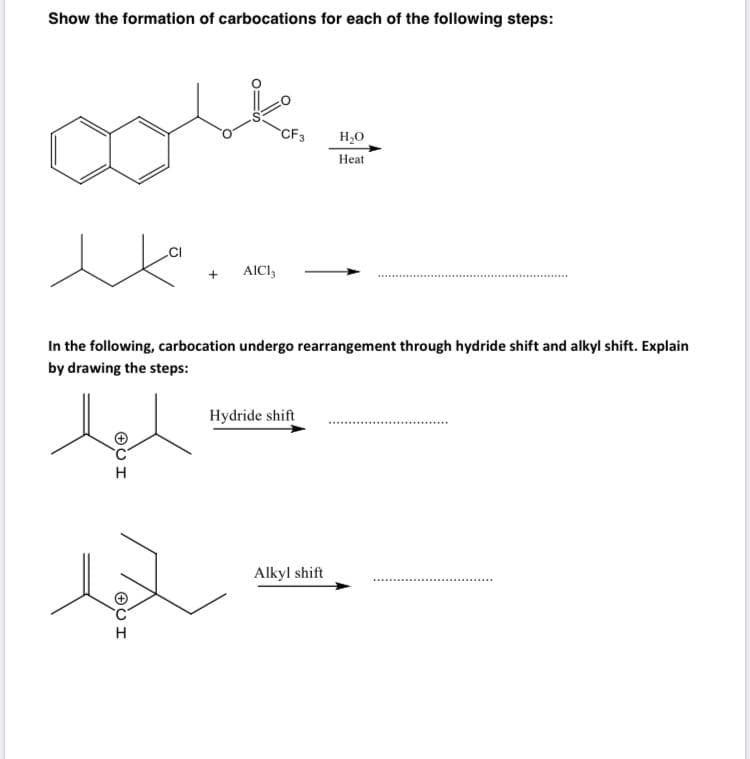
Show the formation of carbocations for each of the following steps: CF3 Н,о Нeat .CI AICI, In the following, carbocation undergo rearrangement through hydride shift and alkyl shift. Explain by drawing the steps: Hydride shift Alkyl shift
Q: 1. H20 H;C- H3C CH, aqueous H,SO, он 2. -CH3 `CH3 h = SN1 Nucleophilic substitution i= Sy2…
A:
Q: HO Provide a mechanism for the reaction below. Illustrate the role played by N,N,N- triethylamine in…
A: Trifluoroacyl chloride is highly electrophilic in nature. The aniline adds to the electrophilic…
Q: If 1,3,5-trimethylcycloheptane will undergo monobromination, how many possible alkyl halides will be…
A:
Q: Consider the following pericyclic reaction. CH3 H3C H. `H H3CO/ H3C H3CO `CH3 K L (a) Name the type…
A: Here we have to say the name of pericyclic process and have to predict the reaction is thermally or…
Q: HO. K2Cr,07 H* H20
A:
Q: Which of the following structures exhibit sp³ hybridized nitrogen atom? H-N CH3 H3C
A: When any Alkane undergoes halogenation Reaction, then haloalkanes are formed. Haloalkanes have…
Q: Using retrosynthetic analysis, determine which compound(s) could lead to the alkane shown below in a…
A: The following product can be obtained by catalytic hydrogenation i.e. H2/ pd.
