Q: Name the following dienes and rank them from most stable to least stable. (Hint: Alkyl groups…
A: Naming Rule : Select the longest parent chain keeping both the double bonds in it. And number it in…
Q: The reaction of Br2 with 1-methylcyclohexene, in the presence of ethylamine (EtNH2), is expected to…
A:
Q: 3. Assign E or Z stereochemistry to the following alkenes: (a) НОСH2 CH3 (b) HO2C H C=C C=C H3C H CI…
A: I am supposed to answer first question only
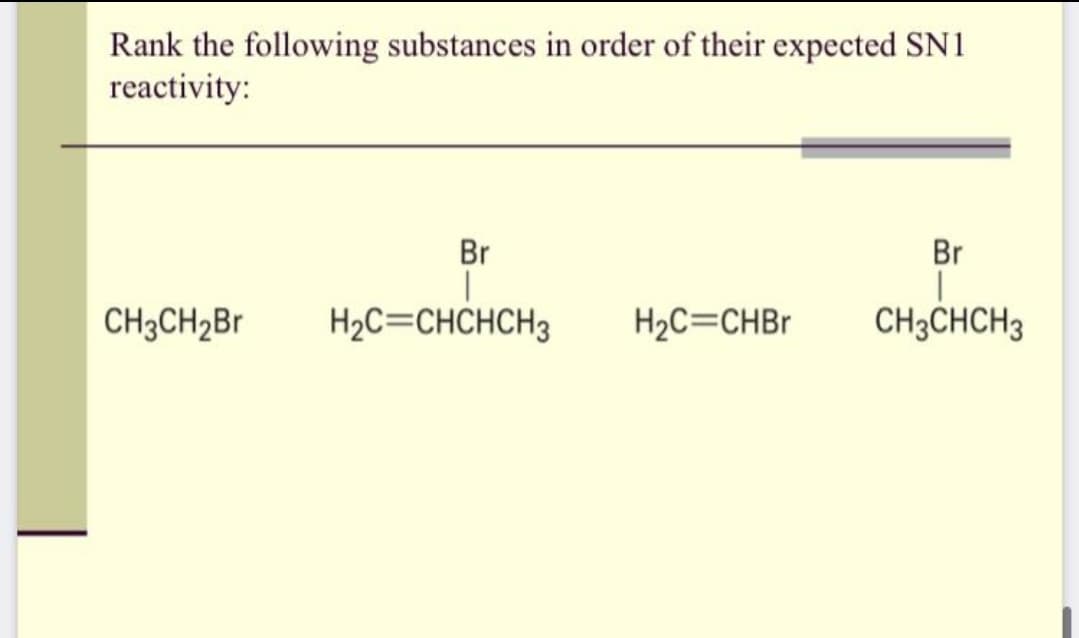
Q: Arrange the compounds below in order of increasing reactivity (least reactive = 1; most reactive =…
A: 1.B 2. A 3. C 4. D
Q: Which two compounds ionize with loss of bromide ion to form the same carbocation? CH3 CH3 CH3 CH2…
A: Halogen which is in conjugation with alkene give carbocation..
Q: Order the compounds below from least to most thermodynamically stable (relative to their constituent…
A: In this question, we want to arrange all these three compound from least stable to most stable. You…
Q: The thermal ring opening of trans-3,4-dimethylcyclobutene may yield two isomers, but only one isomer…
A: Electrocyclic ring opening in which polyana has 4n pi electrons - If the open chain polyene has 4n…
Q: What is the correct order of reactivity of CH3CHO, C2H5COCH3 and CH3COCH3? O CH3COCH3>…
A: The carbonyl compounds give nucleophilic addition reactions. The carbon atom of the carbonyl carbon…
Q: What are the missing reagents and compounds below Br A 03, DMS в Br. Na Na/NH3 D E Answers A B C D E…
A: The missing reagents and compounds in the given reactions above is mentioned below
Q: Identify the following reactions as either SN1, Sn2, E1, or E2: (a) Br CHCH3 CH=CH2 кон (ь) Br OCH3…
A: Applying concept of elimination and substitution reaction.
Q: Rank the following substituents in order of increasing priority. -CH=CH2 -OH -O-CH3 -CH II II O II,…
A: Answer - The correct option is (d) Explanation - The higher the atomic number of the immediate…
Q: Label the following compound as R or S. OH CCH2CH3 (CH3)2CH H.
A: Organic compounds are any chemical compounds that contain carbon-hydrogen bonds in general. Millions…
Q: Predict the major substitution product. Identify first whether it is Sn1/SN2 Br HO OH b. CI a.
A: A. SN1 Mechanism. B. SN2 Mechanism.
Q: CH3 HBr Peroxides CH3 H3C Br -Br CH3 CH3 C. А. ÇH3 ÇH3 Br CH3 `CH2B D. В.
A:
Q: Draw a structural formula for the product of each SN2 reaction. Where configuration of the starting…
A: Given reaction,
Q: Rank the dienes by DECREASING REACTIVITY in Diels-Alder reactions (most reactive on the left). CN…
A: The correct option is (b): V > II > III > I > IV
Q: What diene and dienophile are needed to prepare the following compound? Choose the single best…
A:
Q: Describe the 1H NMR spectrum you would expect for each of the following compounds, indicating the…
A: Since you have posted multiple questions, we are solving the first three sub-parts for you; for the…
Q: Draw the mechanisms Treatment of ‘1-butene’ with HCl/H2O gives ‘B’ Treatment of ‘1-butene’ with…
A: When alkenes react with water in presence of an acid catalyst, then most substituted alcohols are…
Q: What is the expected major product for the following reaction? Br2 CH3OH Br OCH, H3CO, OCH, H3CO Br…
A: The following is a representation of the structure of the provided starting compound:
Q: What is the decreasing order of stability for the following alkenes? II III IV O II>IV>I>II…
A:
Q: NBS CH3CH=CHCH2CH2CH3 CH;CH== CHCHCH½CH3 CH3CHCH=CHCH2CH3 + Br Br A B
A: The carbon next to double bonded carbon is called the allylic carbon. The hydrogen attached to the…
Q: Draw both diastereomers of 2-bromo-3- chlorobutane by editing the two drawings of 2-…
A:
Q: 1. Describe the synthesis of : a) NH 3 CH2 CH2 %3D b) NH3 CH3CH2 CH2 CH2NH2 CH2 CH3. CH3 CHIN-CH3…
A:
Q: Which of the following conformations of 1,2-dibromocyclohexane is the LEAST stable? Select one: Br H…
A: 1st:- both bromines are trans at 180 degree so there are no any interaction with lone pair-lone…
Q: Question attached
A: Diels-Alder reaction is an organic reaction taking place between a conjugated diene and a…
Q: Arrange the following compounds in the increasing order of reactivity in SN' reaction. 1.…
A: Nucleophilic substitution reaction of first order represent the substitution of weak nucleophile by…
Q: 3. Calculate the degree of unsaturation for the following compounds. Propose one structure for each…
A: Degree of unsaturation is given by, Degree of unsaturation =2C+2+N-H-X2 Unsaturation of compound…
Q: (a) or II (b) or II or (c) II or (d) II or (e) II
A:
Q: Arrange the following substituents in order of decreasing priority (highest first). -OCH2CH3 -F OH…
A: Arrange in decreasing order of priority ?
Q: Tell whether each of the following reactions is likely to be SN1, SN2, E1, ElcB, or E2: (c) CI…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Treatment of the following stereoisomer of 1-bromo-1,2-diphenylpropane with sodium ethoxide in…
A:
Q: Which pair of compounds are the most probable main products of the following reaction? CH3 HNO H2SO4…
A: A question based on electrophilic substitution reaction, which is to be accomplished.
Q: 6. Which of the following is most reactive with HBr? а. СН3ОН b. CH3CH2OH (CH3)2CHOH с. d. (CH3)3COH
A: Answer - The correct option is (d) (CH3)3COH Explanation - (CH3)3COH is most reactive with HBr…
Q: Which of the reagents below are expected to convert cyclopentene into cyclopentane? H2O Heat…
A: Given :- cyclopentene → cyclopentane possible reagents :- H2O heat H2, Ni Sulfuric acid To…
Q: What reactants are expected to afford the following Diels-Alder adduct? H3C H. D. DIOT C
A: Here we are required to find the diene and dienophile required for Diels Alder adduct
Q: Draw a structural formula for the product of each SN1 reaction. Where configuration of the starting…
A: In the given reaction, the nucleophile attacks the both front and back side of carbocation in SN1…
Q: Which choice correctly assigns the E/Z configuration of each alkene (I-III) shown below H3C H3CH2C…
A:
Q: Draw a structural formula for the product of each SN2 reaction. Where configuration of the starting…
A: Given:
Q: Assign E or Z configuration (a) H3C CH₂OH C=C CH3CH₂ Cl CO₂H Baca (c) CH3 CH₂OH to the following…
A: Assign E or Z configuration of the given structure---
Q: Write the following transformations. НCN/OH" NH3 Br2 NH3 СООН X1 NH3 H2/Pt СООН X1 X2
A: Solution: Here, we know HCN is a weak acid, so it will not good source of nucleophile (CN-).…
Q: Tell whether the following reaction is likely to be SN1, SN2, E1, E2 or E1cB, and predict the…
A: The given reaction is,
Q: Arrange in decreasing order according to their carbanion character. CH3CH2CH2M9CI CH3CH2CH2K…
A: Here we are required to arrange the following organometallic compound in Decreasing order of…
Q: Molecule Chair conformations and energies (kJ/mol) AE %N1 (kJ/mol) Methylcyclohexane H3C. No…
A:
Q: Which of the following carbocations is the least stable? -CH2* O (CH3)3C* O (CH3)2CH+ O CH2=CHCH,+
A: Carbocation: carbon ion that bears a positive charge on it.
Q: Draw a structural formula for the product of each SN2 reaction. Where configuration of the starting…
A: SN2 reaction or bimolecular nucleophilic substitution has following features: Formation of a…
Q: Predict the products of the following reactions. cyclopentadiene + methyl acrylate, CH2“CH¬COOCH3
A: The structure of cyclopentadiene is shown below. The structure of methyl acrylate is shown below.…
Q: Type of leaving groups favorable for SN2 & SN1 reactions & 3 examples SN2 SN1
A: Leaving group :- A leaving group is a molecular fragment that separates with a pair of…
Q: Arrange the following carbocations in the order of increasing stability (least stable to most…
A: Carbocations are sp2 hybridised Carbon containing species which are unstable cations. These are…
Q: Reagents a. CoHsCHO b. NaOH, ethanol c. Pyrrolidine, cat. H* j. Br2, H* d. H2C=CHCN e. H3O* f. LDA…
A: The enolate is formed from the cyclopentanone by reaction with LDA. The enolate reacts with vinyl…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Determine the Ksp of the following reactions. a. NaC2H3O2 Na+ + C2H3O2- b. HBr H+ + Br c. Zn(OH)2 Zn+2 + 2HO- d. PbCl2 Pb+2 + 2Cl- e. SnSO4 Sn+2 + SO4-Arrange the following group in order of increasing priority. Q) -CH3 -H -Br -CH2CH3One of the following molecules (a)–(d) is d-erythrose 4-phosphate, an intermediate in the Calvin photosynthetic cycle by which plants incorporate CO2 into carbohydrates. If d-erythrose 4-phosphate has R stereochemistry at both chirality centers, which of the structures is it? Which of the remaining three structures is the enantiomer of d-erythrose 4-phosphate, and which are diastereomers?
- identify and interpret the peaks and their characteristics (C=O, 4H, doublet) product name: 2,5-dibenzylidenecyclopentanone (C19H16O)Determine the Ksp of the following reactions PbCl2 Pb+2 + 2Cl-Assign a priority order (from least to highest priority) to each of the following sets of groups: I. -OCH II. -NH(CH3)2 III. CH2NH2 IV. -OH A. IV, I, III, II B. III, II, IV, I C. II, III, IV, I D. III, IV, I, II
- Calculate the Keq for the following reaction, given that Eo Cd2+/Cd = -0.403 V and Eo Zn2+/Zn = -0.763. Overall rxn: Cd2+ + Zn <-> Zn2+ + CdDescribe the E/Z stereochemistry for the molecules shown below: A: Z; B: Z A: Z; B: E A: E; B: E A: E; B: ZDo Sn2 reactions always happen with inversion of stereochemistry, and does the stereocenter always change from R to S or S to R?

