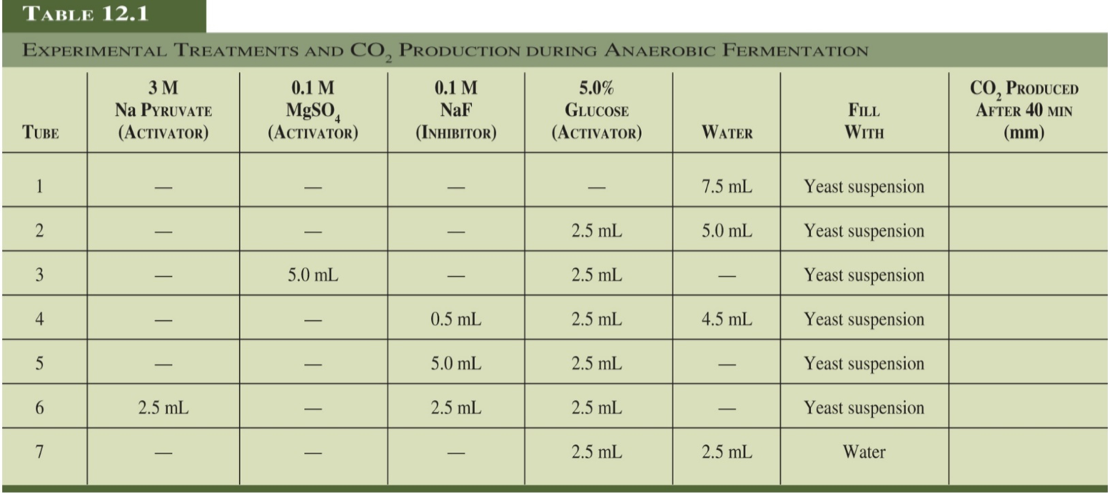
TABLE 12.1 EXPERIMENTAL TREATMENTS AND CO, PRODUCTION DURING ANAEROBIC FERMENTATION 0.1 M MgSO, (ACTIVATOR) CO, PRODUCED AFTER 40 MIN ЗМ Na PYRUVATE 0.1 M 5.0% NaF GLUCOSE FILL (ACTIVATOR) TUBE (ACTIVATOR) (INHIBITOR) WATER WITH (mm) Yeast suspension 1 7.5 mL Yeast suspension 2.5 mL 5.0 mL Yeast suspension 3 5.0 mL 2.5 mL Yeast suspension 2.5 mL 4.5 mL 4 0.5 mL Yeast suspension 5.0 mL 2.5 mL 2.5 mL 2.5 mL Yeast suspension 2.5 mL 6. 2.5 mL 2.5 mL Water
TABLE 12.1 EXPERIMENTAL TREATMENTS AND CO, PRODUCTION DURING ANAEROBIC FERMENTATION 0.1 M MgSO, (ACTIVATOR) CO, PRODUCED AFTER 40 MIN ЗМ Na PYRUVATE 0.1 M 5.0% NaF GLUCOSE FILL (ACTIVATOR) TUBE (ACTIVATOR) (INHIBITOR) WATER WITH (mm) Yeast suspension 1 7.5 mL Yeast suspension 2.5 mL 5.0 mL Yeast suspension 3 5.0 mL 2.5 mL Yeast suspension 2.5 mL 4.5 mL 4 0.5 mL Yeast suspension 5.0 mL 2.5 mL 2.5 mL 2.5 mL Yeast suspension 2.5 mL 6. 2.5 mL 2.5 mL Water
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 22P: Based on your residing of this chapter, what would you expect to be the most immediate effect oil...
Related questions
Question
a. Explain the purpose of each chemical in each test tube set up provided in the table. Use the principles of experimental variables, experimental controls, and controlled variables
b. Predict the amount of carbon dioxide produced from each test tube sample using a scale of 0 to 5, with 5 being the greatest amount of carbon dioxide. Provide an explanation for each sample.
Transcribed Image Text:TABLE 12.1
EXPERIMENTAL TREATMENTS AND CO, PRODUCTION DURING ANAEROBIC FERMENTATION
0.1 M
MgSO,
(ACTIVATOR)
CO, PRODUCED
AFTER 40 MIN
ЗМ
Na PYRUVATE
0.1 M
5.0%
NaF
GLUCOSE
FILL
(ACTIVATOR)
TUBE
(ACTIVATOR)
(INHIBITOR)
WATER
WITH
(mm)
Yeast suspension
1
7.5 mL
Yeast suspension
2.5 mL
5.0 mL
Yeast suspension
3
5.0 mL
2.5 mL
Yeast suspension
2.5 mL
4.5 mL
4
0.5 mL
Yeast suspension
5.0 mL
2.5 mL
2.5 mL
2.5 mL
Yeast suspension
2.5 mL
6.
2.5 mL
2.5 mL
Water
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning
