The first of the two heterolysis reactions below takes place readily, but the second one does not. Explain why. CH2 CI H3C H3C ECi-AI CI CI CI H3C H3C
Q: O A.D O B.A O C.E OD.C O E. B А + HCI - B с +
A: This question belongs to reaction mechanism branch of organic chemistry. Kinetically controlled…
Q: 4. For the following reaction scheme, please convert the starting material to the corresponding…
A: The reactions taking place are given as,
Q: Predict the Majar products of the reaction 1.Oraw MechanisM ( Label each arrow with its Dattern) Brz…
A: Electrophilic addition :- Addition of bromine to alkene is a type of electrophilic addition…
Q: Rank the following substrates in order from slowest SN1 reaction rate to fastest. Br Br Br Br A В
A: The stability of carbocation is given as substituted with Hetero atom > Tertiary > Secondary…
Q: H3C. КОН, Н-О CH3 H
A: Organic reaction mechanisms SN1 reaction mechanisms.
Q: NBS A NBS B CN -N NBS
A: NBS is N-bromosuccinimide which is used as a brominating agent acting as a source of bromine free…
Q: CH3 + HBr CH=CH2 + HBr `CH3 HC=C¯ + D2O H2O -C=C-CH3 H2SO4
A: Given reactions,
Q: y12. Which reaction in each of the following pairs would you anticipate being faster?
A: We have given that Which reaction in each of the following pairs would you anticipate being faster?…
Q: 6. Determine whether each reaction occurs by an SN2, SNI, EI, and/or E2 reaction. Provide a detailed…
A: The higher the temperature, the more likely elimination occurs. The lower the temperature, the more…
Q: Br „CH3 CH3CH2OH H3C"
A: Organic reaction mechanisms
Q: 1. For each of the following pairs of SN2 reactions, indicate which reaction occurs faster: a)…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Br NaOCH3
A:
Q: Consider the substitution reaction shown below. By what mechanism will the reaction proceed? Br…
A: There occur substitution reaction.
Q: Using curved-arrow notation, draw out the mechanism for the following reaction. Be sure to include…
A:
Q: Which conditions will cause the interconversion shown? how? H2 / Lindar catalyst a. LI/ NH3lig) H2 /…
A: Answer :- b) Li/NH3(liq)…
Q: Give a clear handwritten mechanism and product for given below Sn2 reaction..a reaction shows all…
A: SN2 is reaction mechanisms:
Q: Do the reactions below proceed in good yield from left to right as shown?
A:
Q: /hich reaction sequence will most successfully generate the product shown belov (1) HNO3/H2SO4 (A)…
A: the solution is given below -
Q: .CO2CH3 CO2CH3 H20 | H2SO4 CO2CH3 .CO2CH3 РСС HO 3
A: There is some printing mistake in question you can check in image Ist reaction is diels-elder…
Q: 6. Determine whether each reaction occurs by an Sn2, Sn1, E1, and/or E2 reaction. Provide a detailed…
A: Hello. There are multiple subparts to this question. The first three subparts have been solved. To…
Q: (i) which reaction among the pair, is fastest? Explain your (ii) predict the products of faster…
A:
Q: Br A Br B Br C D -Br
A: SN1 reaction The SN1 reaction is a nucleophilic substitution reaction where the rate-determining…
Q: Which mechanism is consistent with the reaction below? (CH3)3CBr+OH-→(CH3)3COH+Br- 1.…
A: Carbocation can be defined as the species in which carbon atom bear positive charge. It can be…
Q: Draw the free energy diagram for each of the reactions in Problem 8.33. For each diagram, include…
A: E1 E1 is a unimolecular Elimination reaction. Here, the removal of an HX take place, this results in…
Q: (a) PHOOC-CH,-C-CH,-COOP. (1) KCN, A (2) H,0® (o) СООН Onl
A:
Q: 6. Determine whether each reaction occurs by an SN2, SN1, E1, and/or E2 reaction. Provide a detailed…
A: → In above base is weak so it show SN2 reaction.
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.
A: Given : structure of reactant. This is SN2 reaction. Removal of cl- and attack of Nucleophile will…
Q: CHa Br, FeBr, NHCOCH, heat H3C CH, SO,, H-SO, Br CF3 heat
A:
Q: The electrophilic addition of Br, to several alkenes was examined. Explain why the relative reaction…
A: In the reaction of Br2 with an alkene, an alkene is a nucleophile and Br is an electrophile.
Q: Draw a detailed, step-wise mechanism for the reaction shown below. SHOW ALL BOND-FORMING AND…
A: In the above reaction, the OH- present in the base NaOH will attack on the carbon making ester bond…
Q: Use curved arrows to show how E is converted to F by a two-stepreaction sequence consisting of a…
A: The reaction involves [1,5] sigmatropic rearrangement and [4+2] cycloaddition. A C-H bond is broken…
Q: (b) Show the mechanism of this reaction using proper arrow push notation. You mu intermediates,…
A: We will write the mechanism of the given conversion
Q: For the dehydrohalogenation (E2) reaction shown, draw the major organic product, including…
A: E2 reaction is a bimolecular elimination reaction to give an alkene. In the transition state, the…
Q: Draw the products of the two step reaction sequence shown below. Use dash and/or wedge bonds to…
A:
Q: 3. Provide full mechanism (curved arrows and intermediates) for the following reaction. OCH3 1.…
A: Answer 3 ) The mechanism for the given reaction = ? Note : As per our company guidelines, we are…
Q: a) b) НО ОН PCC CH2Cl2
A:
Q: Could the reactions shown here take place by a concerted mechanism?
