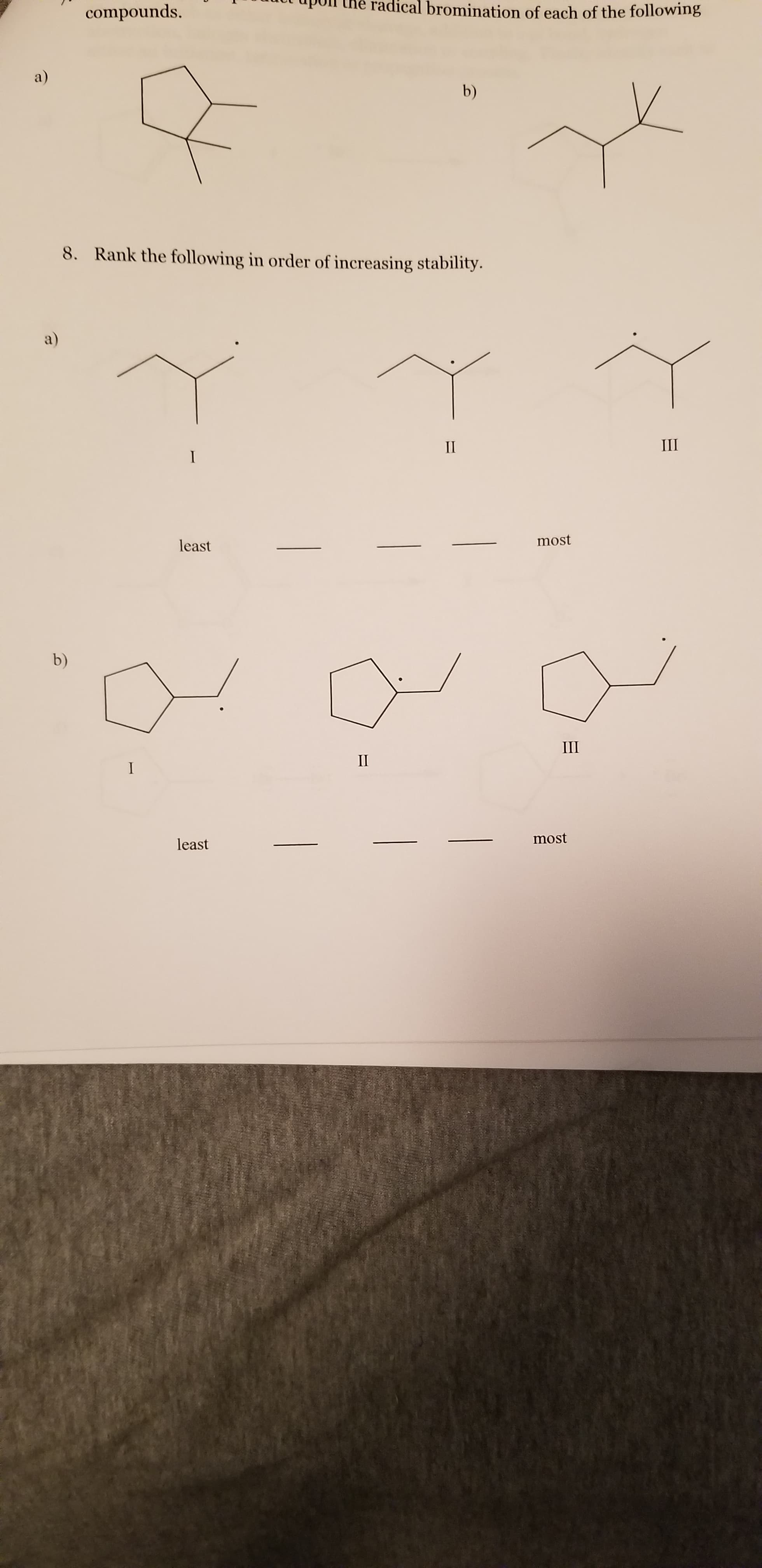
radical bromination of each of the following compounds. a) b) Rank the following in order of increasing stability. 8. a) Ш IП most least III П most least b)
Q: Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much…
A:
Q: at is the correct order of decreasing reactivity ost reactive first) of these compounds toward…
A: When electron donating groups are attached to benzene ring.so, they increase the reactivity towards…
Q: Which of the following is the most likely structure of the following carbocation after rearrangment…
A:
Q: Which of the following is a second chain propagation step in the free radical chlorination of…
A:
Q: Rank the attached alkenes in order of increasing stability.
A: The given alkenes are represented as follows:
Q: Which reaction is predicted to be faster (A or B)? Reactions A and B are of the same type. Classify…
A:
Q: which has faster rate, bromination (under hv) of cyclohexane or bromination (under Hv) of toluene?
A: Alkanes give a halogenation reaction with halogen in the presence of light or heat by free radical…
Q: Which of the following carbocations ( A or B) is more stable? Explain your choice.
A:
Q: Does dehydrogenation of alkyl halides create Zaitsef or Hoffman products? In this example, which is…
A: Alkyl halide undergoes dehydrogenation reaction in presence of a base and form alkene. This kind of…
Q: Explain Radical Halogenation at an Allylic Carbon ?
A: An allylic carbon is a carbon atom bonded to a carbon atom that is bonded to another carbon atom.
Q: Which of the following is a stronger nucleophile in a polar, aprotic solvent? Br
A: A polar aprotic solvent does not have a hydrogen atom that can participate in hydrogen bonding.
Q: Rank the attached carbocations in order of increasing stability?
A: The order of stability of carbocations is: Tertiary carbocation > secondary carbocation >…
Q: Draw the exo and endo product for the reaction of cyclopentadiene and maleic anhy- dride. Which one…
A: 2. Cyclopentadiene and maleic anhydride react readily in Diels-Alder reaction to form an addition…
Q: What happens to the rate of an SN2 reaction under the following conditions? Both [RX] and [:Nu−] are…
A: The rate of SN2 reaction depends upon the concentration of reactant (alkyl halide, RX ) and…
Q: For each pair of compounds, which has the polar bond that will undergo nucleophilic addition more…
A: (a)
Q: a. Which of the following would be more reactive in an electrophilic substitution and why? Use…
A:
Q: А D
A: We have to rank the following in order of reactivity in photobromination reaction from fastest to…
Q: c. Please rank the following radicals in order from least to most SELECTIVE: A В C D Please explain…
A:
Q: a. Rank the follow radicals from least to most stable. CH3 CH3 CH3 .F A B C D Now, explain…
A: Radical is a electron deficient species. Any electron donating group stabilize it and electron…
Q: Which of the following carbocations (A or B) is more stable? Explain your choice.
A: The carbocation A and B are stabilizing by the adjacent heteroatoms. Oxygen and nitrogen atoms…
Q: Which will undergo rearrangement upon SN1? Select one: OH a. HO C.
A: Given : Some Compounds are given To find : Compound that undergo rearrangement on SN1 Reaction…
Q: Rank the following radicals in order of decreasing stability. most stable least stable (сны,снінсн,…
A: The stability of carbon free radicals can be determined by considering the number of hypercojugative…
Q: Which C–H bond in attached compound is most readily broken during radical halogenation?
A: a. During cleavage of C-H bond about 3 types of radical can be formed from the given molecule. Of…
Q: Consider the following reaction being performed with a low concentration. Think about what type of…
A: The product of the given reaction is
Q: A. -Br D. 0-CH3 E. acats Rank in arder of increasing reactivity towards electrophilic aromatic…
A:
Q: b. С. + + + a.
A: The reactive intermediate species in which carbon atom forms only three bonds and contains a…
Q: Rank the following carbocations in order of decreasing stability, putting the most s II Multiple…
A: Stability of carbocation is as 3°>2°>1° We can see that I is a 3° Carbocation as the carbon…
Q: 2 Draw the retro Diels-Alder mechanism and product for each of the compounds shown here. Which retro…
A: Retro Diels-Alder reaction is reverse of the diels alder reaction. A diels alder reaction is a (4+2)…
Q: Carbocation rearrangement of the cation on the right gives: A) B) D) two of the above
A:
Q: What happens to the rate of an SN2 reaction under each of the following conditions? [RX] is…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Rank the following carbocations in terms of stability from lowest to highest. to A B C D
A:
Q: nydrogen and the leaving group have to be antiperiplanar in which reaction? E2
A: There are two types of elimination reactions. First is E1 and other is E2. (Dear student,…
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: Fill in the blank: The reaction of alkene with HBr in presence of peroxide (addition of HBr, ROOR)…
A: The reaction of alkene with HBr in presence of peroxide (addition of HBr, ROOR) is
Q: Draw the products of radical chlorination and bromination of each compound. For which compounds is a…
A: a.
Q: H;C CH3 HN- -c-CH, A в a) Which of the above molecules (A or B) have a higher rate of reaction…
A: We have two compounds, we have to predict the rate of electrophilic aromatic substitution of the…
Q: What happens to the rate of an SN2 reaction under the following conditions? [RX] is tripled, and…
A: The rate of an SN2 reaction under the given condition: [RX] is tripled, and [:Nu−] stays the same.
Q: Arrange the following alkyl halides in increasing reactivity in E2 reaction. Write 1 for the least…
A:
Q: In order of stability, rate the following compounds, with 1 being the most stable.
A:
Q: why does this carbocation go through rearrangemnt by a hydride shift?
A:
Q: The hydrobrominationof 2-methyl-1-butene could theoretically lead to 2 different alkyl bromides; in…
A: We are given an alkenes
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Q: Azulene, an isomer of naphthalene, having fused 5 and 7 membered rings, has the following orbital…
A: Azulene: It is non-benzenoid fused ring system in which 5-membered and 7-membered ring systems are…
Q: Q2 Which compound liberate the lowest heat upon hydrogenation with excess H,? Why? C В A products)
A: Alkenes can be defined as the compound containing double bonds between carbon atoms in that…
Q: Draw the products of radical chlorination and bromination of eachcompound. For which compounds is a…
A: Bromination is more selective than chlorination and secondary halogenation is preferred in case of…
Q: a) place asterisks(*) at the allylic position of compound A. b) Draw the resonance forms of the…
A:
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 3 images

- ochem help Rank the following radicals in the order of increasing stability (see attached image)Which bromocyclohexane starting material would react faster – the cis or trans, and explain why? Draw both chair forms of the starting material to prove your point.Rank the following carbocation in order of increasing stability?
- Does dehydrogenation of alkyl halides create Zaitsef or Hoffman products? In this example, which is created? I think it's Hoffman because there is a more stable/substituted option which is to put the double bond to the right of the positive charge instead of to the left. Or does it depend on the reactants? Further clarification is needed. Thank you in advance!For each one, what are the products? For each subpart, point that the mechanism (SN1, SN2,E1 or E2). Draw major product structureAn SN2 mechanism's top requirement from an aprotic solvent is to ensure that it can stabilize the leaving group.
- which has faster rate, bromination (under hv) of cyclohexane or bromination (under Hv) of toluene?a) place asterisks(*) at the allylic position of compound A. b) Draw the resonance forms of the allylic radical intermediate that accounts for the formation of B and C.For the following reactions: A. Draw the missing reactant, reagent or product including the correct stereochemistry where appropriate. B. Determine the Reaction Pathway (Sn1, Sn2, E1, E2) leading to the desired product C. How many steps are in the Reaction as can be plotted on a Reaction Coordinate?

