Read each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. If you checked yes, give the symbol of an element that matches. Does any element match the description? description An element in Period 1 and Group 1A. Yes No Ar A metalloid in Group 8A. Yes No A noble gas with a higher atomic number than calcium. Yes No A main-group element in Period 6. Yes No I Don't Know Submit O 2020 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy 10 MacBook Pr esc 23 del 1O H. B command option command * 00
Read each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. If you checked yes, give the symbol of an element that matches. Does any element match the description? description An element in Period 1 and Group 1A. Yes No Ar A metalloid in Group 8A. Yes No A noble gas with a higher atomic number than calcium. Yes No A main-group element in Period 6. Yes No I Don't Know Submit O 2020 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy 10 MacBook Pr esc 23 del 1O H. B command option command * 00
Chapter3: Atoms And Elements
Section: Chapter Questions
Problem 65E: Here are three fictitious elements and a molecular view of the atoms that compose them. The molar...
Related questions
Question
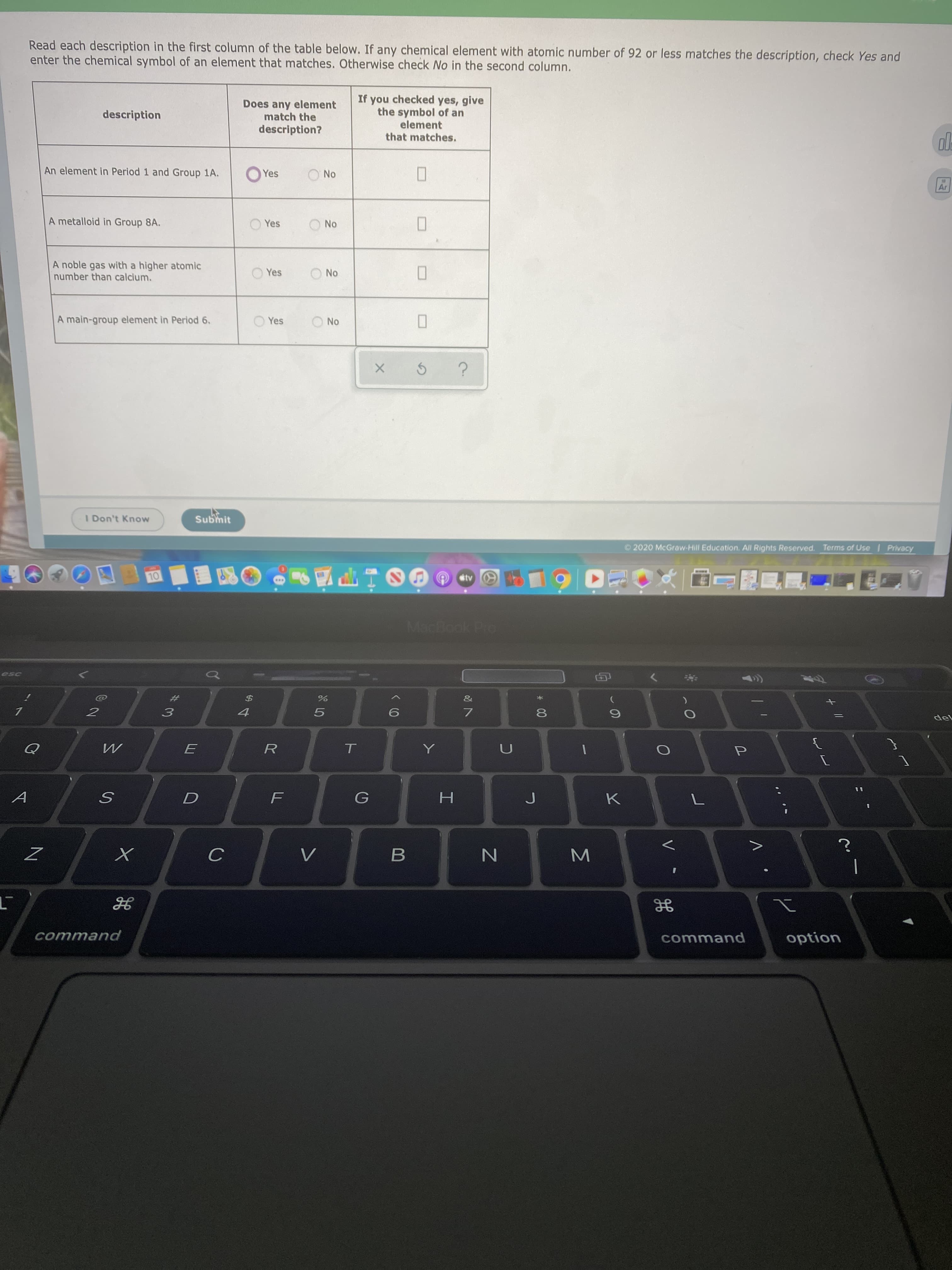
Transcribed Image Text:Read each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and
enter the chemical symbol of an element that matches. Otherwise check No in the second column.
If you checked yes, give
the symbol of an
element
that matches.
Does any element
match the
description?
description
An element in Period 1 and Group 1A.
Yes
No
Ar
A metalloid in Group 8A.
Yes
No
A noble gas with a higher atomic
number than calcium.
Yes
No
A main-group element in Period 6.
Yes
No
I Don't Know
Submit
O 2020 McGraw-Hill Education. All Rights Reserved.
Terms of Use
Privacy
10
MacBook Pr
esc
23
del
1O
H.
B
command
option
command
* 00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Recommended textbooks for you


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning