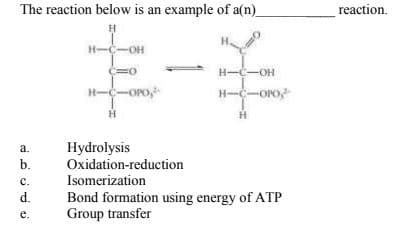
The reaction below is an exampie ol an) reaction. H-C-OH H--OH H-C-OPo, H-C-Oro Hydrolysis Oxidation-reduction Isomerization Bond formation using energy of ATP Group transfer a. b. с. d. e.
Q: Define the following terms: a. electron transport chain b. oxidation-reduction reactions c.…
A: Explanation for electron transport chain, oxidation-reduction reactions, conjugate redox pair,…
Q: ATP hydrolysis is _______ because ________. ATP + H2O ADP + Pi a) Exergonic; Its hydrolysis…
A: ATP is the chemical link between catabolism and anabolism. It is the energy currency of the living…
Q: (d) Derive Michaelis-Menten equation of enzymatic reaction.
A: Michaelis Menten equation shows the relationship between the concentration of the substrate and…
Q: This reaction cannot go to completion as drawn. Why? H2N + CI HN- A) The amine will act as a Lewis…
A: This amide synthesis reaction: They are commonly synthesised by the reaction of acid chlorides with…
Q: Which of the following reactions is feasible at pH 7 ? And, why? FAD + NADH + H+ FADH2 + NAD+ FADH2…
A: Nicotinamide adenine dinucleotide (NAD) and Flavin adenine dinucleotide (FAD) are coenzymes derived…
Q: Beginning with the 1st tetrahedral intermediate, show the complete steps in chymotrypsin mechanism…
A: The peptides are formed by linking different amino acids through peptide bonds. The enzyme involved…
Q: Which statement defines a coenzyme
A: Answer - Option E - An organic molecule closely associated with enzymes
Q: 6. Enzyme X exhibits maximum activity at pH = 6.9. X shows a fairly sharp decrease in its activity…
A: Enzymes are biocatalysts that speed up biological reactions. Active site of enzymes are the pockets…
Q: Identify the statements that are TRUE regarding Catabolic reactions. SELECT ALL THAT APPLY…
A: Catabolic reactions are the reactions which breakdown complex substances into smaller units.…
Q: | Explain how enzymes increare the peed of reaction Explain the most important reason why enzymer…
A: Hi, Thanku for the question. According to the company guidelines ,we can only solve 1 questions at a…
Q: Which of the following is incorrect? a. None; all the other choices are correct O b. Reactions with…
A: Michaelis menton kinetics help in determining the enzymes reaction rate and km. It is plotted by…
Q: In the reaction Na + Cl à Na+ + Cl-, which component is said to become ‘oxidized’ and which is…
A: In this question, we have to define oxidation and reduction in NaCl
Q: Which of the following classifications apply to the formation of 3-hydroxybutyrate from…
A: Ketogenesis It is the process of formation of ketone bodies in the liver during fasting or…
Q: If the AG of a reaction was -31.45 kJoules, you would know that: the reaction most likely leads to…
A: ANSWER;- Correct answer is B. Explain;- If the ΔG of a reaction was -31.45 k joules, you would…
Q: In biological systems, ATP functions bya. providing the energy to drive endergonic reactions.b.…
A: ATP or adenosine triphosphate is known as energy currency of the cells. The molecule contains three…
Q: A certain anabolic biochemical reaction A has AG=17.8 kJ-mol, and is always coupled to another…
A: Some of the reactions that take place inside living organism require an input of energy to take…
Q: Which statements are true and which are false? Modify each false statement so that it reads…
A: Chemical thermodynamics is the study of the interrelation of heat and work with chemical reactions…
Q: Consider the following metabolic pathway and answer the questions. CH,OPO, CHOPO, H--OH HO-C-H…
A: Hi, thank you for posting the question on Bartleby. As per the guidelines, we can answer up to three…
Q: In the partial reactions shown below, is the reactant undergoing oxidation or reduction? (a) Ç00 COO…
A: Oxidation (in terms of oxygen and hydrogen atom transfer): Oxidation in terms of atom transfer is…
Q: What type of reactions is represented below? CH,OH CH2OH C=0 H-C-OH HO-C-H HO-C-H CH,OH CH2OH O…
A: This is a ketotetrose which is a tetrose having a single ketone group at the 2-position.
Q: Which of the following statement is FALSE? a.Catabolic pathways are energetically favorable. b.All…
A: Catabolic pathways are those pathways which function to breakdown complex molecules into simpler…
Q: For the experiments to test amylase activity, you started out with a starch solution to which you…
A: Enzymes in other words are known as biological catalysts, these are mainly used in the digestive…
Q: Lactose--->galactose+glucose represents an enzymatic reaction. What statement about this reaction is…
A: Lactase, also known as lactase-phlorizin hydrolase, is an enzyme found in animals' small intestine…
Q: The reaction ATP to ADP + Pi is typically part of a(n) ________ reaction. a) free radical b)…
A: Enzymes mediate six primary kinds of biochemical processes in living systems. These include group…
Q: 1.) Evaluate the general reaction pictured and select the statement that best explains the process…
A: Introduction:- Chemical reactions occur when chemical bonds between atoms are formed or broken. The…
Q: Reaction below requires a catalytic amount of NAD+. OH HO HO. ОН ОН О `OH
A: Intramolecular cyclization reaction that taking place here involves multiple hydrogenation and…
Q: In the elimination-addition mechanism for phosphate group transfer reactions, the intermediate would…
A: Phosphate ester and anhydride phosphorus atom reactions form the chemical basis for many of the most…
Q: Draw the complete molecular structure of the hydrolysis of a phosphate from ATP. Give the value and…
A:
Q: Curve A (red), is enzyme catalyzed reaction treated with a compound. Curve B (blue), is the same…
A: The given graph represents the Lineweaver-Burk plot. Lineweaver-Burk plot is the plot between 1/V…
Q: The breakdown of large molecules by the enzymatic addition of water is an example of what kind of…
A: large molecules are broken down into small molecules in a process known as hydrolysis, which means…
Q: Determine the type of biochemical reaction that occurs in the reaction shown below. O redox…
A: In the given question, a cyclic pyranose structure is given. It is a pyranose because it has 6…
Q: Propose a mechanism for the following reaction:
A: Asparagine and glutamine are derived from aspartic acid and glutamic acid by the action of the…
Q: If a reaction has a Delta G of 42: a The reaction is energy requiring Ob. The reaction is exergonic…
A: Enzymology is the branch of science which deals with the study of enzymes. It reveals how an enzyme…
Q: What is the role of the small organic non-protein compounds called coenzymes? A. Coenzymes are…
A: Enzymes are the biocatalyst that can increase the rate of biochemical reaction without itself being…
Q: H20 f) OH Chemistry Steps Chemistry Stes Br Chemistry Steps Chemistry Steps Chemistry Steps Br h)…
A: Solution is attached below.
Q: Given the following reaction, identify the class of the enzyme involved. CO (CH;2 ATP + NE H- NH,…
A: The international commission on enzymes has classified the enzymes based on the type of reactions…
Q: Krebs Cycle / Tricarboxylic Acid Cycle Enzyme Function Step Reactants / Substrate Products 1
A: Introduction: The Krebs cycle occurs in mitochondria where the acetyl group of acetyl-CoA is…
Q: Use numbers 1 to 5 to identify each carbon atom in the product of this reaction. What coenzyme is…
A: Amino acids have two functional group namely carboxyl and amino group. The above compound is a…
Q: me 1: CH3 HNO, / H,SO, NaOC1 CH HO, CH,OH /H* NO, HNO,/ H,SO, OCH NaOC1 HNO, H,SO он но. NO2 NO2 CH…
A: A metabolic pathway is a step by step series of interconnected biochemical reactions that convert a…
Q: the substrates must be a high erall reaction is exergonic
A: Answer Substrate level phosphorylation in which One of the substrates must be a higher free energy…
Q: Consider the mechanism below. (i) Identify Asp X's function in the first step of the reaction. Ex…
A: The reaction mechanism shown here is an enzyme catalyzed reaction to synthesize sugar acid and here…
Q: Catalysts are believed to lower the activation energy of thetransition state in a chemical reaction.…
A: Proteins that function as biological catalysts are known as enzymes (biocatalysts). Catalysts help…
Q: Hydrolysis is an example of which type of reaction?
A: Hydrolysis is the chemical breakdown of substances by water . Catabolic reactions often employ…
Q: For the reaction shown, which statement is true? B Transition state EA Reactants AB AG<0 Products…
A: Reactions occur when there are changes in the reactants that break the bonds and make new bonds to…
Q: Which of the following statements is true for all exergonic reactions? O a. The reaction proceeds…
A: Enzymes are a class of proteins except for ribozymes that increase the pace of the chemical…
Q: Given the active site diagram below, which of the indicated components is/are necessary for proton…
A:
Q: Explain briefly the following: 1-Oxidation-Reduction Reaction. 2- The influence of temperature on…
A: Oxidation is the reaction in which an atom loses its electrons. Reduction is the process by which an…
Q: For the reaction shown in Figure 1, name molecules X and Y. What do A and B represent? Name the…
A: The glycolysis is the first step of cellular respiration that occurs within the cytoplasm of every…
Q: What type of reaction is the third step (formation of keto group from a secondary alcohol) of fatty…
A:
Q: P P₂ 2 E*-P₁-P₂ E*-P₂ E* E E-S₁ E-S₁-S₂ Which of these 2 options represents a Sequential Ordered…
A: Enzymes are very important ad highly effective catalysts, having the ability to enhance the reaction…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Which statement is FALSE? a. For S P, a catalyst shifts the reaction equilibrium to the right. b. After a reaction, the enzyme involved becomes available to catalyze the reaction again. c. A reaction may not occur at a detectable rate even though it has a favorable equilibrium. d. Substrate binds to an enzyme's active site. e. Lowering the temperature of a reaction will lower the reaction rate.Which of the following statement is FALSE? a.Catabolic pathways are energetically favorable. b.All energetically unfavourable reactions have a positive delta G. c.Reactions with a positive delta G can occur through reaction coupling with an energetically favourable reaction such that the overall delta G is negative. d.Carbon monoxide is the most energetically favourable form of carbon under normal conditions.Some cool enzyme (CE) catalyzes the conversion of compound F to compound A. Which of the following must be true of CE? a) CE can facilitate conversion of compound A into compound F. b) CE binds more tightly to the substrate F than to the product A. c) CE decreases the energy of the transition state between compounds F and A. d) CE facilitates net conversion of F to A whenever F is present.
- The hydrolysis of ATP has a large negative ΔG'°; nevertheless it is stable in solution due to: a. entropy stabilization. b. ionization of the phosphates. c. resonance stabilization. d. the hydrolysis reaction being endergonic. e. the hydrolysis reaction having a large activation energy.Which statements are true and which are false? Modify each false statement so that it reads correctly. a. In a closed system, neither energy nor matter is exchanged with the surroundings. b. State functions are independent of the pathway. c. A process is isothermic if ΔH = 0. d. The sign and magnitude of ΔG give important information about the direction and rate of a reaction. e. At equilibrium, ΔG = ΔG°. f. For two reactions to be coupled, they must have a common intermediate.What is an example where molecules or aroma within a cell physically overcome some barrier to their reaction going forward thag is exergonic one? (hint: think about what a catalyst physically does during a reaction).
- Which one of the following statements is true of enzyme catalysts? a. Their catalytic activity is independent of pH. b. They are generally equally active on D and L isomers of a given substrate. c. They can increase the equilibrium constant for a given reaction by a thousand fold or more. d. They can increase the reaction rate for a given reaction by a thousand fold or more. e. To be effective, they must be present at the same concentration as their substrate.You are observing an enzyme driven reaction. To the reaction mixture you add a chemical X which inhibits the reaction. If you add more substrate, the reaction rate approaches the Vmax of the uninhibited reaction. Furthermore, the structure of X is similar to the natural substrate. What kind of inhibitor is X?Which of the followingdescribe superior properties of enzymes (biological catalysts) over traditional chemical catalysts? a. They are mostly and generally operative under mild temperature, pressure, and pH conditions b. They are regulated only by substrate concentration c. They do not effect the reaction equilibrium, but lower the reaction's activation energy d. They are recycled at the end of the reaction Choose all that apply
- Explain briefly the following: 1-Oxidation-Reduction Reaction. 2- The influence of temperature on enzyme action.“Enzyme increases the rate of a reaction by lowering the activation energy. In doing so, it also changes the equilibrium of that reaction.” Do you agree/disagree with this statement? And why?Which of the following best indicates a reason why Hisneeds to be a good proton donor (not acceptor) in the catalytic triad mechanism? So that it can create the alkoxide ion. So that it can create a hydroxyde ion. None of the other options is suitable because His's only role is to be a proton acceptor. So that the first product forms and leaves with the correct number of H's. So that it can stabilize the transition state.

