Table 1. Data for standard addition method Volume of Tablet Volume of standard Absorbance Final Volume solution (mL) (mL) 5.00 0.00 0.59 100.0 mL 5.00 1.00 0.74 100.0 mL 5.00 2.00 0.85 100.0 ml 5.00 3.00 1.02 100.0 ml 5.00 4.00 1.17 100.0 mL The standard solution- The solution was prepared by dissolving 0.400 g of acetyl salicylic acid in 250.0 mL of water. The same method as described by the calibration method. Tablet solution : The solution was prepared following the method described in the calibration method by reacting tablet with NaOH and finally diluting to 250.0 mL. Use this data to prepare the standard addition curve. Please remember to find the concentration of each standard solution in the volumetric flask before you plot the data.
Table 1. Data for standard addition method Volume of Tablet Volume of standard Absorbance Final Volume solution (mL) (mL) 5.00 0.00 0.59 100.0 mL 5.00 1.00 0.74 100.0 mL 5.00 2.00 0.85 100.0 ml 5.00 3.00 1.02 100.0 ml 5.00 4.00 1.17 100.0 mL The standard solution- The solution was prepared by dissolving 0.400 g of acetyl salicylic acid in 250.0 mL of water. The same method as described by the calibration method. Tablet solution : The solution was prepared following the method described in the calibration method by reacting tablet with NaOH and finally diluting to 250.0 mL. Use this data to prepare the standard addition curve. Please remember to find the concentration of each standard solution in the volumetric flask before you plot the data.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.13QAP
Related questions
Question
Please help me find the concentration of each standard solution in the v-flask, and prepare a curve.
The solution was diluted with ddH2O.
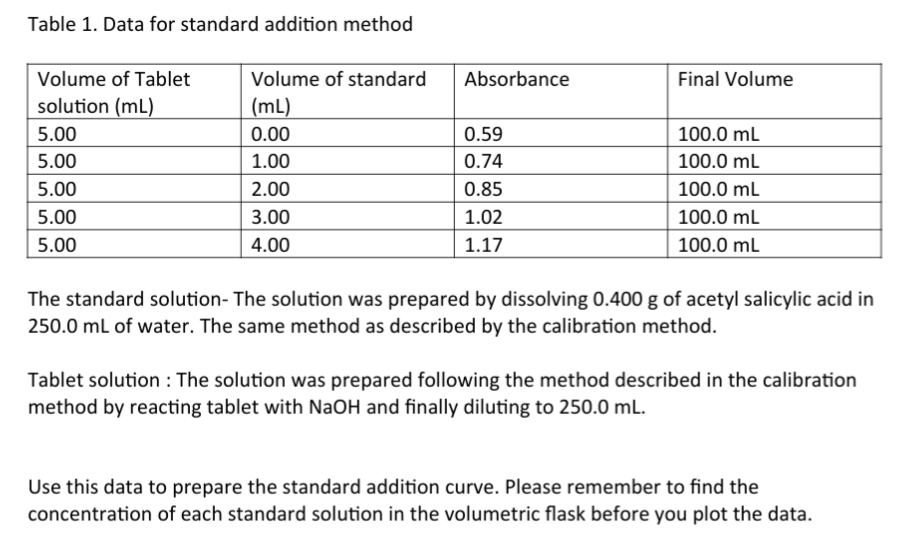
Transcribed Image Text:Table 1. Data for standard addition method
Volume of Tablet
Volume of standard
Absorbance
Final Volume
solution (mL)
(mL)
5.00
0.00
0.59
100.0 mL
5.00
1.00
0.74
100.0 ml
5.00
2.00
0.85
100.0 mL
5.00
3.00
1.02
100.0 ml
5.00
4.00
1.17
100.0 mL
The standard solution- The solution was prepared by dissolving 0.400 g of acetyl salicylic acid in
250.0 ml of water. The same method as described by the calibration method.
Tablet solution : The solution was prepared following the method described in the calibration
method by reacting tablet with NaOH and finally diluting to 250.0 mL.
Use this data to prepare the standard addition curve. Please remember to find the
concentration of each standard solution in the volumetric flask before you plot the data.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
