The enthalpy of solution for the dissolving of a KBr sample was determined following Part C of the Experimental Procedure in this experiment. Complete the following table for Trial 1 for determining the enthalpy of solution of KBr. 1. Mass of salt (g) 2. Moles of salt (mol) 3. Mass of water (g) 5.00 25.0 25.0 18.1 4. Initial temperature of water (°C) 5. Final temperature of water (°C) Calculations for Enthalpy (heat) of Solution for the Dissolution of a Salt 1. Temperature change of solution, AT (°C) 2. Heat change by water (J) Show calculation. 3. Heat change of salt (J), specific heat capacity of KBr = 0.439 J/g.°C) 4. Total enthalpy change (J) Equation 25.11. Show calculation. 5. AH, (J/mol salt) Equation 25.12. Show calculation. %3D
The enthalpy of solution for the dissolving of a KBr sample was determined following Part C of the Experimental Procedure in this experiment. Complete the following table for Trial 1 for determining the enthalpy of solution of KBr. 1. Mass of salt (g) 2. Moles of salt (mol) 3. Mass of water (g) 5.00 25.0 25.0 18.1 4. Initial temperature of water (°C) 5. Final temperature of water (°C) Calculations for Enthalpy (heat) of Solution for the Dissolution of a Salt 1. Temperature change of solution, AT (°C) 2. Heat change by water (J) Show calculation. 3. Heat change of salt (J), specific heat capacity of KBr = 0.439 J/g.°C) 4. Total enthalpy change (J) Equation 25.11. Show calculation. 5. AH, (J/mol salt) Equation 25.12. Show calculation. %3D
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter4: Energy And Chemical Reactions
Section: Chapter Questions
Problem 65QRT
Related questions
Question
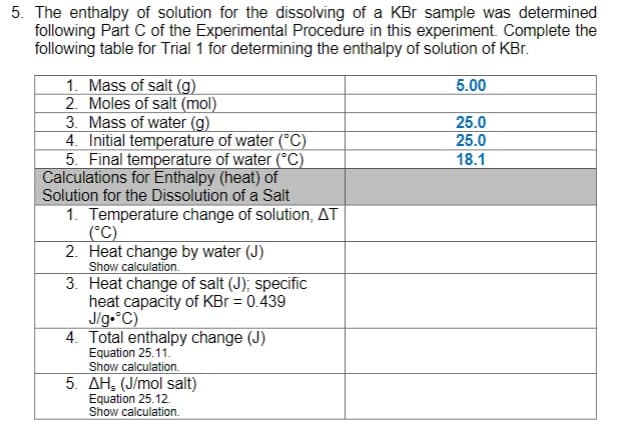
Transcribed Image Text:5. The enthalpy of solution for the dissolving of a KBr sample was determined
following Part C of the Experimental Procedure in this experiment. Complete the
following table for Trial 1 for determining the enthalpy of solution of KBr.
1. Mass of salt (g)
2. Moles of salt (mol)
5.00
3. Mass of water (g)
25.0
25.0
4. Initial temperature of water (°C)
5. Final temperature of water (°C)
Calculations for Enthalpy (heat) of
18.1
Solution for the Dissolution of a Salt
1. Temperature change of solution, AT
(°C)
2. Heat change by water (J)
Show calculation.
3. Heat change of salt (J); specific
heat capacity of KBr = 0.439
J/g.°C)
4. Total enthalpy change (J)
Equation 25.11.
Show calculation.
5. AH, (J/mol salt)
Equation 25.12.
Show calculation.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,