Sofia Gaseous ethane (CH, CH,) will react with gaseous oxygen (O,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,O). Suppose 15.3 g of ethane is mixed with 82. g of oxygen. Calculate the minimum mass of ethane that could be left over by the chemical reaction. Round your answer to 2 significant digits. x10 Check Explanation O 2019 McGraw-Hill Education. All Rights Reserved. Terms of Use I Prive 2: 12/ 99+) Type here to search hp delete 144
Sofia Gaseous ethane (CH, CH,) will react with gaseous oxygen (O,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,O). Suppose 15.3 g of ethane is mixed with 82. g of oxygen. Calculate the minimum mass of ethane that could be left over by the chemical reaction. Round your answer to 2 significant digits. x10 Check Explanation O 2019 McGraw-Hill Education. All Rights Reserved. Terms of Use I Prive 2: 12/ 99+) Type here to search hp delete 144
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 21A
Related questions
Question
100%
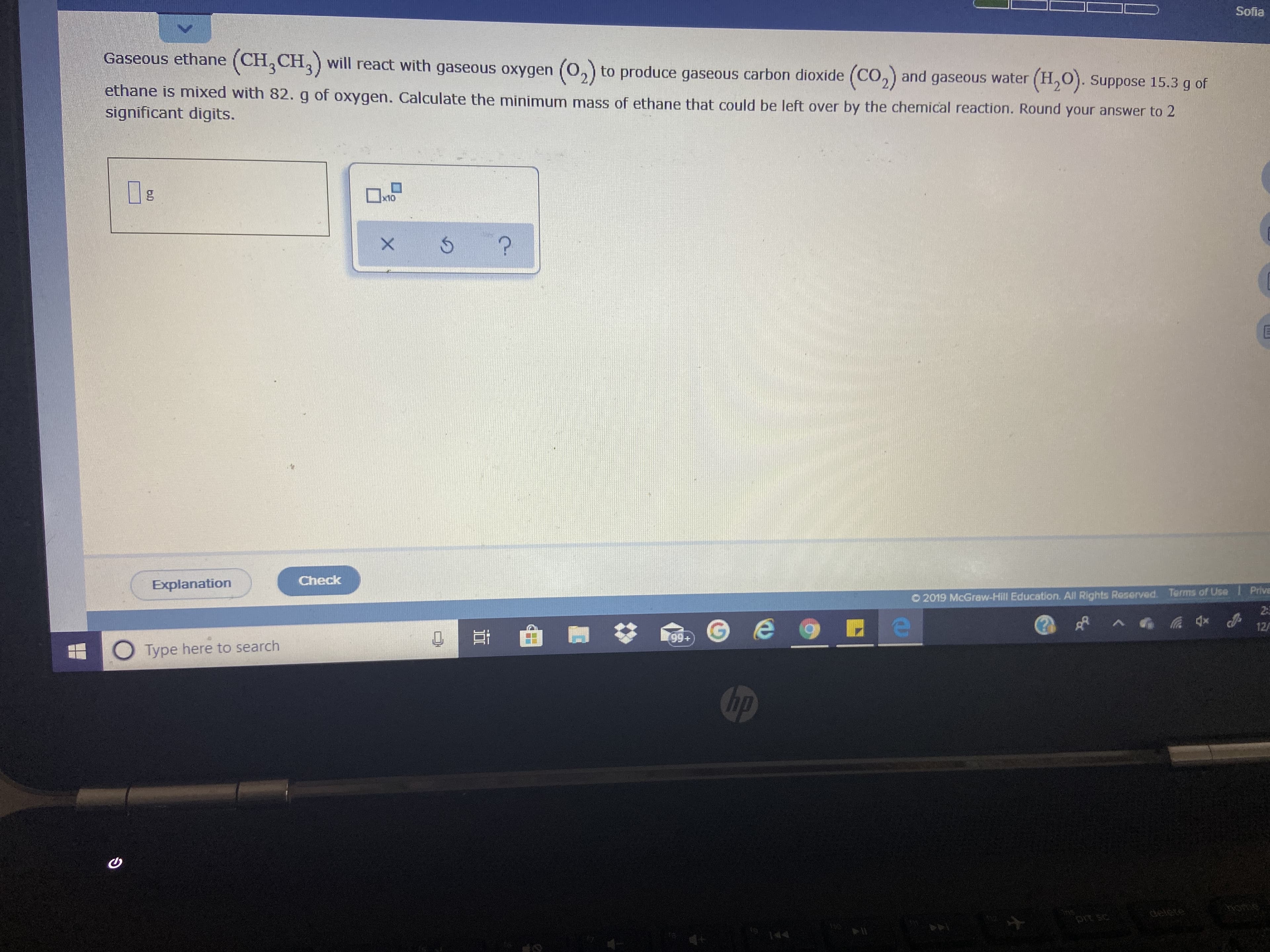
Transcribed Image Text:Sofia
Gaseous ethane (CH, CH,) will react with gaseous oxygen (O,) to produce gaseous
carbon dioxide (CO,) and gaseous water (H,O). Suppose 15.3 g of
ethane is mixed with 82. g of oxygen. Calculate the minimum mass of ethane that could be left over by the chemical reaction. Round your answer to 2
significant digits.
x10
Check
Explanation
O 2019 McGraw-Hill Education. All Rights Reserved. Terms of Use I Prive
2:
12/
99+)
Type here to search
hp
delete
144
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning