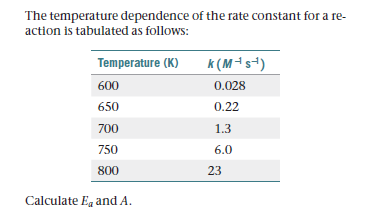
The temperature dependence of the rate constant for a re- action is tabulated as follows: Temperature (K) k (M-s-) 600 0.028 650 0.22 700 1.3 750 6.0 800 23 Calculate Eq and A.
Q: The number of vacancies in lead at 25 °C is 1.66x1019/m3 and at 227°C the number of vacancies is…
A: Let Ea is the activation energy for the vacancy formation.
Q: If the zero order elimination is 0.125 hr -1 and the t1/2 (hr) is 7.5 hr, the initial conc. Co is-…
A: Dear student Since you have posted multiple questions., we will solve only 1st question for you if…
Q: The manganese(II)-catalyzed redox reaction between cerium(IV) and thallium(I) is thought to proceed…
A: In a multistep mechanism, the slow step is the rate determining step.
Q: The value of steric factor, P for the reaction between NO and Cl2 giving NOCl and Cl is 0.018 at 298…
A: Given = steric factor= 0.018 experimental value of A is 4.0 x 109 L mol-1 s-1
Q: S₂02 +31-2SO²- + 13 R = K[S₂0²-][1-] Trial [S₂0,2], (M) [1], (M) Initial Rate (M/s) 1 0.018 0.036…
A: What is the rate constant for the reaction given the rate law and data ?
Q: 3. In a study of fumarase from pig the following kinetic parameters were found in one direction of…
A: I have attached the detailed solution below. At equilibrium v=0.
Q: For the reversible reaction A+B C + D the enthalpy change of the forward reaction is +21 kJ/mol. The…
A: The enthalpy of a reaction is the energy change when reactant is converted to the product. The…
Q: Why do some reactions with -AG values not appear to occur under those conditions? The reaction…
A: Delta G negative that means the reaction is spontaneous but some reactions does not occur even when…
Q: Determine the rate law, rate constant(k) of the reaction with the given information. What's the…
A: Rate law for the given equation is: Order of the reaction = m+n+p 1) To get m value, consider…
Q: The composition of a liquid-phase reaction 24-B was monitored spectrophotometrically. The following…
A: Given, Reaction : 2A→B t/min 0 10 20 30 40 ∞ Concentration (mol/L) 0 0.089 0.153 0.200 0.230…
Q: Kinetic data was collected for the following reaction at 298 K. 2AB(g) → A2(g) + B2(g) Time (s) [AB]…
A: The kinetics data given is,
Q: The following initial rate data are for the reaction of hypochlorite ion with iodide ion in 1 M…
A: We know that Rate constant : Rate constant is the proportionality constant in the equation that…
Q: For a certain reaction, a graph in which the natural log of vapor pressure is plotted along the…
A:
Q: (i) (ii) (iii) Construct a suitable plot and determine AH and A* S. Determine the Gibbs energy of…
A: b) i) slope = - ∆H0 / R = - 12174 or - ∆H0 / 8.314 = - 12174 or ∆H0 = 101215 J / mole = 101.2 KJ /…
Q: Q4. lodide ions are oxidised to iodine by hydrogen peroxide in acidic conditions. H₂O₂(aq) + 2H+(aq)…
A:
Q: Please watch this video and solve the following two questions (…
A:
Q: 2]o (M) [OH-]o (M) Initial Rate (M/s) 1 0.0500 0.100 5.75 x 10-2 2 0.100 0.100 2.30…
A:
Q: Calculate the activation energy, in kJ/mol, for the redox reaction Sn2+ + 2C03+ Sn4+ + 2Co2+ ---->…
A: From the Arrhenius equation: k1 and k2 are rate constants at temperature T1 and T2. Ea: Activation…
Q: It is decided to test the exothermic reaction A P with 8000 kg of reactant A and an initial…
A: 1) we have to assume the given reaction carried out in a isothemal reactor that means the reactor is…
Q: ) use integral rate law to show that the Successive half-live s reaction keep of Second oider…
A: The half-life of a reaction, t1/2, is the amount of time needed for a reactant concentration to…
Q: Which of the following reaction will be NONSPONTANEOUS? a. Br2(aq) + 2I-(aq) → I2(aq) + 2Br-(aq) b.…
A:
Q: 17-28. At 400 K oxalic acid decomposes according to H,C,O,(g) → CO,(g) + HCHO,(g) The rate of this…
A: The given reaction is :
Q: Begining with 1 M concentrations of each reactant and product at pH=7 and 25.0 degrees C, calculate…
A:
Q: pters 9 and 11) References) The following experimental data were obtained for the reaction of NH,*…
A: Given,
Q: What is the rate for the first order reaction A → Products when [A] = 0.259 M? (k = 0.360 1/min).…
A: In first order reaction rate depends upon the concentration of one reactant only
Q: OTMS OTf Cu(OAc)2 (y mol%) Additive DCE [0,5M] 70°C 0,5 to 16h
A: This reaction is an arylation of the silyl-protected enolate, which gives the following reaction.
Q: In the reaction --> S + P --> Q + V --> you measure the concentrations of all the compoents in the…
A: The reaction given is, => S + P ↔ Q + V ΔGo = +0.13 KJ/mol = 130…
Q: Consider the reaction: 4 NH3(g) + 5 O2(g) → 4 NO₂(g) + 6 H₂O(1) AfH° -46.19 0.00 90.37 -285.85…
A: Using the given values of heat of formation, we have to calculate the enthalpy change for the…
Q: 3. Some values of the rate constant for the alkaline hydrolysis of ethyl iodide over the temperature…
A: Answer : - Energy of activation = Ea = 83.140 kJ/mol Pre-exponential Factor = A = 1623346…
Q: 2) Use the data below to determine the rate law and the value of k for the following reaction H2O2…
A:
Q: State 2 important requirements for studying fast gas phase reactions and explain your answer.
A: Fast reaction can be studied by flow methods. Those reaction which carried out in short span of time…
Q: "A solid catalyst with highly dispersed active metal on supported material is typically considered…
A:
Q: For A⟶products, time and concentration data were collected and plotted as shown. [?] (?) t (s)…
A:
Q: Eyring equation in term of concentration can be written as ats キH kT %3D where n= 1 or 2 Given: E =…
A: Entropy is used to determine the randomness and disorderness of the system.
Q: 8) B + -C forward rate constant: ka, backward rate constant: kb Reactions are 1st order in both…
A: Intrpretation - To determine the half life for the forward and backward reaction which is given…
Q: 6. The determines the rate constant, while determines the favorability of a reaction under standard…
A: As per the Arrhenius equation, the activation energy and rate constant are related together. So, the…
Q: What is the value of K. expression for the following reaction? Iz(aq) + 5HsIOg(aq) 5710;"(aq) + 9H2O…
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: 2. You may have clearly been able to distinguish the difference in the time required for the initial…
A: Given, HCl reacts with Zinc .
Q: 1) MeJNH " 8 = 8 5 5 8 2) NaBH4 Reaction3 is B U
A: We have to find the product of the reaction.
