Shown here are the resonance contributors for the cationic intermediate in the electrophilic addition of bromine to the ortho position of 4-methylphenol. For each of the first three resonance structures, add one double-barbed curved arrow to show the delocalization of the electron pair to form the structure to the right. Br: LBr: The final structure is the major resonance contributor and explains why the - OH group is... a deactivator and an ortho/para director. (Continued from above.) an activator and a meta director. a deactivator and a meta director. an activator and an ortho/para director.
Shown here are the resonance contributors for the cationic intermediate in the electrophilic addition of bromine to the ortho position of 4-methylphenol. For each of the first three resonance structures, add one double-barbed curved arrow to show the delocalization of the electron pair to form the structure to the right. Br: LBr: The final structure is the major resonance contributor and explains why the - OH group is... a deactivator and an ortho/para director. (Continued from above.) an activator and a meta director. a deactivator and a meta director. an activator and an ortho/para director.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter19: Eas: Electrophilic Aromatic Substitution
Section: Chapter Questions
Problem 32CTQ
Related questions
Question
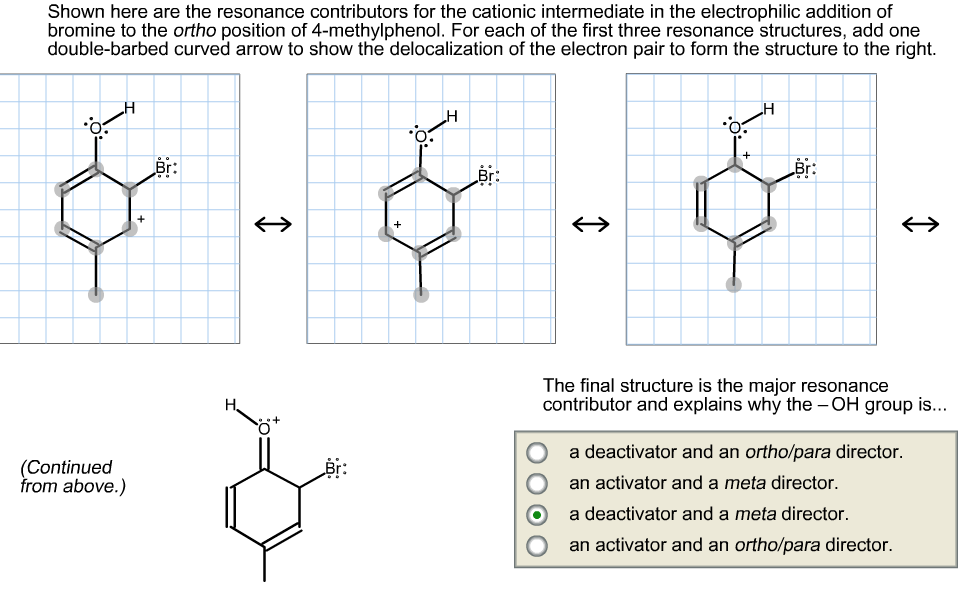
Transcribed Image Text:Shown here are the resonance contributors for the cationic intermediate in the electrophilic addition of
bromine to the ortho position of 4-methylphenol. For each of the first three resonance structures, add one
double-barbed curved arrow to show the delocalization of the electron pair to form the structure to the right.
Br:
LBr:
The final structure is the major resonance
contributor and explains why the - OH group is...
a deactivator and an ortho/para director.
(Continued
from above.)
an activator and a meta director.
a deactivator and a meta director.
an activator and an ortho/para director.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning