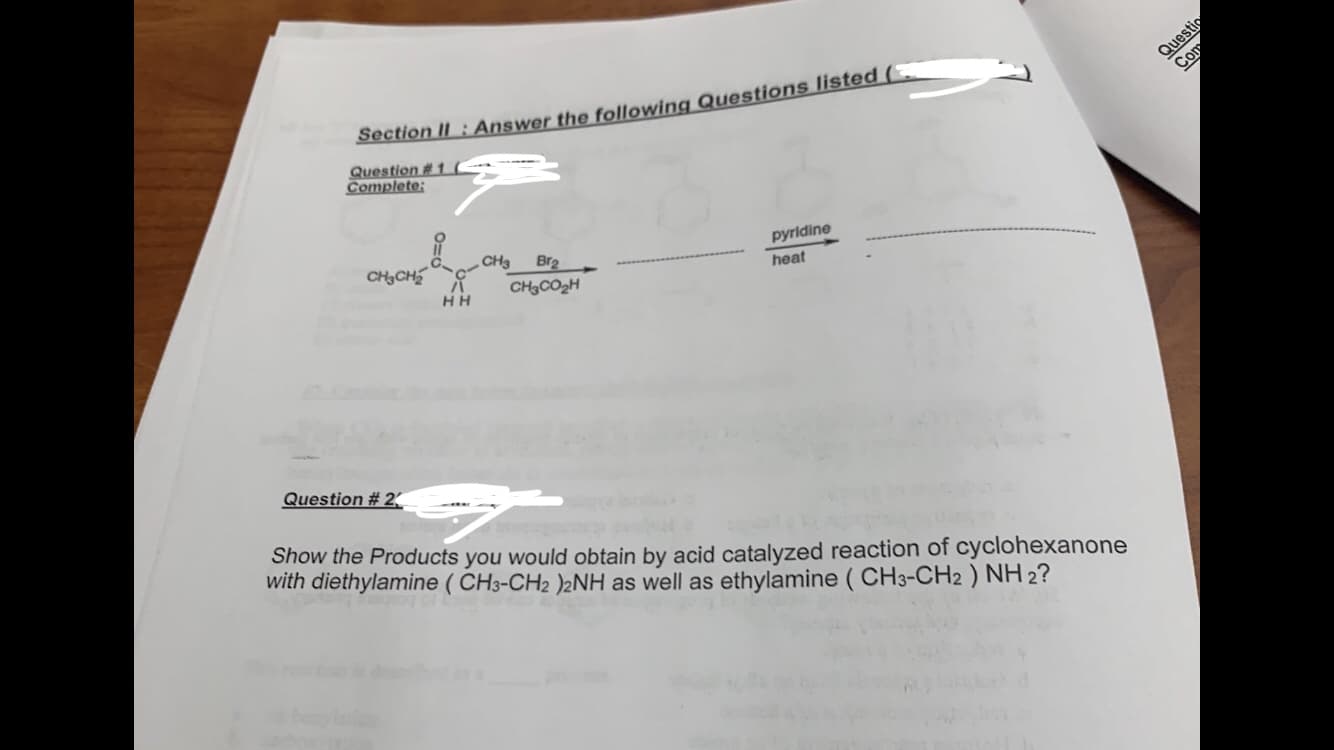
pyridine heat снэсогн НН Question # 2 Show the Products you would obtain by acid catalyzed reaction of cyclohexanone with diethylamine (CH3-CH2 )2NH as well as ethylamine (CH3-CH2) NH2?
Q: H. CrO3 Aqueous Acetone Heat 1. HCI 2. НО Na tBuO Br Нeat
A:
Q: One synthetic advantage of olefin metathesis is that the catalyst tolerates a variety of functional…
A: Alkene metathesis is the exchange of substituents across the double-bonded parent alkenes. There is…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. lithium…
A: Given starting material is ketone.
Q: The alkene shown below is treated sequentially with ozone (O3) and zinc/acetic acid. Draw structural…
A: The above reaction is addition of ozone referring as ozonolysis.
Q: The reaction of 2-methylpropene with HBr in ether is described below. Answer the following questions…
A: 1.) In this reaction , HBr adds on double bond to give a single bond . In this reaction , first H+…
Q: S g) h) OH OH PCC PCC CH₂Cl₂ CH₂Cl₂ K₂Cr₂O7 H₂SO4 H₂O Excess K₂Cr₂O7 H₂SO4 H₂O OH
A: 1). Reagent - PCC, CH2Cl2 PCC (Pyridinium chlorochromate) oxidize alcohols into Carbonyls. PCC…
Q: HURRY ASAP I WILL RATE NO NEED FOR EXPLANATION WHICH OPTIN Which one of the following alcohols can…
A:
Q: 5. Which among the following compounds can show E-Z system. a. 3-bromo-2-chloropent-2-ene b.…
A: The objective of the question is to find out which among the given compounds can show E-Z system.
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. lithium…
A: The incomplete reaction is given as follows:
Q: Find the TRUE statement(s) among the choices below. You may choose one or more statements. An…
A: Here we have to determine the true statement from the following options.
Q: Identify reagents that can be used to complete the following transformation. OH O BH3-THF O 1)…
A:
Q: QUESTION 20 Which observation is NOT consistent with an SN1 mechanism for the conversion alkyl…
A:
Q: tng with sodium azide as your source of nitrogen and using any other reagents of your choice, show…
A: ->NaN3 can acts as nucleophile and can give nucleophilic substitution reaction. ->Reducing…
Q: Which substance would undergo the following reaction? H i. O3 ii. Zn, HOAC Select one: O…
A: These reactions are the well examples of the ozonolysis and reduction reaction where alkenes and…
Q: QUESTION 5 Show how you would synthesis the following compounds starting from benzene? Write the…
A: We have to synthesise the given compound starting from Benzene. We have to make use of different…
Q: Question 28 In what order would you use the reagents given to transform compound A to compound B.…
A: In organic chemistry, inter conversion of organic molecule takes place from one form to another form…
Q: w the product(s) for the following reaction. (Z)-3-hexene + MCPBA nich reagent(s) and/or…
A: 1) (Z) - 3- hexene reaction with mCPBA 2) Ring opening of epoxide
Q: The most common reducing agent used for the conversion of ester into aldehyde. * a-H2/Ni, Heat…
A: esters are converted into aldehyde by DIBAL-H Option"d" is correct.
Q: Which of the reagents below could be used to convert 2-pentanol to 2-chloropentane. heat ? `CO2Et а.…
A:
Q: Select the expected major product of the reaction shown 1. LDA, -78 C 3-methyl-4-hexanone 2. CH3CH2I…
A: LDA is a good base, so it takes hydrogen atom from carbon, now an enolate is formed. This reaction…
Q: State the reaction happening in each step of the synthesis sequence shown below. reaction type…
A:
Q: Which of the following reactions does NOT give 3-methylbutan-2-ol as the main product? Select all…
A: Given target product is, 3-methylbutan-2-ol
Q: show the steps for the synthesis of the two products below. the starting material is benzene.
A: Since you have asked multiple questions, we will solve first one for you. For the remaining one,…
Q: Select the carbon in the structure below that would be lost as CO2 after treatment with the…
A:
Q: Select the best reagent expected to convert 3-heptyne to cis-3-heptene. A. NaNH2, NH3 B. Na, NH3…
A: Given We know about the alkyne reaction Select the best reagent expected to convert 3-heptyne to…
Q: ng déscribes the final major product of the reaction of 2-propanone with excess methanol and…
A: Aldehyde and ketones reacts with alcohol in presence of catalyst HCl.
Q: Practice o14 For each of the following, provide the product or reagent as necessary H20 NABH4 +…
A:
Q: E Module 13 HW Chapter 21 - Selected Exercises Question 2 of 9 -/1 E Identify the reagents you…
A:
Q: Which of the following are true? Chose all the correct answers. O 1,3-Cis disubstituted cyclohexane…
A: When the groups are present on the equotorial position are more stable than axial position. In axial…
Q: View Policies Current Attempt in Progress Propose an efficient synthesis for the following…
A: Remove the double bond using hydrogen bromide and expand the carbon chain with CHCNa and make…
Q: catalytic hydrogenation ( or deuteration) adds H2 (or D2) by a syn electrophilic addition. Which…
A: The product of syn-addition of D2 to (E)-2-pentene and (Z)-2-pentene is to be determined.
Q: From the table of available reagents select the one(s) you would use to convert 2-phenylethanol to…
A: In this question, we want to prepare following two products from the starting material…
Q: CH3 1 mole Br, H3C F (Show stereochemistry of product)
A:
Q: Show how the Suzuki and'or Heck reactions can be used to prepare the following compounds: Part A…
A: Note : Suzuki reaction : Boronic acid + bromo benzene. Heck reaction : alkene + bromo benzene.
Q: Which of the following compounds is expected to be a major product of the following reaction? Select…
A:
Q: Which of the following reactions form the product shown. 2 N2OH 1. NaH O BrCH,CH,Br 2. BrCH CH-Br…
A: Given, NaH ( sodium hydride) is a strong base. NaOH (sodium hydroxide) is a base. Hydronium ion…
Q: Given the set of compounds below: isopentane 4-methylcyclopent-1-ene toluene Which compound/s will…
A:
Q: Provide a synthesis of the following molecule starting from toluene. CH3 ber CH3 Draw the 4 possible…
A:
Q: a. Which of the following mechanistic steps makes the most sense given the structures and charges…
A: A reaction will proceed in the forward direction if it form a stable intermediate or transition…
Q: Paragraph Styles Edi 7. Answer ALL parts of the question (a) Predict the organic products formed…
A:
Q: d) 2. H20i OH Cold HO, mC PRA, Places OH groues wits Same Skerochmi sty. HO TSCI OH e) gets rid of…
A: The question is based on the concept of organic reactions. we have to identify the reagents…
Q: Which one of the structures below represents the major product obtained from the reaction shown in…
A: we have to predict the product form for the given reaction
Q: Arrange the following compounds in increasing reactivity with SN1 mechanism. Type the letter labels…
A: Here we are required to arrange the compound in the increasing order of reactivity toward SN1…
Q: Parsol®MCX (see structure below) is a popular UV B sunscreen used in many formulations. How would…
A: Applying concept of wittig reaction.
Q: Propose the structure for hydrocarbon that gives the following products from ozonolysis reaction.…
A: Alkenes can undergo ozonolysis to form alcohols, aldehydes, ketones. Alkynes undergo ozonolysis to…
Q: Q2(17 Marks): outline all steps in a possible laboratory synthesis of the following compounds: (…
A: Given: conversion To find: To carry out all the above conversion which are given. Solution: To…
Q: Draw the major organic product of the reaction shown. (CH3CH2)3N (CH3)2CHCH,CHCOZ…
A: The reactants given in the reaction shown are leucine and di-tert-butyl dicarbonate. The reagent…
Q: What are the products of the following reaction? Would you expect them to have higher or lower Amax…
A: Hello dear, welcome on bartleby; The value of λmax depends upon conjugation, on increasing…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Several sulfonylureas, a class of compounds containing RSO2NHCONHR, are useful drugs as orally active replacements for injected insulin in patients with adult-onset diabetes. These drugs decrease blood glucose concentrations by stimulating b cells of the pancreas to release insulin and by increasing the sensitivity of insulin receptors in peripheral tissues to insulin stimulation. Tolbutamide is synthesized by the reaction of the sodium salt of p-toluenesulfonamide and ethyl N-butylcarbamate . Propose a mechanism for this step.Following is a retrosynthesis for the coronary vasodilator ganglefene. (a) Propose a synthesis for ganglefene from 4-hydroxybenzoic acid and 3-methyl-3-buten-2-one. (b) Is ganglefene chiral? If so, which of the possible stereoisomers are formed in this synthesis?Why do you suppose only symmetrical ethers are prepared by the sulfuric-acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal reactivity?
- Amines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2 reaction when treated with silver oxide as base. Pentylamine, for example, yields 1-pentene. Propose a structure for the intermediate, and explain why it readily undergoes elimination.(a) Arrange the following compounds in an increasing order of their indicated property :(i) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)(ii) CH3CH2CH (Br) COOH, CH3CH (Br) CH2COOH,(CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)(b) How would you bring about the following conversions :(i) Propanone to Propene (ii) Benzoic acid to Benzaldehyde(iii) Bromobenzene to 1-phenylethanolDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb. CH3COClc. (CH3CO)2Od. excess CH3Ie. (CH3)2C = Of. CH3COCl, AlCl3g. CH3CO2Hh. NaNO2, HCli. Part (b), then CH3COCl, AlCl3j. CH3CHO, NaBH3CN
- The formula for the danger sign pheromone for a species of ant is C7H7O. When treated with I2 NaOH, this pheromone produces iodoform and n-hexanoic acid. How is this pheromone structure?What products would you expect from the oxidation of thefollowing compounds with (i) CrO3 in aqueous acid? (ii) withPCC?(a) tert-butanol(b) cyclohexanol(c) cyclohexanoneQ1: What condensation products would you expect to obtain by treatment of the following substance with sodium ethoxide in ethanol?(a)Ethyl butanoate (b) Cycloheptenone(c) 3,7-Nonanedione (d) 3-phenylpropanal
- Undecanal is commercially available, but 6-methyl-1-bromoheptane is not. Propose two methods for the synthesis of 6-methyl-1-bromoheptane using precursors containing fewer than five carbon atoms (Cn n < 5). A protecting group will probably be required.Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CN2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (i) Draw the structural formula of compounds W, X and Y