Q: 2. Given that the organometallic catalyst (C6H5)Co(CO)2 acts to catalyze the following "triyne…
A: The first step is the coordination of the organometallic complex with the alkyne bonds of…
Q: Which combination of reagents A-D is suitable for the synthesis of the following compound: Br + Li B…
A:
Q: CH;CHCH-CH, CH3
A:
Q: 2. What Alkene would be used to synthesize 3-bromohexane? + HBr CH;CH,CHCH,CH,CH; br 3. Write down…
A: Reaction proceed through the most stable carbocation. It will give the major product . Carbocation…
Q: What are the intermediates and final product of the following synthesis? 1. LIAIH4, Et,0, 0 °C 2A…
A:
Q: Predict the product for the following reaction fuming H2SO, SO3H H. SO2 SO3 OH III IV O II 0 0 o o o
A:
Q: 0. Which two structures ionize with loss of bromide to form the same carbocation? Br Br Br 20 Br I…
A: In a chemical reaction; the substance which involves in conversion is said to be reactant whereas…
Q: Following are two diastereomers of 3-bromo-3,4-dimethylhexane. On treatment with sodium ethoxide in…
A: Answer (1): Both diastereomer of 3-bromo-3,4-dimethylhexane treat with sodium ethoxide in…
Q: CLG 12 – Delocalized Electrons II (10/18/21) 1. 2,3-dimethyl-1,3-pentadiene can conceivably give…
A:
Q: n-Bu Na NH2 1. -CN CN 2 Br NaBr NH3 toluene, reflux n-Bu OH Aqueous acetone 2. H20 HBr optically…
A: The first reaction is a double alkylation of a nitrile compound. The second reaction is a…
Q: Write the expected substitution product(s) for each reaction and predict the mechanism by which each…
A: The given alkyl halide is an example of secondary alkyl halide since the bromide is attached to a…
Q: 4. Show the detailed mechanism for the following reactions. Label steps (Sn2, protonation..), and…
A: We have given the organic reaction and we have to find the mechanism of the reaction.
Q: Carbocations undergo 1,2-methyl shift or 1,2-hydride shift to produce a more stable intermediate.…
A: The mechanism can be shown as:
Q: 1. Give the chemical mechanism and the major for the following reactions using curved arrow…
A:
Q: 1. Mechanisms and synthesis will be important parts of Exams 2, 3, and the final (and Chem228).…
A: Since your question has multiple sub-parts, we will answer the first three questions for you. If you…
Q: CH3 CH3 p-TSCI A pyridine 1. LIAIH4 →B CH;CO, Na+ HC ОН DMSO 2. H,0 lit.. CH13 НО CH13 (R)-2-Octanol…
A:
Q: The mechanism of bromination of an alkene, which involves a cyclic bromonium ion, explains why…
A: Note: Addition of bromine to cis-2-butene produces cyclic bromonium ion. This bromonium ion is…
Q: Arrange the following alkenes in order of increasing stability: (I) (II) (III) (IV) O a. II, IV, II,…
A: In this question we have to tell the stability of alkene.
Q: CH3 CH3ČCHCH3 CH3
A: The stability of the carbocation can be understood by the hyperconjugation structures. As the number…
Q: Predict the product of the following reaction. HNO, H2SO4 NO2 SO,H NO3 NH2 II IV
A:
Q: What is the expected major product for the reaction below? EGMU 2020737654 t-BUOK Br ? t-BUOH 4.…
A: This reaction is an elimination reaction . H and Br should be anti to each other. As it is a anti…
Q: 10 mol % Pd(OAc)2 BUNCI, K₂CO DMF, 100 °C OMe Pd(OAc)2 PhyP (20%) BC Reaction 2 BnO₂C7 OBn MeOOC…
A:
Q: Why is it necessary to incorporate the benzyl group on the chloroamine used to convert B to C?
A:
Q: 11. In an organic chemistry lab, chemists were attempting to convert compound I into compound III…
A: The Carbon atom which contains positive charge and bonded with three other bonds is known as…
Q: Suggest a mechanism and predict the major product for th 1) LIAIH, 2) H0
A: When ketones reduced by strong Oxidising agent like lithium aluminum hydride (LiAlH4) then finally…
Q: Rank the following organobromides in order of increasing rates of ionization to its carbocations and…
A:
Q: 1. determine the type of pericyclic reaction (sigmatropic, electrocyclic, cycloaddition) 2. draw the…
A:
Q: Predict the structure of the products (A-E) of the following reactions. Br BuzSnH OMe AIBN Toluene,…
A:
Q: 2) Provide a structure for the expected product of the following reaction. 1) Mg Br -? H3C 2) 3)…
A: Halides reacts with Mg and produces Grignard reagents i.e RMgX. In the reaction between epoxy and…
Q: Match the following SN1 and E1 reactions to the correct energy diagram. ASsume all reactions will…
A:
Q: H,O a) H,SO, Br2 b) H, с) Pt
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: 2. Provide reagents and reaction conditions that will give the product shown as the onl major…
A: Let us discuss each reaction.
Q: 3. Give the major product(s) and provide the mechanism of formation for the major product: ELOH…
A:
Q: 4. using retrosynthetic analysis, determine which compounds could lead to the alkene shows below in…
A: Potassium tert-butoxide can carry out elimination to give least substituted alkene. So the product…
Q: 3) Consider the synthesis below. Identify P1 and deduce What are the reagents/reactants A and B A…
A: Given:
Q: Explain the mechanism-Carboxylation—Reaction of RMgX with CO2
A: Carboxylation reaction of Grignard reagent (G.R) can occur by passing CO2 gas through the G.R and…
Q: 8. show electron flow). Suggest a detailed step-by-step reaction mechanism for the reactions shown…
A: First reaction is ring contraction reaction. And second one is expansion reaction.
Q: 3. Each of the following carbocations can rearrange to a more stable ion. Propose structures for the…
A: Carbocation is a species in which carbon carries positive charge.
Q: Identify the main expected product when the epoxide below reacts with PhMgCI, followed by acid…
A: 1. Epoxide is opened with Grignard at less substituted side (Sn2 reaction). 2. 6 pi electrons are…
Q: 6. Complete the following reaction scheme (g) CH H3C. `CH 1) 9-BBN 2) H,О, NaOH H3C (h) H2O, H2SO4.…
A:
Q: Rearrangment (р 1283) CH, H,C. ZnBr, H,C CH, Br HBr CH 93% yield он (Manning P. Cooke, Jr.) I need…
A: The mechanism of reaction is provided below in attach image.
Q: Write a mechanism for the following reaction. You MUST use curved arrows to represent electron…
A:
Q: The nitroso group, −N=O, is one of the few nonhalogens that is an ortho- and para- directing…
A: One the basis of resonating structure we will decide Nitroso benzene is ortho para directing or…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Help with the following ochem reaction scheme... Consider 3,4-dimethylpiperidine being subjected to the below reaction scheme: step 1) CH3I (excess) step 2) NaOH, heat step 3) CH3I (excess) step 4) NaOH, heat Provide the bond line structures for the major organic product obtained in each step and then discuss the regiochemistry for step 2.Explain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?
- In the presence of acid such as H2SO4, 3,4-dimethyl-1-pentene undergoes isomerization to 2,3-dimethyl-2-pentene (shown below). Write a complete mechanism, showing all steps and using curved arrows to explain this observation.The hydroboration–oxidation of internal alkynes produces ketones.(a) When hydroboration–oxidation is applied to but-2-yne, a single pure product isobtained. Determine the structure of this product, and show the intermediates in itsformation.(b) When hydroboration–oxidation is applied to pent-2-yne, two products are obtained.Show why a mixture of products should be expected with any unsymmetricalinternal alkyne.Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Show how free-radical halogenation might be used to synthesize the followingcompounds. In each case, explain why we expect to get a single major product.(a) 1-chloro-2,2-dimethylpropane (neopentyl chloride)The hydroboration–oxidation of internal alkynes produces ketones.(a) When hydroboration–oxidation is applied to but-2-yne, a single pure product isobtained. Determine the structure of this product, and show the intermediates in itsformation.Carbocations often rearrange, as shown below. Draw in the hydrogens on the two carbons involved in the rearrangement, and show formal arrow-pushing to illustrate the transformation.
- The following reaction is similar to the dehydration of 2-methylcyclohexanol. However, a significant amount of a terrasubstitued alkene is expected. Predict the structure for this tetrasubstituted alkene. Also, using curved arrows provide a reasonable mechanism for its formation.Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Conformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.

