QUESTION 21 Answer the following questions given the table below Vapor Pressure n-heptane kPa 101.400 117.8 136.1 156.7 179.5 204.8 232.7 263.5 297.3 334.2 Temperature T(F) T(K) T (C) 209.3 371.6 98.5 218.6 376.8 103.6 228.0 382.0 108.8 237.3 387.2 114.0 246.6 392.4 119.2 256.0 397.5 124.4 265.3 402.7 129.6 274.7 407.9 134.8 284.0 413.1 140.0 293.3 418.3 145.2 Styrene kPa 24.363 29.144 34.660 40.992 48.224 56.447 65.753 76.243 88.018 101.185 (a) At 124.4C what is the K value for Styrene? (b) At 124.4C what is the relative volatility for n-heptano? D 30 X 1.000 0.815 0.658 0.522 0.405 0.303 0.213 0.134 0.064 0.001 у 1.000 0.947 0.883 0.807 0.717 0.612 0.490 0.349 0.187 0.003
QUESTION 21 Answer the following questions given the table below Vapor Pressure n-heptane kPa 101.400 117.8 136.1 156.7 179.5 204.8 232.7 263.5 297.3 334.2 Temperature T(F) T(K) T (C) 209.3 371.6 98.5 218.6 376.8 103.6 228.0 382.0 108.8 237.3 387.2 114.0 246.6 392.4 119.2 256.0 397.5 124.4 265.3 402.7 129.6 274.7 407.9 134.8 284.0 413.1 140.0 293.3 418.3 145.2 Styrene kPa 24.363 29.144 34.660 40.992 48.224 56.447 65.753 76.243 88.018 101.185 (a) At 124.4C what is the K value for Styrene? (b) At 124.4C what is the relative volatility for n-heptano? D 30 X 1.000 0.815 0.658 0.522 0.405 0.303 0.213 0.134 0.064 0.001 у 1.000 0.947 0.883 0.807 0.717 0.612 0.490 0.349 0.187 0.003
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
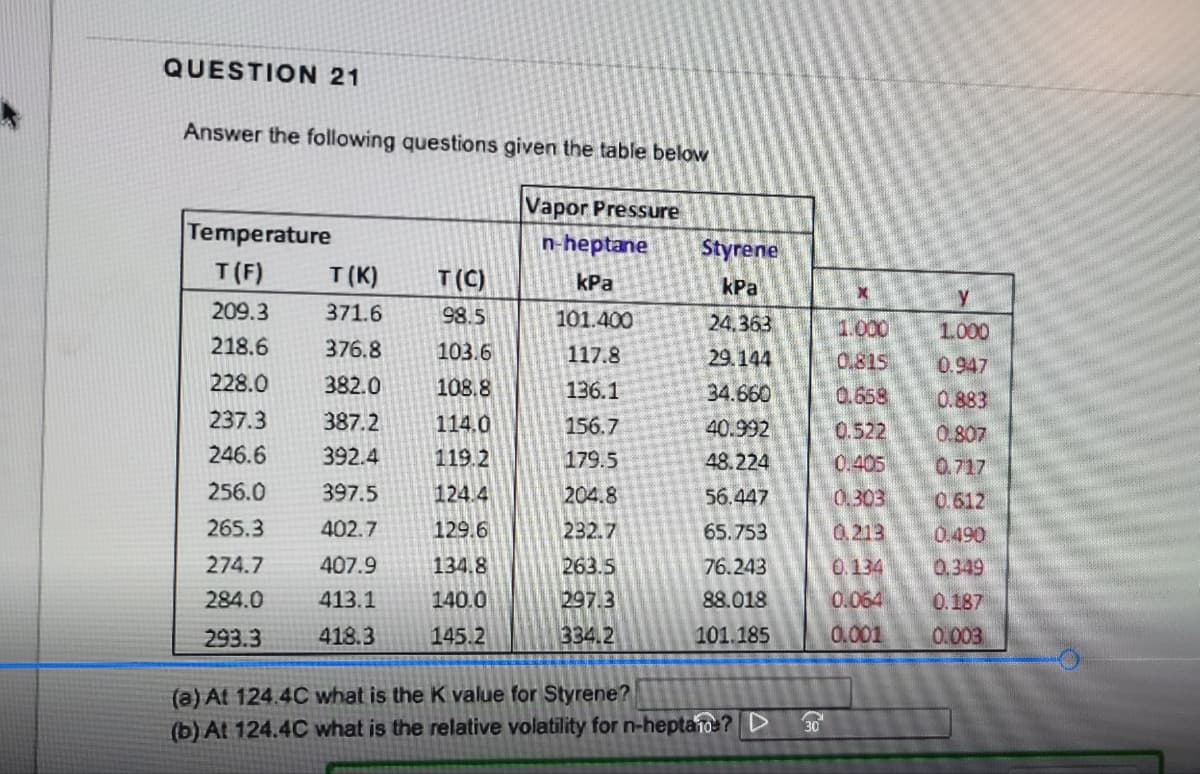
Transcribed Image Text:QUESTION 21
Answer the following questions given the table below
Vapor Pressure
n-heptane
kPa
Temperature
T(F)
209.3
218.6 376.8
228.0
382.0
237.3
387.2
246.6
392.4
397.5
402.7
407.9
413.1
418.3
256.0
265.3
274.7
284.0
293.3
T(K)
371.6
T (C)
98.5
103.6
108.8
114.0
119.2
124.4
129.6
134.8
140.0
145.2
101.400
117.8
136.1
156.7
179.5
204.8
232.7
263.5
297.3
334.2
Styrene
kPa
24.363
29.144
34.660
40.992
48.224
56.447
65.753
76.243
88.018
101.185
(a) At 124.4C what is the K value for Styrene?
(b) At 124.4C what is the relative volatility for n-heptano? D
30
X
1.000
0.815
0.658
0.522
0.405
0.303
0.213
0.134
0.064
0.001
Y
1.000
0.947
0.883
0.807
0.717
0.612
0.490
0.349
0.187
0.003

Transcribed Image Text:QUESTION 19
A distillation column with a partial reboiler and a total condenser is being used to separate a mixture of Comp A (1),
Comp B (2). and Comp C (3). The feed is 40.0% mole Comp A. 30.0% mole Comp B and 30.0% mole Comp C. The feed
is input as a saturated vapor. We desire 96.50% recovery of the Comp B in the bottoms and 99.50% recovery of the Comp
A in the distillate. The reflux is returned as a saturated liquid, and CMO can be assumed. Equilibrium can be represented
as constant relative volatilities. Choosing Comp B (2) as the reference component. a12 = 1.40 and a32 = 0.80.
Use a basis of 100 moles of feed/h.
Find the number of equilibrium stages required at total reflux.
will increase
LECTION 20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The