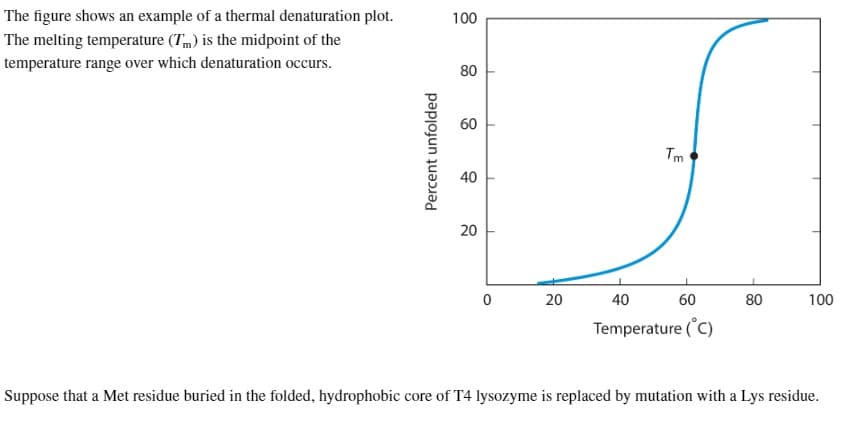
The figure shows an example of a thermal denaturation plot. The melting temperature (Tm) is the midpoint of the temperature range over which denaturation occurs. Percent unfolded 100 80 60 40 20 0 20 Tm 40 60 Temperature (°C) 80 100
Q: Enzyme Urease Chymotrypsin Alcohol Dehydrogenase Lysozyme Enzyme Official Name (write N/A if…
A: Urease- an enzyme produced by many bacteria, fungi etc. It hydrolyzes urea into ammonia and carbon…
Q: The enzyme involved in the formation of fructose-6-phosphate is a/an A. isomerase B. aldolase C.…
A: Fructose-6-phosphate is an intermediate of glycolysis. Isomerases are the enzymes that catalyze…
Q: Determine the interactions shown in the figure at point P * M N R.. CH₂ O Hydrophobic interaction O…
A: the interactions shown in the figure at point P a)Electrostatic interaction b)Covalent bonding…
Q: Mention all the enzymes and reagents used in the everse Transcriptase Polymerase Chain Reaction…
A: RT PCR is Reverse transcription polymerase chain reaction. This a process used to generally…
Q: Mixtures of amino acids can be analyzed by first separating the mixture into its components through…
A: In ion exchange chromatography the column support matrix is constituted of beads to which charged…
Q: Draw and name an aldose and ketose having five (5) carbon atoms. Identify in your drawing the point…
A: Carbohydrates are a class of macromolecules that plays an important role as energy source for the…
Q: What is a codon, how many RNA nucleotides are in a codon, How many of these code for amino acids,…
A: Transcription is the process by which the genetic information stored in the DNA is copied onto an…
Q: Detergents disrupt hydrophobic interactions by coating hydrophobic molecules with molecules that…
A: Hemoglobin is an oligomeric protein with four polypeptides. The individual polypeptides are joined…
Q: What is the purpose of a bacterial smear? What is the purpose of a bacterial smear? It spreads the…
A: Introduction: The bacterial smear technique is a routine procedure performed to prepare the…
Q: 6. How many mL of 6M HCI solution should be diluted to make 200 mL of a 1.5M solution?
A: Diluted solution can be prepared by diluting the concentrated sample of HCl.
Q: 384 Protein Purification Methods Q4.1- What chemical properties of sodium dodecyl sulfate (SDS)…
A: SDS-PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis) is a type of electrophoresis…
Q: cell division, what do you think is this other explanation for the loss of body fat in cancer…
A: Cancer patient when undergo chemotherapy for treatment have a feeling of nausea and headache along…
Q: II. True or False a. An effort is usually made to fragment the polypeptide backbone first, before…
A: Proteins are the heteropolymers of twenty standard amino acids that differ from each other on the…
Q: 10. Vitamin B6: coenzyme form, functional groups, mechanism of action, biological role, sources,…
A: Enzymes are biological catalysts that catalyse biochemical reactions. Most enzymes are made up of…
Q: The word root erythr/o means? Don't copy
A: The identification of affixes in biological terms can help in the understanding the complex terms.…
Q: is device used to measure temperature.
A: The device used to measure temperature is .........
Q: referred to feedback inhibition as “Goldilocks regulation”. Describe how feedback inhibition keeps…
A: Biochemical pathways and reactions are occurring constantly in cells. These pathways convert the…
Q: Explain the role of zinc in biomolecules .
A: Zinc is the micro nutrient and is considered as the third most abundant element in the body. It has…
Q: 1. By mistake, a student placed microdrops of solutions of alanine and leucine on the same point on…
A: All the 20 standard amino acids except proline have a primary amine group . Ninhydrin is a very…
Q: how important is biochemistry during this time of CoVid 19 pandemic? explain it further.
A: CoVid 19 pandemic is due to an outbreak of viral infection that is caused by SARS CoV 19, a variant…
Q: Discuss how the following buffer systems work in the human body: a) Carbonate buffer system b)…
A: All biological processes are pH dependent. Even a slight change in pH can result in a large change…
Q: You have been observing an insect that defends itself from enemies by secreting a caustic liquid.…
A: Dissociation of formic acid is as follows: HCOOH + H2O ↔ HCOO- + H3O Here, HCOOH –> Formic acid…
Q: A. Will decreasing the amount of energy needed to make an exergonic reaction occur cause the…
A: The thermodynamics of a chemical reaction focuses on the energetics of the products and the…
Q: The phosphate groups in the sugar-phosphate backbone of each strand of a DNA molecule have a pKa of…
A: A DNA molecule is a strand of polynucleotides, where each nucleotide consists of a phosphate group,…
Q: Which of the following statements about trans-fatty acids is/are FALSE? A. Healthier than saturated…
A: Introduction: Fats belong to a chemical group known as lipids. They are found in animals and…
Q: In most cases the peptide bond is in the trans conformation. What statement below best explains this…
A: In proteins the adjacent amino acids are held together via peptide bonds . A peptide bond is an…
Q: The following are true statements regarding passive facilitated transport EXCEPT The process…
A: Diffusion is a defined biological process by which molecules transport through organs. Diffusion is…
Q: can you explain using a drawing presenting the factors like pH, temperature, substrate…
A: Introduction Enzymes are known as bio-catalyst. It increases rate of a chemical reaction by…
Q: An allosterically regulated enzyme in the R state is unable to bind substrate at the active site.…
A: Allosteric site : It is a place or site on an enzyme where the molecule which is not a substrate can…
Q: how would you prepare a 3-fold dilution series
A: Serial dilution : A substance is diluted in a solution step by step in a serial dilution procedure.…
Q: Differentiate the four qualitative tests for proteins BIURET, XANTHOPROTEIC, NINHYDRIN, & MILLON’S
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Using an arrow, draw the site of cleavage for the following peptide that is reacted by: Pepsin…
A: Any peptide or protein can be cleaved by numerous protein digestive enzymes such as pepsin, trypsin,…
Q: Please answer fast In affinity chromatography, which of the following strategies is used to elute…
A: In affinity chromatography, elutions of proteins are done under mild conditions to preserve the…
Q: QUESTIONS: 1. Explain why a vegetable and fruit diet alkalinize blood pH. 2. What are the four major…
A: Diet is a combination of substances that we take throughout a day. These can be a combination of…
Q: Water is present in the cell at a concentration of 55.5 M, whereas P, is present at a concentration…
A: Glyceraldehyde phosphate dehydrogenase is a 37 kDa enzyme which catalyzes sixth step of…
Q: Vitamin B3: coenzyme form, functional groups, mechanism of action, biological role, sources, daily…
A: Vitamins are essential nutrients that are required in lesser amounts but are very essential for…
Q: A cell in a hypertonic solution swells because of a net loss of water by osmosis. True False
A: A hypertonic solution is a solution that contains high solute concentration. Osmosis is the…
Q: A mixture of the following amino acids (glu, leu, val, arg, ser, phe) was obtained upon complete…
A: The following amino acids (glu, leu, val, arg, ser, phe) were obtained from complete hydrolysis of a…
Q: Lipid movement showing migration of the lipid from one monolayer to another is called? flexion…
A: INTRODUCTION lipids - Energy-rich biomolecules made mostly from carbon and hydrogen atoms; generally…
