This portion of the boundary is at temperature Tres 600 K Reservoir at Tres W - Air initially at 300 K, 1 bar. Finally, T2 600 K
This portion of the boundary is at temperature Tres 600 K Reservoir at Tres W - Air initially at 300 K, 1 bar. Finally, T2 600 K
Elements Of Electromagnetics
7th Edition
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Sadiku, Matthew N. O.
ChapterMA: Math Assessment
Section: Chapter Questions
Problem 1.1MA
Related questions
Question
A closed, rigid tank contains 4 kg of air initially at 300 K, 1 bar. As illustrated in the figure below, the tank is in contact with a thermal reservoir at 600 K and heat transfer occurs at the boundary where the temperature is 600 K. A stirring rod transfers W = -480 kJ of energy as shown in the diagram.The final temperature is 600 K. The air can be modeled as an ideal gas with cν = 0.733 kJ/kg · K and kinetic and potential energy effects are negligible.
Determine the amount of entropy transferred into the air and the amount of entropy produced, each in kJ/K.
| Q/Tb = | kJ/K |
| σ = | kJ/K |
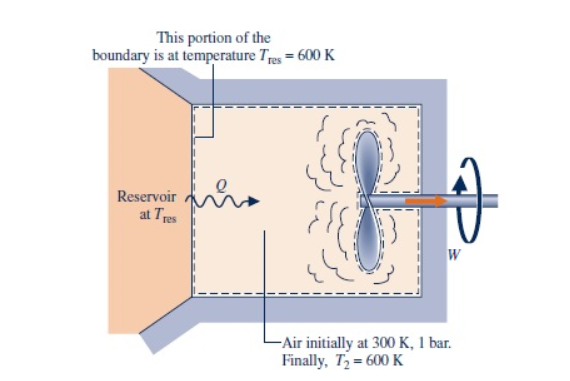
Transcribed Image Text:This portion of the
boundary is at temperature Tres 600 K
Reservoir
at Tres
W
- Air initially at 300 K, 1 bar.
Finally, T2 600 K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:
9781118170519
Author:
Norman S. Nise
Publisher:
WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:
9781337093347
Author:
Barry J. Goodno, James M. Gere
Publisher:
Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:
9781118807330
Author:
James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:
WILEY