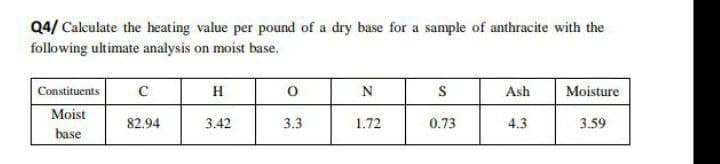
Q4/ Calculate the heating value per pound of a dry base for a sample of anthracite with the following ultimate analysis on moist base. Constituents Moist base с 82.94 H 3.42 0 3.3 N 1.72 S 0.73 Ash 4.3 Moisture 3.59
Q: 5. Complete this sequence of reactions. Provide a mechanism for each step along the way. 1) CH,MgBr…
A: In the first step of the reaction, the Grignard reagent reacts with the ketone to form tertiary…
Q: (a) Determine whether this hydrogen peroxide reaction is first-order or second-order by first…
A: A first-order reaction is one in which the rate of reaction depends only on one reactant…
Q: AlP Express answer as an ion.
A: Since, Ionic compound are those compound which on dissociation produced cation and anion. Thus,
Q: A certain liquid has a vapor pressure of 92.0 Torr at 23.0 °C and 217.0 Torr at 45.0 °C. Calculate…
A: Given , Initial temperature , T1=23.0°C=(23+273)K=296 KVapor pressure , P1 = 92.0 torrFinal…
Q: Low concentrations of warfarin near the detection limit gave the dimensionless instrument readings:…
A: Given: Instrument Readings 195.5 166.9 152.5 164.5 209.5 150.3 213.3 191.9 124.9…
Q: If for the reaction, ax + bY --> products, the rate law is determined to be r=K[X]¹[Y then the order…
A: Given: r = [X]1[Y]2 To determine order of the reaction
Q: PRE-LABORATORY ASSIGNMENT You must finish the pre-laboratory assignments before beginning the…
A:
Q: The molar heat of vaporization of water is 42 kJ/mol. How much energy is released by the…
A:
Q: Drag "+" for a positive reaction, and "-" for a negative reaction
A: For some reaction, the chemical tests represented by abbreviations are performed and based on the…
Q: An electromagnetic ray whose wavelength in 4x10-4 and when it passes through a transparent medium,…
A: The relation between the wavelength (λ) and the frequency (ν) is - (λ)×(ν)= c Here c is the speed of…
Q: When solid lead(II) phosphate is in equilibrium with its ions, the ratio of lead(II) ions to…
A: Given : lead(ii) phosphate is at equilibrium with its ions
Q: what is the solution?
A: Introduction A solution can be anything that can be made by mixing two or more substances. Soda…
Q: What is the mole fraction of solute in a 3.79 ?3.79 m aqueous solution?
A: The number of moles of solute present in one-kilogram of solvent is known as the molality of the…
Q: Which of the following single-replacement A. Cu(NO3)2 (aq) + Pb (s) B. Cu (s) + Pb(NO3)2 (aq)…
A: A more reactive metal can replace a less reactive metal ion from the solution of the salt of the…
Q: The freezing point of pure benzene (CoHo) is 5.49 °C. The freezing point of a solution made using…
A: A decrease in the freezing point is a colligative property. The decrease in the freezing point is…
Q: 67. The boiling point of Chloroform was raised by 0.325K when 5.141 × 10 kg of a non- volatile…
A: Colligative properties are properties of a solution that depend on the number of solute particles…
Q: is reduced in the following reaction: Cr₂O72- + 6S2032 + 14H+ → 2Cr³+ + 3S406² +7H₂O O Cr₂O7²- O…
A: Introduction The oxidation number of an atom is a measure of the degree of oxidation of an atom in a…
Q: 7. Using the bond dissociation energies provided below, draw an energy diagram for the two…
A: Suppose a reaction A2 + B2 –> C2 Then ΔHrxn given as ΔHrxn = Σ Bond energy of reactants -Σbond…
Q: The two compounds in A ar The two compounds in Bar the same molecule skeletal isomers functional…
A:
Q: 6) Draw an energy profile diagram with AH = - 10 kJ and E. (Activation Energy)=+ 25 kJ. Label:…
A:
Q: Write the electron configuration of the following atoms and identify what Family and period do they…
A: The electronic configuration and the class of the elements are given below
Q: + d. What is the rate of this reaction between 20.0 and 50.0 seconds? Use the balanced equation…
A: The rate of a chemical reaction is the change in concentration of a reactant or product over time.…
Q: Exercises 1. Draw the Lewis structures of the following compounds. a. CH CI b. NO3 c. 0₂- e. CO₂ g.…
A: This question is related to valence bond theory. In the given question, 10 molecules are given and…
Q: Figure 3-4 C=C DOG B о C A N H
A:
Q: A titration of 50.0 mL 0.010 M propanoic acid with 0.010 M KOH was carried out. Indicate the pH at…
A: Answer: This question is based on hydrolysis of salt. Salt of weak acid and strong base gets…
Q: The solubility of O2 in water is 5.85 x 10-4 M at 25 oC and 0.45 atm of O2 pressure . What will the…
A:
Q: Which has the larger AHhydr, Ca²+ or Sc³+? 1. Ca²+ 2. Sc3+ 3. they are the same 4. impossible to…
A: Water is a polar solvent and it dissolves ionic compounds. The negatively charged ion of an ionic…
Q: A0.1 M CH₂COOH solution is titrated with 0.1 M NaOH. At what point in the titration will the pH of…
A: During a titration of an acid with the base, the pH value changes as the concentration of the base…
Q: 11. Calculate the change in internal energy for the combustion of 1.0 mol of propene given that the…
A: The change in enthalpy is given to be -2058 kJ. The number of moles of propene combusted is 1.0 mol.…
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine…
A: In the given reaction, the gastric juices contain hydrochloric acid(HCl). First, write down the…
Q: You may want to reference Section 3.7 (Pages 80-87) while completing this problem. Using the values…
A: Condensation:- Condensation is a process of converting water vapor to water. This process is the…
Q: The decomposition reaction M H rightwards arrow M to the power of plus plus H to the power of minus…
A:
Q: A 1.384 g sample contains only vitamin C (CH₂O) and sucralose (C₁2H₁9C1₂O₂). When the sample is…
A:
Q: See Figure 3-1. Match the letter to the correct functional group.
A: Given : structure of molecule
Q: (c) Explain in chemical terms why treatment of ethanol with H₂SO4 and heat is not the best way to…
A: The Williamson ether synthesis is a chemical reaction used to synthesize ethers from alcohols and…
Q: Reaction 4 [copper (II) oxide → copper (II) sulfate] Sulfuric acid (H₂SO4) is corrosive. If you get…
A: The given reaction IV is - CopperII oxide→Copper (II) sulfate This reaction involves the addition of…
Q: If a reaction is first order with a rate constant of 0.0450 s¹, how much time is required for 65% of…
A: The given data is as follows: The rate constant for first order reaction = 0.0450 s-1 Let the…
Q: c. 2-hexene +HCI/H₂O → d. 3-methyl-1-pentyne + 2 moles HCI/H₂O →
A: These reactions goes through carbocation intermediate. More stable carbocation intermediate leads to…
Q: Which statement is the best description of carbon trifluoride, CHF33 polar bonds, non-polar molecule…
A: CHF3, also known as carbon trifluoride
Q: A) B) C) 3. Suggest suitable starting alkenes for the synthesis of each of the following compounds?…
A:
Q: 14. HCI НО ` ags/
A: In this reaction, The asymmetric alkene reacts with HCl such that the chlorine is added to the…
Q: If the K₂ of a weak acid is 1.6 x 10-8, the K, of its conjugate base partner must be which of the…
A:
Q: what will be in the HNMR of the following compound?
A: NMR spectroscopy (Nuclear Magnetic Resonance spectroscopy) is a analytical technique that uses the…
Q: 1.74. What is the ratio of the populations of two energy states whose energies differ by 1000 J at…
A:
Q: D A C B
A: Introduction Hydrobromination is a reaction in which bromine is added to an alkene or alkyne…
Q: onsider the table. Metal Tm (K) ΔHfus (kJ/mol) Tb (K) ΔHvap (kJ/mol) Li 454 2.99 1615 134.7 Na 371…
A: Given, Tm (K)=302 ΔHfus(kJ/mol)=2.10 Tb (K)=942 ΔHvap (kJ/mol)=66 To calculate, ΔSfus and ΔSvap
Q: The density of an (NaCl) ionic solid is 3.34 g/cm3 and the length of an edge is 482 pm. a. How…
A: Given: density of an (NaCl) ionic solid = 3.34 g/cm3 length of an edge (a) = 482 pm…
Q: < 2:19 A) S-¹ B) M S-¹ What are the units for the rate constant of a first-order reaction? C) M-¹ s¹…
A: Introduction The rate constant of a reaction is an important parameter that describes how quickly a…
Q: We mixed some ethylene glycol with 346 g water to reduce the equilibrium vapor pressure of water…
A: Answer: When a non-volatile solute is added in a solvent it causes a depression in freezing point…
Q: Identify the splitting patterns for each non-singlet hydrogen and draw the corresponding qualitative…
A:
Step by step
Solved in 2 steps with 1 images

- Method of separation/chemical reaction (with the brief process) of 3 KG's OF SODIUM BORATE AND 1.5 KG's HYDROCHLORIC ACIDThe fat in a 1.821 g sample of potato chips is extracted with supercritical CO2. After extraction, the residue weighs 1.139 g. What is the fat content (% w/w) of the potato chips?A sample of an iron ore was prepared for Fe3+ analysis as following: 3.4g of the sample was added anddissolved in acid environment then diluted to 250 mL using volumetric flask. After that, 10 mL of the resultingsolution was transferred by pipet to a 50-mL volumetric flask and continue to be diluted. The scientists foundout that this solution gives the concentration of Fe3+ as 2.3 mg/L. Find the weight percentage of Fe3+ in theoriginal sample.