Q: 1. Draw the following potentialenergy diagrams and fill in the missing values. Potential energy of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Determine the rate law and the value of k for the following reaction using the data provided.…
A:
Q: Calculate AT From the data, calculate q, the number of Joules absorbed by the acid-base…
A: Balanced chemical equation, HCl (aq) + NaOH (aq) → H2O (l) + NaCl (aq)
Q: The manganese(II)-catalyzed redox reaction between cerium(IV) and thallium(I) is thought to proceed…
A:
Q: Determine the activation energy for the redox reaction Q2* + 2 Rat→ Q** + 2 R2*. Rate Constant…
A: The rate constant of a reaction depends on the temperature of the reaction. The minimum amount of…
Q: Calculate the AS°rxn of the following reaction at 223°C and standard pressure. 1st attempt O See…
A:
Q: 9) The temperature dependence of the rate constant for a re- action is tabulated as follows:…
A: The Arrhenius equation is the relationship between the temperature and rate constants and also…
Q: 800 705 kJ 600-- ---- 497 kJ PE (kJ) 400 200 175 kJ 100 Reaction Progress Created by E. Lee for…
A:
Q: er 2 800 705 kJ 600 497 kJ PE (kJ) 400 200 175 kJ 100 Reaction Progress Ceated by E Lee for Virtual…
A:
Q: 6. To observe the effect of temperature on reaction rate, the reaction between MnO4 aq) and C204(ag)…
A: Given: T (oC) T (K) 1/T (1/K) k (M.S-1) ln(k) 20 293 3.413*10-3 5.03*10-5 -9.897 35 308…
Q: Calculate AT From the data, calculate q, the number of Joules absorbed by the acid-base…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The reaction: H2 + I2 = 2HI has an Eactivation = 2000 J/mol. Suppose that it has K=7.5x10-5 M-1s-1 at T=700K, find K at T=800K.Why proposed mechanisms is not reasonable. Chose the correct option and explain.Catalyst is described as any substance that increases the rate of a reaction without itself being consumed. Give a modern and recent application of a catalyst used in a chemical reaction. a ) the commercial or trade name of the catalyst b ) the properties of the catalyst (e.g., shape, size, colour, price). c ) the advantages and the disadvantages of the chosen catalyst. d )catalytic reaction (e.g., the reactant, product, reaction equation, phase, selectivity, promoter, catalyst support, operating temperature & pressure).
- Determine the activation energy from the following data. Temperature (oC) K (s-1) 189.7 2.52x10-5 251.2 3.16x10-3 Group of answer choices 7.74 kJ/mole 158 kJ/mole 0.574 kJ/mole 19.0 kJ/mole 0.653 kJ/moleThe overall reactions A --> D occurs via the following mechanism with the given reaction energies and barriers for each step Assume that a single step controls the rate of the reaction and that any steps preceding the rate-determining step can be considered quasi-equilibrium. What are the values for apparent activation barriers you would measure for the following. i) Step 1 is the rate-determining step. ii) Step 2 is the rate-determining step.(a) show a plot of Energy (vertical axis) versus Reaction Pathway for the reaction R to P which is an endothermic process (+50 kJ/mole) with an energy of activation (Eact) of 250 kJ/mole. (b) mark the location of the activated complex. (c) for the reverse reaction determine the energy of activation
- Consider the following mechanism in aqueous solutions. At 298 K the equilibrium constant for the first step is 5.75⋅10−35.75⋅10-3 k1k1 Step1: A + A −−→←−−←→ A + B k−1k-1 k2k2 Step2: B →→ P When [A] is large, the second step is slow and the first step is fast and at equilibrium. Under these conditions the following experimental data is gathered. The third column in the table is a check to make sure [A] is sufficiently large. The ratio should be greater than 100 when the absolute uncertainties are ±0.01±0.01 Note that as time goes on here, the assumption we are making will not work. Time…Use Cleland notation to describe Ordered and Random sequential reactions and a Ping Pong reaction.what are the Reasonable Mechanism
- From the data given below calculate the value of ΔS° for the reaction. T = 298.15 K. A(g) + 1B(g) ---> 1C(s) + D(l)ΔfH°/(kJ mol-1) -393.23 -44.71 -332.49 -284.45ΔfG°/(kJ mol-1) -392.07 -15.56 -197.3 -231.43Determine the activation energy from the following data. Temperature (oC) k (s-1) 288 0.0521 318 0.332b) what is the velocity of a reaction when [S] = 20 mM, where Vmax = 100 umol/mL*sec, and 4 Km = 2mM? can you show me how he got 98 umol / mL*sec, very close to Vmx, as [S]>>Km, at zero order of the kinetics. step by step please