A:
Q: Which reaction proceed according to an E2 mechanism? toe. 3D II ethanol heat Br heat Br DMSO towe.…
A: E2 elimination generally occurs in the presence of heat, complete in one step and form alkene. As…
Q: ProVide the major prochuct for each following reactuns H more Than one 5 formed, Circk the major…
A: 1) first allylic cation is formed which is electrophile and it attack on Benzene .Ring and product…
Q: Which of the following substrates will have the fastest rate of reaction with NaCN in DMSO? O A) B)…
A: Given: Alkyl halide + NaCN in polar aprotic solvent DMSO
Q: Draw the structure of all products of the mechanism below. H-C=C -OH + H-Ö-CH, H. it
A: Due to presence of lone pair electrons in the O atom, alcohols like methanol can act as nucleophiles…
Q: Rank the following substrates in order from slowest SN2 reaction rate to fastest. Br Br Br H3C Br A…
A: SN2 stands for nucleophilic substitution reaction of order 2. In this reaction, the rate of the…
Q: Which of the following alkene addition reactions occur/s specifically in syn fashion?…
A: If we say a reactions occur/s specifically in syn fashion, the adding atoms should be on same side.…
Q: Determine the step by step mechanism of the major product of the following reaction below.…
A: Alkanes show characteristic reaction on treating with halogen molecules in light. This is recognized…
Q: For SN1 Explain the order in which 1o (primary) alkyl halides reacted (fastest to slowest) and…
A: The rate of the SN1 reactions depends upon the concentration of alkyl halide only. This kind of…
Q: 1. CH3M9BR 2. H30+
A: The reagent used are the Grignard reagent. Follow the below picture for mechanism.
Q: Please show a plausible mechanism. This question is related to Pericyclic reactions CH3 CH3 550 °C…
A: First step of the reaction involves reversible electrocyclic sing opening to a ketene aldehyde.…
Q: i) NaNO2, HCI ii) H2O, heat NH2 (e) H3C NaNH2 NH3 (1) (f)
A: Major products of following organic reaction
Q: Identify the nucleophile in the reaction below: ÇH3 C-OH ÇH3 ČH3 + 2 H20 ČH3 + H30 O CH3 I. III. H3O…
A: Answer : II. H2O
Q: Find major product of this - show reaction mechanism and all selectivity. а.) Brz hy b.) Rha COAC)4…
A: Let us solve these reactions.
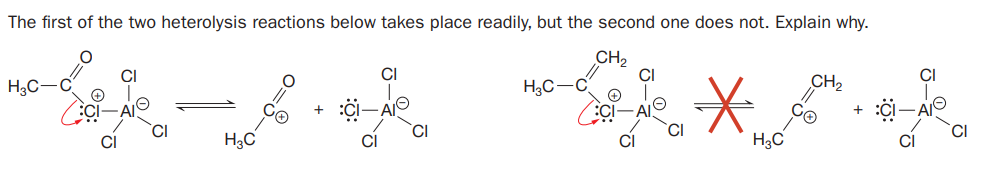
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. The reaction of CH3CH2Br with NaOH occurs by this mechanism.Explain the possible mechanism (Sn1/Sn2/E1/E2) given the reaction. Show the transfer of electrons/groups, and name the product.Shown below are two reactions; Reaction 1 is a Friedel-Crafts acylation while Reaction 2 is a Friedel- Crafts alkylation. State which reaction will occur faster and justify your answer, with words and structures. Explain these all: e- density / Deactivation / Resonance stable / faster the carbonation formed because it's gonna have smaller activation energy
- Which of the following (I-III) is not a step in the mechanism of the reaction shown below.For each of the following reactions, state the mechanism the reaction will follow (E1,E2, SN1, SN2, etc.) and givethe major product(s) of the reaction.Complete all the elimination reactions/show the products, and for each reaction clearly and thoroughly explain which mechanism (E1, E2, SN1, etc.) is predominant and how it effects the product formation.
- Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?For which reaction mechanisms—SN1, SN2, E1, or E2—are each of thefollowing statement true? A statement may be true for one or moremechanisms. The mechanism involves carbocation intermediates.Shown below are two reactions; Reaction 1 is a Friedel-Crafts acylation while Reaction 2 is a Friedel- Crafts alkylation. State which reaction will occur faster and justify your answer, with words and structures. Mention: e- density / Deactivation / Resonance stable / faster the carbonation formed because it's gonna have smaller activation energy
- What is the complete mechanism using curved arrow formalism of the two products shown below? Explain why one is major and the other is minor product formation.Complete the following reactions with the majority products and the missing reaction conditions. Indicate stereochemistry if applicable. Also indicate the type of reaction that took place (SN1, SN2, E1, E2, E1bc)Give the major organic product(s) for each step of the following sequence of reactions



