The conversion of cyclopropane to propene in the gas phase is a first-order reaction with a rate constant of 6.7 x 10-4 s-¹ at 500°C. (a) (b) (c) CH, CH₂ CH₂ cyclopropane CH3-CH=CH₂2 propene If the initial concentration of cyclopropane was 0.25 M, what is the concentration after 8.8 min? How long (in minutes) will it take for the concentration of cyclopropane to decrease from 0.25 M to 0.15 M? How long (in minutes) will it take to convert 74 percent of the starting material? 1
The conversion of cyclopropane to propene in the gas phase is a first-order reaction with a rate constant of 6.7 x 10-4 s-¹ at 500°C. (a) (b) (c) CH, CH₂ CH₂ cyclopropane CH3-CH=CH₂2 propene If the initial concentration of cyclopropane was 0.25 M, what is the concentration after 8.8 min? How long (in minutes) will it take for the concentration of cyclopropane to decrease from 0.25 M to 0.15 M? How long (in minutes) will it take to convert 74 percent of the starting material? 1
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter33: Automated Methods Of Analysis
Section: Chapter Questions
Problem 33.6QAP
Related questions
Question
Transcribed Image Text:ALEKS X M Sign In
3.4 - Work - Microsoft Edge
//prod.reader-ui.prod.mheducation.com/epub/sn_2f0a6/data-uuid-9487cf7940e9479f8d43c65f2dbcdadb
807 of 1513 >
|_ Essent x organix Integra x Auth
(a)
(b)
(c)
X
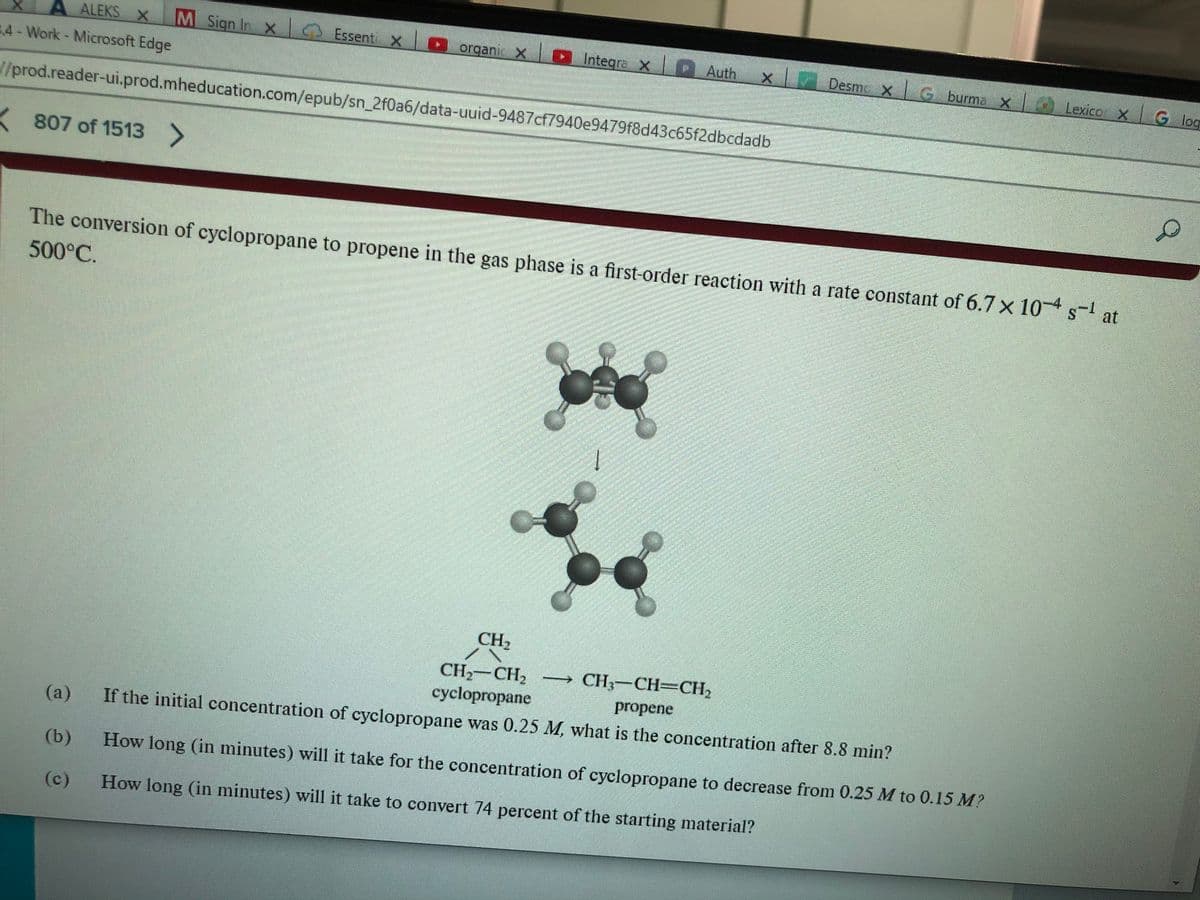
The conversion of cyclopropane to propene in the gas phase is a first-order reaction with a rate constant of 6.7 x 10-4 s-¹ at
500°C.
S
C
Desm_ x | Ġ burm; x | Lexico x G
G log
CH₂
CH₂ CH₂ - CH₂-CH=CH₂
cyclopropane
propene
If the initial concentration of cyclopropane was 0.25 M, what is the concentration after 8.8 min?
How long (in minutes) will it take for the concentration of cyclopropane to decrease from 0.25 M to 0.15 M?
How long (in minutes) will it take to convert 74 percent of the starting material?
D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
How did u get 2.303? I don't understand how you came up with that number and can you plz type the formula used to get t.
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning