Search... nvellum.ecollege.com/course.html?courseld3D15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001 Ae. AOL Video - Serving the best vi. ine Sh... TripAdvisor
Search... nvellum.ecollege.com/course.html?courseld3D15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001 Ae. AOL Video - Serving the best vi. ine Sh... TripAdvisor
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Transcribed Image Text:Search...
nvellum.ecollege.com/course.html?courseld3D15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001
Ae. AOL Video - Serving the best vi.
ine Sh... TripAdvisor
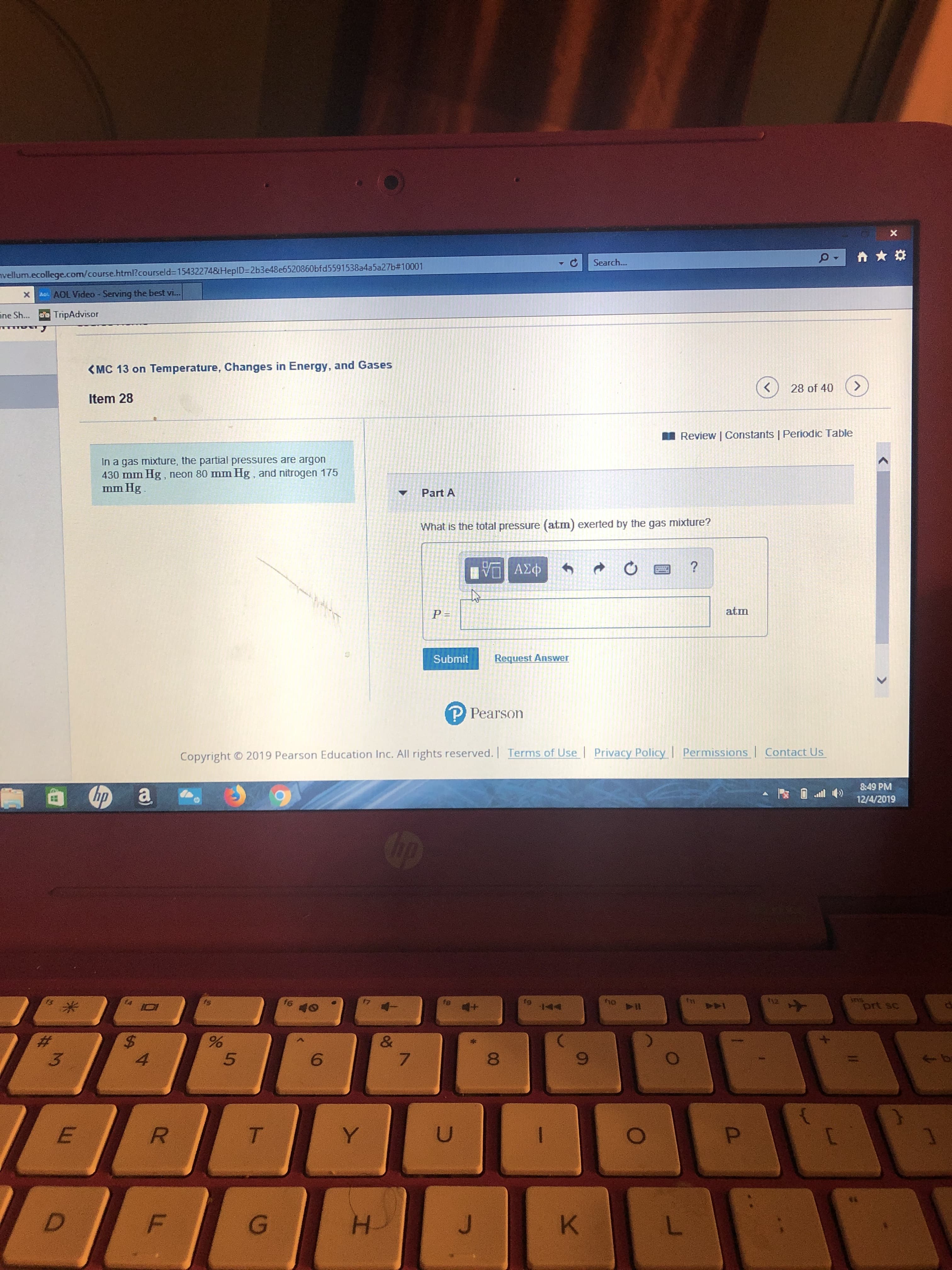
<MC 13 on Temperature, Changes in Energy, and Gases
28 of 40
Item 28
Review | Constants | Periodic Table
In a gas mixture, the partial pressures are argon
430 mm Hg, neon 80 mm Hg, and nitrogen 175
mm Hg.
Part A
What is the total pressure (atm) exerted by the gas mixture?
Vα ΑΣφ
atm
Request Answer
Submit
P Pearson
Contact Us
Permissions
Privacy Policy
Terms of Use
Copyright © 2019 Pearson Education Inc. All rights reserved.
8:49 PM
12/4/2019
ip
STAS
prt sc
12
fg
f6
f4
144
%23
3
%$4
&
8.
7.
T.
P.
Y.
D.
H.
F.
%24
LI
%23
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

