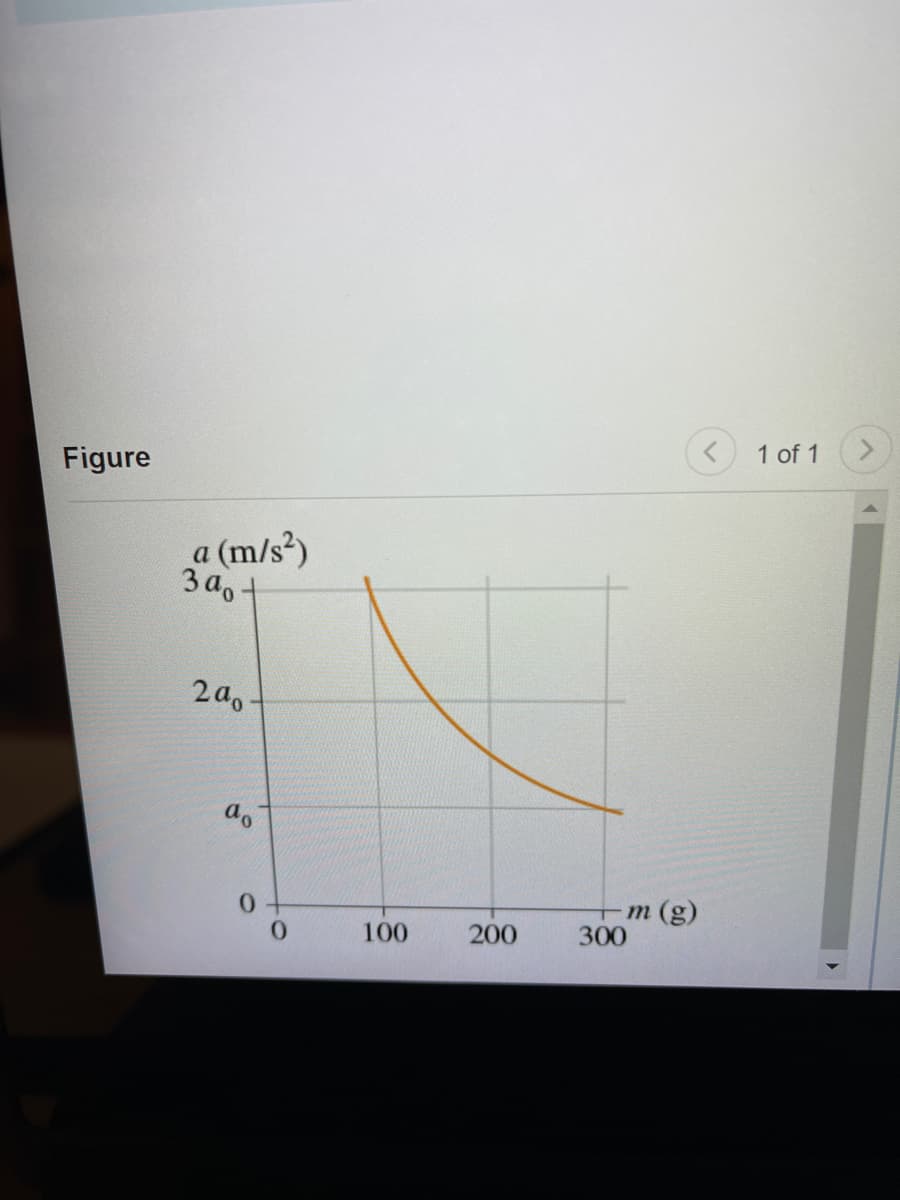
The following figure shows the acceleration of objects of different mass that experience the same force. Suppose a0 = 6.0m/s^2. What is the magnitude of the force? Express the answer in two significant figures.
Q: Which portion of this heating curve for water should have the least steep slope?
A: Heat capacity is defined as the amount of heat required to raise the temperature by 1 degree…
Q: The figure below shows the unusual path of a confused football player. After receiving a kickoff at…
A:
Q: Melissa is a 15-year-old high school student. Over the last week, she had been feeling tired and…
A: Immunity is defined as the ability of an organism to resist a particular infection by the action of…
Q: Hiker Harry walks 4 km east then 7 km 25o north of west. What is the magnitude and direction of…
A:
Q: Starting with the following equation, BCl₃(g) + LiAlH₄(s) → B₂H₆(g) + LiAlCl₄(s) calculate the…
A:
Q: A polyprotic acid is an acid that __________. A) Contains more than one hydrogen B) Can react…
A: Polyprotic acid is an acid and it can donate the multiple protons, that means it contains rich…
Q: OVERVIEW- In order to monitor certain bodily functions, patients are sometimes given radioactively…
A: In a neutral atom, Atomic number = Number of protons = number of electrons Also, in an atom or ion,…
Q: Convert 32.5% to parts per thousand (ppt).
A: ppt = 10 x percentage
Q: Aqueous solutions of magnesium nitrate and sodium phosphate are mixed together, forming solid…
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both…
Q: 1. Name and encircle the functional groups present in each molecule. a) Histrionicotoxin (poison…
A: Please find the answer attached as handwritten note
Q: You are given sodium acetate, NaOH (1 M), NaHCO3, and Na2CO3. Determine which two of these four you…
A: Given: pH of buffer = 9.9 Concentration of buffer = 0.1 M And volume of buffer solution = 100 mL =…
Q: In the figure the ideal batteries have emfs & = 21.3 V, E2 = 9.68 V, and ɛg = 5.10 V, and the…
A:
Q: Kepler's 1st law says that our Solar System's planets orbit in ellipses around the Sun where the…
A: Given: The perihelion distance=Rp=2 AU The semi-major axis=a=1.5 AU
Q: A 275-g sample of nickel at l00.0°C is placed in 100.0 mL of water at 22.0°C. What is the final…
A: Heat gained by a substance = mass x specific heat capacity x (Tfinal - Tinitial) Ni is at a higher…
Q: both chemical and flame tests are used to positively identify carbon dioxide gas. true false
A: Yes both chemical and flame test are used to positively identify carbon dioxide gas so it is true .…
Q: Potassium hydroxide (KOH) has a lattice energy of −789.0 kJ/mol−789.0 kJ/mol and a heat of hydration…
A: Volume of solution could be heated can be calculated from the energy required for heating and the…
Q: You accidentally knock a full bucket of water off the side of the well shown in Figure. The bucket…
A:
Q: Use the electron arrangement interactive to determine the order in which the orbitals are filled.…
A: Electrons are filled in the subshells in increasing order of their energy. Which is decided by (n+l)…
Q: A pulsar is a rapidly rotating neutron star that emits a radio beam the way a lighthouse emits a…
A:
Q: Determine the damping coefficient of a spring-mass-damper system with a mass of 150 kg and stiffness…
A: Given, Mass, m=150kg Stiffness, k=2700N/m Time period, T=1.8s Natural angular frequency,…
Q: When a mixture of 10.5 g of acetylene (C2H2) and 10.5 g of oxygen (O2) is ignited, the resultant…
A: Introduction If the mass of each reacting species is given, then the mass of product species can be…
Q: Formula for density is D=m/v If an object with a mass of 35 grams has a volume of 70cm cubed and its…
A:
Q: Learning Goal: To apply the law of conservation of energy to an object launched upward in the…
A: When there is no energy exchange between a system and the environment, then the total energy of the…
Q: block with mass m = 12.0kg sits at the top of a ramp with height y1=8.5m. The ramp makes an angle θ…
A: Work done = force × displacement
Q: A 2.5 kg , 20-cm-diameter turntable rotates at 110 rpm on frictionless bearings. Two 510 g blocks…
A:
Q: Write a balanced chemical equation based on the following description: the reaction of powdered…
A:
Q: One day, after pulling down your window shade, you notice that sunlight is passing through a pinhole…
A: Given data :Central distance, w=2 cm=0.02 mDistance from the window shade to the wall, D=4 mVisisble…
Q: Blood flows through a major artery at 1.0 m/s over a distance of 0.40 m, then at 0.60 m/s for…
A:
Q: List the following compounds from highest boiling to lowest boiling: Rank the compounds from highest…
A: The factors that affect the boiling point of a compound are, Molecular mass Hydrogen bonding Polar…
Q: Be sure to answer all parts. Identify the correct Lewis electron-dot symbol for the ion formed…
A: Lewis’s structure: In the Lewis structure of an atom, we write the valance electron by a dot. To…
Q: A solution has a density of 0.889 g/mL and has 5.9 g of toluene (C7H8) and 231 g of benzene.…
A: The given data is as follows: The density of the solution = 0.889 g/mL The mass of toluene = 5.9 g…
Q: The hardness of water is usually expressed in parts per million by mass of CaCO3. What is the…
A: Parts per million (ppm) :- The number of parts by weight solute present in million (106) parts by…
Q: A 1-kilogram mass is attached to a spring whose constant is 18 N/m, and the entire system is then…
A: Mass of object m=1 Kg, Force constant k=18 N/m. Damping force F=11 v where v is the instantaneous…
Q: For the reaction A(g) + 1/2B(g) > 2C(g) When C is increasing at a rate of 0.025 M/s, how fast is B…
A:
Q: In fruit flies red (A) eyes are dominant to apricot (a) eyes, and normal (P) wings and dominant to…
A: Given: In fruit flies red (A) eyes are dominant to apricot (a) eyes, and normal (P) wings and…
Q: If you had to prioritize the removal of plants from an area, rank the following in order of highest…
A: Given below is the arrangement of plants from higher priority to lower priority according to their…
Q: A meteoroid changed velocity from 1.0 km/s to 1.8 km/s in 0.03 seconds. What is the acceleration of…
A: Initial velocity of meteoroid (u) = 1 km/s = 1000 m/s Final velocity of meteoroid (v) = 1.8 km/s=…
Q: 1) Nucleotide differences for a DNA sequence on chromosome 1 are shown for five species. Which…
A: A number of nucleotide differences in the genes or in the genes of two species can reveal important…
Q: A snowmobile with an initial speed of 5.6 m/s travels 24.0 m in 2.0 s. What final speed does it…
A: Given data: Initial velocity (V0) = 5.6 m/s Distance traveled (x) = 24.0 m Time (t) = 2.0 s…
Q: Write the balanced dissociation equation for solid iron(II) carbonate in aqueous solution. If it…
A: Salt dissociates to give corresponding ions in aqueous solutions. Dissociation of potassium…
Q: The facial series of the skull comes in contact with the brain. True False
A: Introduction:- The brain is the organ that acts as the central nervous system for all vertebrates…
Q: The specific rotation for compound D is -8.5° dm-1(g/mL)-1, what is the specific rotation of…
A: The specific rotation ([α]) of a compound is mathematically expressed as: [α]=αC×l…
Q: Which compound is the correct structure for the given NMR? A 1. B 2. C (1) (3) (2) (2) (2) 11 10 9…
A: Proton NMR spectroscopy is a technique used for the identification of a compound.
Q: a) Consider the reaction of hydrogen bromide with oxygen, given below: 4 HBr(g) + 1 O2(g) ---->2…
A: Given: 4 HBr(g) + 1 O2(g) ---->2 Br2(g) + 2 H2O(g)
Q: the owing question. Color Element Red Strontium Orange Calcium Yellow Sodium Green Copper Potassium…
A: When electron fall back from excited state to lower state ,they emit energy in form of light.
Q: 7. Explain the significance of AB and Rh blood groups in blood transfusions Discuss a variety of…
A: The classification of blood into A,B, AB and 0 groups, depending on the presence and absence of A…
Q: EXERCISE 6.9 Gaining Insight into Depositional Environments of Sedimentary Rocks Name: Section:…
A: Different types of sediments are deposited/found at different types of places. The above given…
Q: The indicated organ is the Copyright Nielsen & Miller
A: The structural and functional centre for the entire system comprising brain and spinal cord is…
Q: Use standard enthalpies of formation to calculate ΔH∘rxnΔHrxn∘ for the following reaction:…
A: Given reaction: CaO(s)+CO2(g)→CaCO3(s) We have to calculate the ΔH∘rxn using the standard enthalpies…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images
