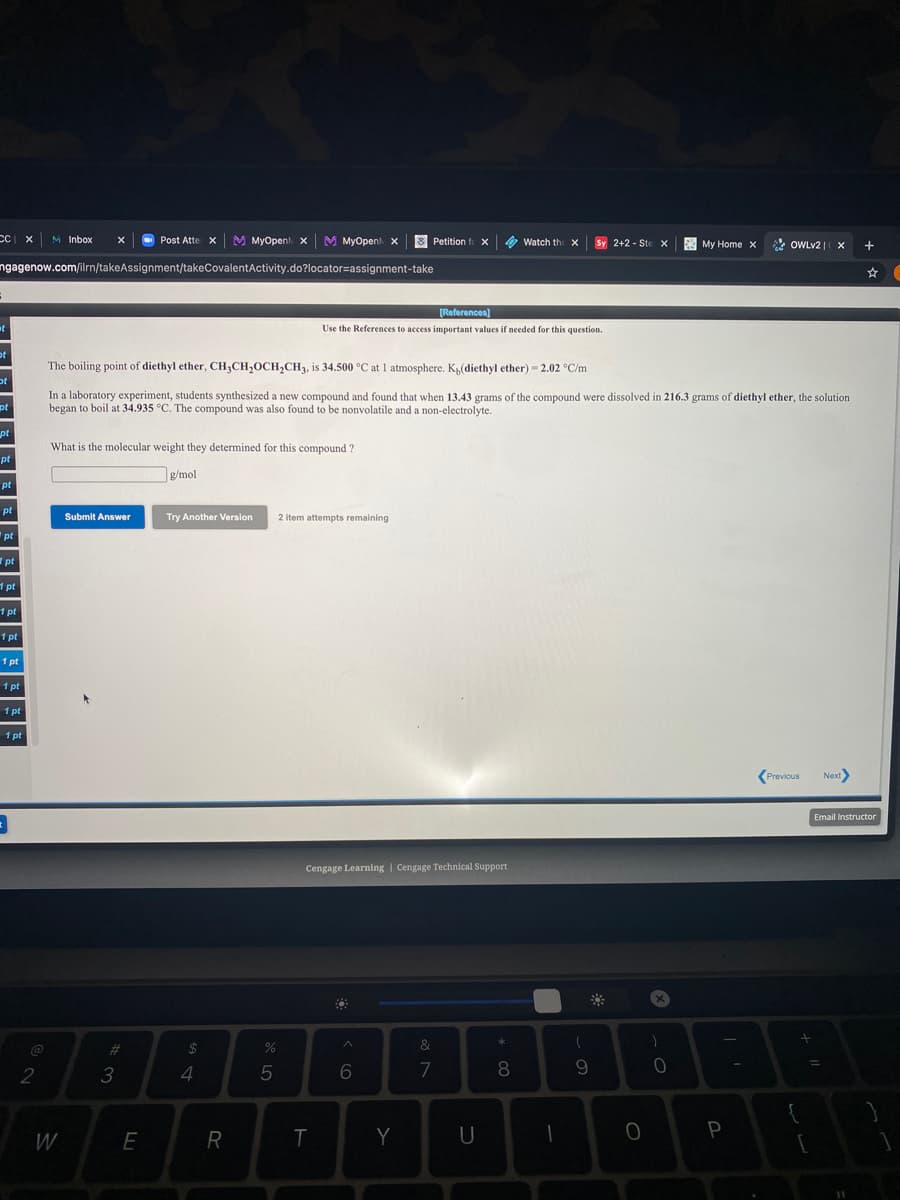
The boiling point of diethyl ether, CH3CH,OCH,CH3, is 34.500 °C at 1 atmosphere. K4(diethyl ether) = 2.02 °C/m In a laboratory experiment, students synthesized a new compound and found that when 13.43 grams of the compound were dissolved in 216.3 grams of diethyl ether, the solution began to boil at 34.935 °C. The compound was also found to be nonvolatile and a non-electrolyte. What is the molecular weight they determined for this compound ? g/mol
The boiling point of diethyl ether, CH3CH,OCH,CH3, is 34.500 °C at 1 atmosphere. K4(diethyl ether) = 2.02 °C/m In a laboratory experiment, students synthesized a new compound and found that when 13.43 grams of the compound were dissolved in 216.3 grams of diethyl ether, the solution began to boil at 34.935 °C. The compound was also found to be nonvolatile and a non-electrolyte. What is the molecular weight they determined for this compound ? g/mol
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 109AE: Patients undergoing an upper gastrointestinal tract laboratory test are typically given an X-ray...
Related questions
Question
Transcribed Image Text:M Inbox
Post Atte x
M MyOpen xM MyOpen x
S Petition f x
O Watch the x
Sy 2+2 - Ste x
E My Home x
* OWLV2 | x
ngagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator=assignment-take
[References)
Use the References to access important values if needed for this question.
The boiling point of diethyl ether, CH3CH,OCH,CH3, is 34.500 °C at 1 atmosphere. K(diethyl ether) - 2.02 °C/m
In a laboratory experiment, students synthesized a new compound and found that when 13.43 grams of the compound were dissolved in 216.3 grams of diethyl ether, the solution
began to boil at 34.935 °C. The compound was also found to be nonvolatile and a non-electrolyte.
What is the molecular weight they determined for this compound ?
pt
g/mol
pt
pt
Submit Answer
Try Another Version
2 item attempts remaining
pt
pt
1 pt
1 pt
1 pt
1 pt
1 pt
1 pt
1 pt
Previous
Next
Email Instructor
Cengage Learning | Cengage Technical Support
2$
3
4
7
8.
9.
W
R
Y
U
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning