Dielectric Constant Of Water
Water constitutes about 70% of earth. Some important distinguishing properties of water are high molar concentration, small dissociation constant and high dielectric constant.
Electrostatic Potential and Capacitance
An electrostatic force is a force caused by stationary electric charges /fields. The electrostatic force is caused by the transfer of electrons in conducting materials. Coulomb’s law determines the amount of force between two stationary, charged particles. The electric force is the force which acts between two stationary charges. It is also called Coulomb force.
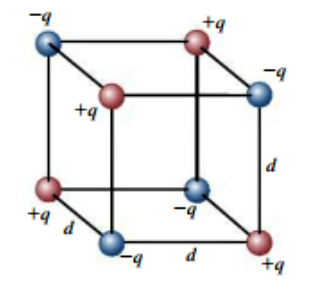
The figure shows eight point charges arranged at the corners of a cube with sides of length d. The values of the charges are +q and −q, as shown. This is a model of one cell of a cubic ionic crystal. In sodium chloride (NaCI), for instance, the positive ions are Na+ and the negative ions are CI−. (a) Calculate the potential energy U of this arrangement. (Take as zero the potential energy of the eight charges when they are infinitely far apart.) (b) In part (a), you should have found that U < 0. Explain the relationship between this result and the observation that such ionic crystals exist in nature.
(HINT: This is a configuration energy problem. However, it is lengthy to solve.)
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images









