL of 10 M HCI?
A: Dilution decreases the concentration of the solution that is same moles are distributed over the…
Q: Prepare 100 ml each of 0.1 M HCL and 0.1 M NaOH.
A: Molarity is a way of expressing concentration in which moles of solute present per liter of solution…
Q: Solution 1) 50.00mL of a solution containing 0.15M malonic acid, CH2(CO2H)2, and 0.020M manganese…
A:
Q: A concentrated ammonia solution is 17.4 M ammonium hydroxide (NH4OH). How do you prepare a 250 mL of…
A: Given: Concentration of concentrated ammonia solution = 17.4 M Concentration of diluted ammonia…
Q: How would u prepare 10ml of a 0.25% m/v HCI solution of 1% m/v HCI was available? How much 1% m/v…
A:
Q: Describe how you will prepare 100 mL of 1 M HCl from concentrated HCl. Show calculations below.
A: By dissolving 3.65g of HCl in 100mL water we can make 1M HCl solution.
Q: Water is known as the universal solvent. What property of water allows it to dissolve ions such as…
A: Water is called the universal solvent because it is capable of dissolving more substances than any…
Q: Which solution is more concentrated: a 1 MH2SO4 solution or a 1 N H2SO4 solution? Explain why. 9.
A: Which solution is more concentrated a 1 M H2SO4 solution or a 1 N H2SO4 solution? Explain why.
Q: Which of the following statement/s is/are correct? 1. 3.00 N H2SO4 is equivalent to 6.00 M H2SO4.…
A:
Q: Which of the following is the least soluble in water? Select one: O a. NaCl (sodium chloride) O b.…
A: The NaCl which is commonly known as sodium chloride is strongly soluble in water on dissolving and…
Q: How many grams of Acetic acid are needed in 10L solution of 0.1M concentration?
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Calculate the boiling point (in oC) of an aqueous solution which is 0.347 m in potassium carbonate.…
A: Answer:- This question is answered by using the simple concept of the elevation in the boiling point…
Q: What is the equilibrium concentration of CH3 COOH if 0.150 moles each of CH3 COOH and NaOH are added…
A:
Q: We mix 25 mL of 10 M HCl(aq) with 15 mL of 10 M KOH(aq). If both solutions are initially at 85 ∘C,…
A: From given The amount of heat liberated from the reaction of 25 mL of 10 M HCl (aq) with 15 mL of 10…
Q: How many grams of sodium bromide do you need to prepare a 1 liter solution of 0.050M NaBr? Show your…
A:
Q: a. Reaction with NaOH NaOH OH NaOH -OH b. Reaction with NaHCO3 NaHCO3 `OH c. Reaction with HCI HCI…
A: Since you have submitted multiple subparts, we will solve the first three subparts for you. Kindly…
Q: 12. In each pair, predict which would have a greater percentage of hydrate in water and explain. 12…
A: Hydration of a molecule depends upon extent of polarization of molecule.
Q: How much water should be added to 14mL of 15% alcohol solution to reduce the concentration to 7%?
A: Given, V1 = 14 ml M1 = 15% V2 = ? M2 = 7 %
Q: Explain how Compound A and Compound B will dissolve in water using the correct vocabulary including…
A: We are given two compounds; NaCl and CO2.
Q: Prepare 100 ml each of 0.1 M HCL and 0.1 M NaOH
A: Molarity is a way of expressing concentration which indicates the moles of solute present per liter…
Q: Calculate the molarities of the following solutions 7. 2.3 moles of sodium chloride in .45 liters of…
A: Given, here7) number of moles of sodium chloride =2.3 molevolume of solution ( water ) =0.45 L8)…
Q: a) The vapor pressure of ethanol is 100 mmHg at 34.9°C. Determine the vapor pressure at 63.5°C…
A: Answer a) Initial pressure,P1 = 100 mm Hg Final pressure, P2 = ? Initial temperature, T1 = 34.9 °C =…
Q: What is the partial pressure of carbon dioxide in a container that holds 5 moles of carbon dioxide,…
A:
Q: Kb for NH 3 = 1.8 x 10 and Ka for HF = 3.5 x 10 -4 -4 Which type of salt is NH 4F? %3D
A: Since the strength of acid and base is proportional to the value of Ka and Kb respectively Hence…
Q: How many grams of sodium hydroxide (M.wt-58.5) can be added to 2.5 liter of a solution mixture of…
A: Given information, M.wt of NaOH = 58.5 g/mol Volume of mixture = 2.5 L Concentration of ammonia =…
Q: If water was used to rinse the pipette used for the NaOH solution aliquots, how would this affect…
A: Rinsing a pipette:- It is always advised that first, we shall rinse the pipette with distilled water…
Q: Phosphoric acid is usually obtained as an 85% phosphoric acid solution. If it is 15M.Calculate: a.…
A:
Q: How many grams of Acetic acid are needed in 10L solution .1 ?of 0.1M concentration
A: Given Volume of solution = 10L Molarity of solution = 0.1 M
Q: Caffeine In water @ 100°C
A:
Q: Give a clear explanation handwritten answer of each compound....
