ter 06 em 17 Part A For the reaction, calculate how many grams of the product form when 1.6 g of Cl2 completely reacts. Assume that there is more than enough of the other reactant. 2 Na(s) +Cl2 (g)-2 NaCl(s) Express your answer using two significant figures. VO A ΑΣφ ? 3.2 mNaCl= Previous Answers Request Answer Submit Incorrect; Try Again; 3 attempts remaining Part B P Pearson 2019 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy PolicyI Pern Copyright FIC F7 F9 F6 F5 F8 F2 F3 F4 $ 4 # & 0 3 5 7 X
ter 06 em 17 Part A For the reaction, calculate how many grams of the product form when 1.6 g of Cl2 completely reacts. Assume that there is more than enough of the other reactant. 2 Na(s) +Cl2 (g)-2 NaCl(s) Express your answer using two significant figures. VO A ΑΣφ ? 3.2 mNaCl= Previous Answers Request Answer Submit Incorrect; Try Again; 3 attempts remaining Part B P Pearson 2019 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy PolicyI Pern Copyright FIC F7 F9 F6 F5 F8 F2 F3 F4 $ 4 # & 0 3 5 7 X
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 6ALQ: You have a chemical in a sealed glass container filled with air. The setup is sitting on a balance...
Related questions
Question
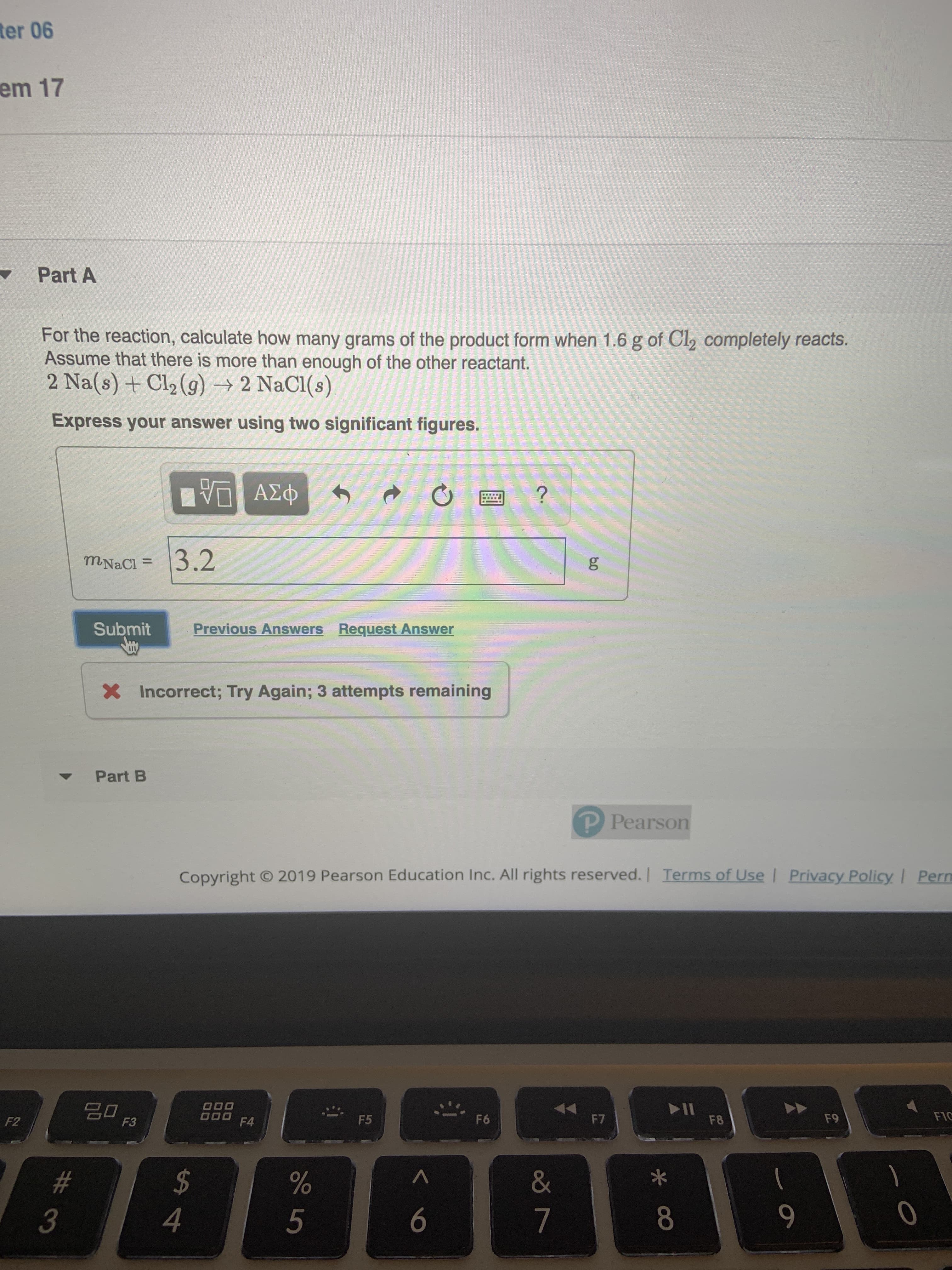
Transcribed Image Text:ter 06
em 17
Part A
For the reaction, calculate how many grams of the product form when 1.6 g of Cl2 completely reacts.
Assume that there is more than enough of the other reactant.
2 Na(s) +Cl2 (g)-2 NaCl(s)
Express your answer using two significant figures.
VO A
ΑΣφ
?
3.2
mNaCl=
Previous Answers Request Answer
Submit
Incorrect; Try Again; 3 attempts remaining
Part B
P Pearson
2019 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy PolicyI Pern
Copyright
FIC
F7
F9
F6
F5
F8
F2
F3
F4
$
4
#
&
0
3
5
7
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning