Table 11.1 (data) m Test 15-cm Test Tube 10-cm Test Tube Tube Amount of KIO3 Trial Number Amount of Water Amount of NaHSO3 oln Reaction Time 20 drops 20 drops 20 drops 35 drops 30 drops 20 drops 5 drops 1:26 10 drops l0:42 20 drops 1:21 2 3 Table 11.2 (report) oncentration ria Number Concentration Reaction-Rate Change Reaction Rate Change betweern Trials 1 and 2 betweern Trials 1 and 3 2 3
Table 11.1 (data) m Test 15-cm Test Tube 10-cm Test Tube Tube Amount of KIO3 Trial Number Amount of Water Amount of NaHSO3 oln Reaction Time 20 drops 20 drops 20 drops 35 drops 30 drops 20 drops 5 drops 1:26 10 drops l0:42 20 drops 1:21 2 3 Table 11.2 (report) oncentration ria Number Concentration Reaction-Rate Change Reaction Rate Change betweern Trials 1 and 2 betweern Trials 1 and 3 2 3
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter20: Kinetics
Section: Chapter Questions
Problem 20.87E: Pyrolysis involves heating compounds to break them into smaller molecules, and typically involves...
Related questions
Question
100%
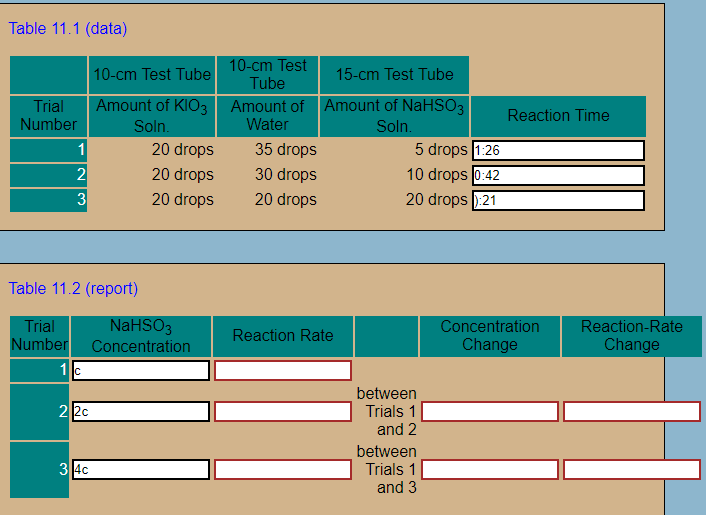
Transcribed Image Text:Table 11.1 (data)
m Test
15-cm Test Tube
10-cm Test Tube
Tube
Amount of KIO3
Trial
Number
Amount of
Water
Amount of NaHSO3
oln
Reaction Time
20 drops
20 drops
20 drops
35 drops
30 drops
20 drops
5 drops 1:26
10 drops l0:42
20 drops 1:21
2
3
Table 11.2 (report)
oncentration
ria
Number Concentration
Reaction-Rate
Change
Reaction Rate
Change
betweern
Trials 1
and 2
betweern
Trials 1
and 3
2
3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,