This sketch of a neutral molecule is shaded red or blue wherever the electrostatic potential at the molecule's surface isn't zero. What could the chemical formula of the molecule be? electrostatic potential map possible chemical formula (check all that apply) So3 NO2 HCN 0 CICN HF ? X Check Explanation Privacy Terms of Use 2019 McGraw-Hill Education. All Rights Reserved. Shc Alyssa Flores-....pdf Chromatograp....pdf MacBook Pro F11 DII F10 F9 F8 F7 F6 20 O00 F 5 F 4
This sketch of a neutral molecule is shaded red or blue wherever the electrostatic potential at the molecule's surface isn't zero. What could the chemical formula of the molecule be? electrostatic potential map possible chemical formula (check all that apply) So3 NO2 HCN 0 CICN HF ? X Check Explanation Privacy Terms of Use 2019 McGraw-Hill Education. All Rights Reserved. Shc Alyssa Flores-....pdf Chromatograp....pdf MacBook Pro F11 DII F10 F9 F8 F7 F6 20 O00 F 5 F 4
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 70P: Ozone (O3) has a nonzero dipole moment. In the molecule of O3 , one of the oxygen atoms is directly...
Related questions
Question
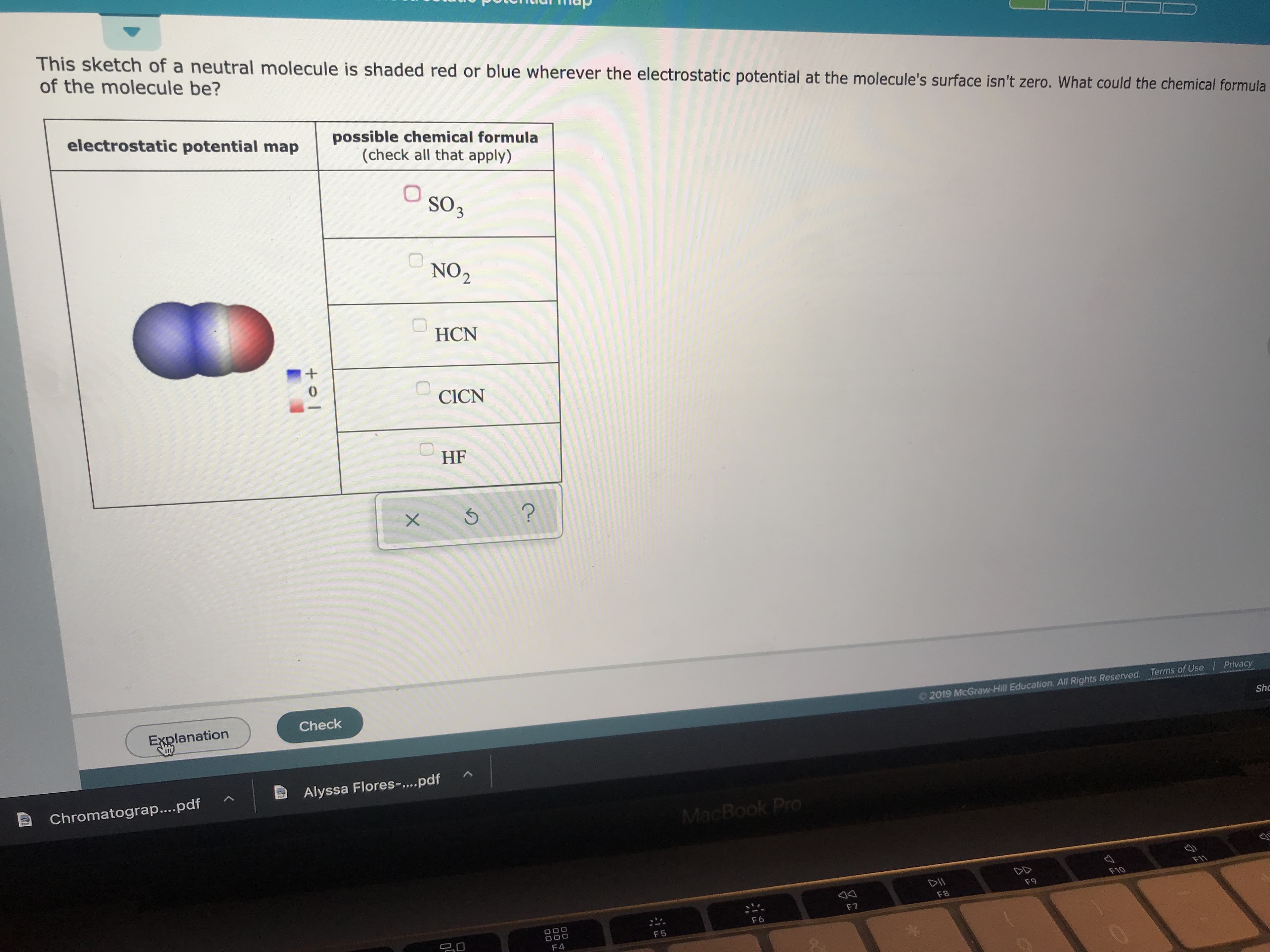
Transcribed Image Text:This sketch of a neutral molecule is shaded red or blue wherever the electrostatic potential at the molecule's surface isn't zero. What could the chemical formula
of the molecule be?
electrostatic potential map
possible chemical formula
(check all that apply)
So3
NO2
HCN
0
CICN
HF
?
X
Check
Explanation
Privacy
Terms of Use
2019 McGraw-Hill Education. All Rights Reserved.
Shc
Alyssa Flores-....pdf
Chromatograp....pdf
MacBook Pro
F11
DII
F10
F9
F8
F7
F6
20
O00
F 5
F 4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,