Q: We know that cannabis increases appetite; however, a recent study showed that cannabis consumption…
A: Cannabis is also known as marijuana. It is a psychoactive drug from the Cannabis plant. Clark et.…
Q: Which of the following is/are (a) ketopentose? A. threose B. xylulose C. arabinose D. mannose
A:
Q: A) What would you expect to happen if you added ddCTP to a DNA synthesis reaction in vitro in large…
A: ddCTP is 2',3'-Dideoxycytidine-5'-triphosphate. It does not contain a 3’OH group in the deoxyribose…
Q: 7. What is the definition of a glycoside?
A: The naturally occurring sugars from plants can be found associated with some other groups . These…
Q: Collagen and keratin are examples of what type of protein? A. fibrous B. simple C. globular…
A: Proteins are unbranched polymers constructed from 20 standard amino acids. Amino acid residues…
Q: 2. How many grams of glycerine, C3H5(OH) (MM = 92 g/mol) is mixed with 36 g H₂O to make a solution…
A:
Q: The octapeptide gly-cys-met-asn-lys-ala-tyr-gly was hydrolyzed consecutively by CNBr and then…
A: The octapeptide gly-cys-met-asn-lys-ala-tyr-gly was hydrolyzed consecutively by CNBr and then…
Q: How do you explain Mr Willis’ dark urine and dark stools? Would Mr Willis benefit from any…
A: G6PD(glucose-6-phosphate dehydrogenase) is an enzyme which helps in proper functioning of RBCs. Its…
Q: Which of the following fatty acids will produce propionyl-CoA in its/their last β-oxidation cycle?
A: Fatty acids metabolism involves β-oxidation which happens in the mitochondrial matrix. In…
Q: How to draw titration of amino acids ?what happen when ph increase from low to high? 1. what does it…
A: Amino acids are biomolecules that have an amino group and a carboxyl group linked to the same carbon…
Q: What is the glycosidic linkage present in the dissacharide? A. α-(1→1)-α B. α-(2→1)-β C.…
A: Introduction: The glycosidic linkage is formed by the process of condensation reaction and a bridge…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- In each of the following cases, predict whether the pKa value of the first residue will be upshifted or downshifted relative to the typical pKa value (Table 2.1) based on the microenvironment. Briefly (in no more than 2-3 sentences) explain your reasoning. (a) A His residue adjacent to two Arg residues on the surface of a protein(b) A Cys residue adjacent to an Asp residue in the active site of an enzyme(c) A Lys residue buried in the hydrophobic core of a globular proteinWhich of the following statements regarding Anfinsen's denaturing experiments with ribonuclease A are valid? (i) Exposing the denatured protein to air oxidation and then dialysis to remove urea restored the protein to its original functionality. (ii) Removing urea by dialysis and then allowing air oxidation of the denatured protein restored the protein to its original functionality. (iii) Denaturing the protein with both urea and β-mercaptoethanol yielded an inactive protein. (iv) Protein folding is determined by its primary sequence.What is the expected outcome of the ion exchange chromatography using lysozyme as outlined above? Will the protein be separated successfully or not using the materials stated in the above? Provide a brief explanation to your reasoning please answer correctly, not written assigment
- The answer given in problem 31. 17 is inadequate for me, and I simply don't understand the explanation for the answer... The problem is: Ribosoms were isolated from bacteria grown in a "heavy" medium (13C and 15N) and from bacteria grown in "light" medium (12C and 14N). These 70S ribosoms were added to an in vitro system engaged in protein synthesis. An aliquot removal serveral hours later was analyzed by density-gradient centrifugation. How many bands of 70S ribosoms would you expect to see in the density gradient? Is it possible to give a more comprehensive explanation? :)The last residue of the protein (tail) is Tryptophan, and the first residue (head) is labeled with IAEDANS acceptor. Estimate the length of the protein (head to tail) if the efficiency of RET measures at 0.01 Please show step by step and how to get the 34.014 numberConsider the proteins in Figure 1. Assume they are treated with chymotrypsin to cut them into fragments, and then the fragments are separated by gel electrophoresis. What would the fragment patterns look like in the gel for the three different proteins, assuming we can separate polypeptides that differ in size by very small amounts? Complete Figure 2 below to show the location of the uncut polypeptides (- enzyme lanes) and the cut fragments (+ enzyme lanes).
- The idea that primary sequence determines tertiary structure first came from experiments in the 1950's about: A. the renaturation of RNaseA (ribonuclease A). B. the denaturation of lysozyme. C. the 3D structure of lysozyme. D. the role of Hsp70 in protein folding. E. the role of PDI (protein disulfide isomerase) in protein folding.Describe the molecule shown in the image. Evaluation at Atomic Resolution of the Role of Strain in Destabilizing the Temperature Sensitive T4 Lysozyme Mutant Arg96-->HisYou gathered your data from Lab 4, and found the following values: Total Activity of lysozyme in HEW: 14 units Total Activity of lysozyme in carb 1: 21 units Total amount of protein in HEW: 70 mg Total amount of protein in Carb 1: 15 mg Calculate the extent of purification of lysozyme in Carb 1. 150% 7 fold 70% 15 fold
- How many copies of a protein need to be presentin a cell in order for it to be visible as a band on an SDSgel? Assume that you can load 100 μg of cell extract ontoa gel and that you can detect 10 ng in a single band by sil-ver staining the gel. The concentration of protein in cellsis about 200 mg/mL, and a typical mammalian cell has avolume of about 1000 μm3 and a typical bacterium a vol-ume of about 1 μm3. Given these parameters, calculatethe number of copies of a 120-kd protein that would needto be present in a mammalian cell and in a bacterium inorder to give a detectable band on a gel. You might try anorder-of-magnitude guess before you make the calcula-tions.In addition to in the information given in the pictures, researchers provide further descriptions for the figure stating: Players were also able to restructure b-sheets to improve hydrophobic burial and hydrogen bond quality. Automated methods have difficulty performing major protein restructuring operations to change b-sheet hydrogen-bond patterns, especially once the solution has settled in a local low-energy basin. Players were able to carry out these restructuring operations in such scenarios as strand swapping (Fig. 3) and register shifting. In one strand-swap puzzle, Foldit players were able to get within 1.1 A° of the native structure, with the top-scoring Foldit prediction being 1.4 A° away. A superposition between the starting Foldit puzzle, the top-scoring Foldit solution, and model 1 of the native NMR structure 2kpo (Protein Data Bank) are shown in Fig. 3b. Rosetta's rebuild and refine protocol, however, was unable to get within 2 A ° of the native structure (Fig. 3a, yellow…The following data describe the catalysis of cleavage of peptide bonds in small peptides by the enzyme UTSAse (the arrow indicates the peptide bond cleaved in each case). Substrate Km(mM) kcat(s-1) PAPA↓G 4.0 26 PAPA↓A 1.5 37 PAPA↓F 0.64 18 what features of amino acid sequence dictate the specificity of the proteolytic cleavage? Large hydrophilic R-groups Large hydrophobic R-groups Neutral R-groups Small hydrophilic R-groups Large hydrophobic R-groups Negatively charged R-groups Positively charged R-groups