A: Solubility Product is the dynamic equilibrium that exists when a chemical compound in solid state is…
Q: Calculate the morality of a solution prepared by dissolving 11.5g of solid NaOH in enough water to…
A: Molarity is defined as Molarity = moles of solute / volume of solution in L Volume of solution…
Q: 1. How many ml of NaOH in preparing 50 g of it in 1N of solution (Na- 23, O -16, H-1)
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Describe how you would prepare 1 litre of 0.1M NaOH using NaOH pellets.
A: To prepare the 1 L of 0.10 M of NaOH solution, 0.1 mol of NaOH has to be added into the solution.…
Q: What volume of 0.25 M HCl must be diluted to prepare 0.96 L of 2.0×10-²M HCl? L
A:
Q: Prepare 250mL of a 0.75M solution of phosphoric acid from 75.0% phosphoric acid that is pure. The…
A:
Q: How would you prepare 1.50 L of a 0.70-M solution of each of the following? NiCl2 from the salt…
A: To find: weight of NiC12.6H2O We know that Molecular weight of NiC12.6H2O is: 237.69
Q: What volume of 0.25 M HCl must be diluted to prepare 1.4 L of 7.0×10-2M HCl? _________ L
A: Molarity of a solution is used to express the concentration of the unknown solution. It can be…
Q: H Which of these statements are true for a neutral, aqueous solution at 25 °C? [H+] = [OH-] pH =…
A: Given, The true statement for a neutral, aqueous solution at 25 °C is:
Write the balanced chemical equations for the reactions that occur when each possible hydrated salt is heated. Include the chemical names of each hydrate and anhydrous salt. Hint: Remember, Hydrate + Heat
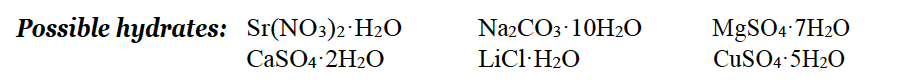
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- A natural product (MW = 150) distills with steam at a boiling temperature of 99°C at atmospheric pressure. The vapor pressure of water at 99°C is 733 mm Hg.a. Calculate the weight of the natural product that codistills with each gram of ater at 99°C.b. How much water must be removed by steam distillation to recover this natural product from 0.5 g of a spice that contains 10% of the desired substance?Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW: Na: 23, K: 39, Cl: 35, H: 1, C: 12, O: 16) Calculate the total amount of chloride expressed in mmol/L present in the prepared solution 60.34 mmol/L 90.10 mmol/L a 111.11 mmol/L b 29.76 mmol/L c 80.61 mmol/L d 20.27 mmol/L1 a) what volume of 3.0M nitric acid would be needed inorder to prepare 150.0ml of 1.3M nitric acid? b) what volume of 6.11M sulfuric acid would be needed to prepare 250.0ML of 1.3M sulfuric acid c) what volume of 2.5M potassium chloride would be needed inorder to prepare 175.0ML of 0.42M potassium chloride.
- 37 - Which of the following is considered a STRONG electrolyte? A) NH4NO3 B) PbCl2 C) HC2H3O2 D) CH3OH E) C12H22O11How would you prepare 1.5 L of approximately 0.1 M KMnO4?Calculate grams of sucrose that must be added to 112.5 grams of water to prepare a 12.5 % by mass solution. a. 32.1 b. 16.1 c. 8.04 d. 0.244 e. 48.2
- Starting with the solid and adding water , how would you prepare 2.00L of 0.685MNi(NO3)2 CuCl2C6H8O6 ( vitamin C)8. Paraform precipitate is formed in formaldehyde solution when stored at a temperature: A) Below + 18 ° С B) Above + 18 ° С C) Above + 9 ° C. D) Below + 9 ° C.The number of moles of H2O produced in reaction of 50 mL 1.0 M CH3COOH and 50 mL 1.0 M NaOH.