- During what century was the balance intruduced for chemicak processes?The chief chemist of the Brite-Metal Electroplating Co. isrequired to certify that the rinse solutions that are discharged from the company’s tin-plating process into the municipal sewer system contain no more than 10 ppm (parts per million) by mass of Sn2+. The chemist devises the following analytical procedure to determine the concentration. At regular intervals, a 100-mL (100-g) sample is withdrawn from the waste stream and acidified to pH = 1.0. A starch solution and 10 mL of 0.10 M potassium iodide are added, and a 25.0-mA current is passed through the solution between platinum electrodes. Iodine appears as a product of electrolysis at the anode when the oxidation of Sn2+ to Sn4+ is practically complete and signals its presence with the deep blue color of a complex formed with starch. What is the maximum duration of electrolysis to the appearance of the blue color that ensures that the concentration of Sn2+does not exceed 10 ppm?Determine the percentage Fe in a sample of limonite from the following data:Sample : 0.5000g ; KMnO4 used = 50.00 ml ; 1.000ml of KMnO4 is equivalent 0.005317 g Fe,FeSO4 used = 6.00 ml; 1.000ml FeSO4 is equivalent 0.009200 g FeO ( ans 44.59 %)
- A sample known to consist of NaOH orNaHCO3, or Na2CO3 or possible compatible mixtures of these, together with inert matter. With methyl orange, a 1.10 g sample requires 31.40 mL of HCl (of which 1.00 mL ≈ 0.0140 g CaO). With phenolphthalein, the same weight of sample requires 13.30 mL of the acid. Calculate the percentage of inert matter in the sample.What is meant by non-stoichiometric defect? Ionic solids which have anionicvacancies due to metal excess defect develop colour. Explain with the helpof suitable example.A chemist received different mixtures for analysis with the statement that they contained NaOH, NaHCO3, Na2CO3, or compatible mixtures of these substances, together with inert material. From the data given, identify the respective materials, and calculate the percentage of each component. 1.000g samples and 0.2500N HCl were used in all cases. (A) For Sample W: With PP, 24.32ml was used. A duplicate sample required 48.64ml with MO. (B). For Sample X: The addition of PP caused no color change. With MO, 38.47ml of the acid was required. (C). For Sample Y: To cause a color change in the cold with PP, 15.29ml of the acid was necessary, and an additional 33.19ml was required for complete neutralization. (D) For Sample Z: The sample was titrated with acid until the pink of PP disappeared; this process required 39.96ml. On adding an excess of the acid, boiling, and titrating back with alkali, it was found that the alkali was exactly equivalent to the excess acid added.
- A sample of iron ore weighing 800 mg was treated with HNO3 , boiled to dryness and redissolved in dilute HCl. After filtration and removal of undissolved silica, the liquid was passed through a Walden reductor. The collected sample was titrated with 0.0210 M KMnO4 , requiring 12.0 mL to reach the end point. Calc %Fe (55.845) in the ore.An acid solution containing 2 per cent by mass of NaNO3, and an unknown concentration of HNO3 is used to regenerate a strong acid resin. After sufficient acid had been passed over the resin for equilibrium to attained, analysis showed that 10 per cent of resin sites were occupied by sodium ions. What was the concentration of HNO3 in the solution, if its density were 1,030 kg/cu.m.?Pyrolusite (MnO2) is dissolved in hydrochloric acid:MnO2 + HCl → MnCl2 + H2O + Cl2The chloride was passed into potassium iodide solution where it liberated iodine:Cl2 + KI → KCl + I2The iodine liberated was estimated by adding sodium thiosulfate, the reaction beingI2 + Na2S2O3 → NaI + Na2S4O6If 5.6 g of crystallized sodium thiosulfate, Na2S2O3 5H2O ,were used up, how many grams of manganese were present?MW (g/mole): I =126.9; Mn =54.94; Cl = 35.45; K = 39.09; Na = 23; S = 32



