PRINTER VERSION 1 BACK FULL SCREEN NEXT CALCULATOR BASIC STOICHIOMETRY T01/S01 Based on the balanced equation, Molar Mass (g/mol) Mg + Cl2 - M9CI2 24.305 Mg Cl2 MgCl2 Avogadro's No. 6.022x1023 mol1 calculate the molecules of CI, required when 91 formula units of MgCl, are formed. 70.906 95.211 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
PRINTER VERSION 1 BACK FULL SCREEN NEXT CALCULATOR BASIC STOICHIOMETRY T01/S01 Based on the balanced equation, Molar Mass (g/mol) Mg + Cl2 - M9CI2 24.305 Mg Cl2 MgCl2 Avogadro's No. 6.022x1023 mol1 calculate the molecules of CI, required when 91 formula units of MgCl, are formed. 70.906 95.211 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter8: Chemical Composition
Section: Chapter Questions
Problem 128CP: itamin B12 , cyancobalamin, is essential for human nutrition. Its molecular formula is...
Related questions
Question
Please provide detailed steps as I am using this to study, along with explanations as to what you did on each step
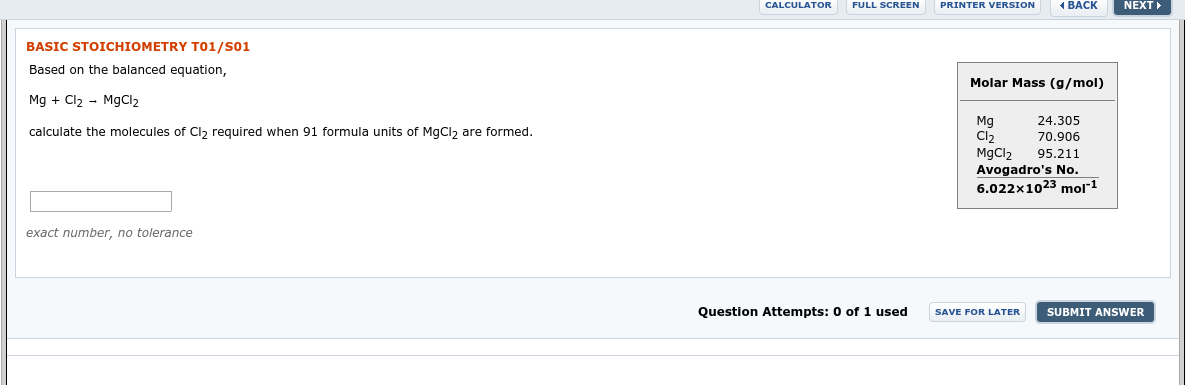
Transcribed Image Text:PRINTER VERSION
1 BACK
FULL SCREEN
NEXT
CALCULATOR
BASIC STOICHIOMETRY T01/S01
Based on the balanced equation,
Molar Mass (g/mol)
Mg + Cl2 - M9CI2
24.305
Mg
Cl2
MgCl2
Avogadro's No.
6.022x1023 mol1
calculate the molecules of CI, required when 91 formula units of MgCl, are formed.
70.906
95.211
exact number, no tolerance
Question Attempts: 0 of 1 used
SUBMIT ANSWER
SAVE FOR LATER
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 6 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning