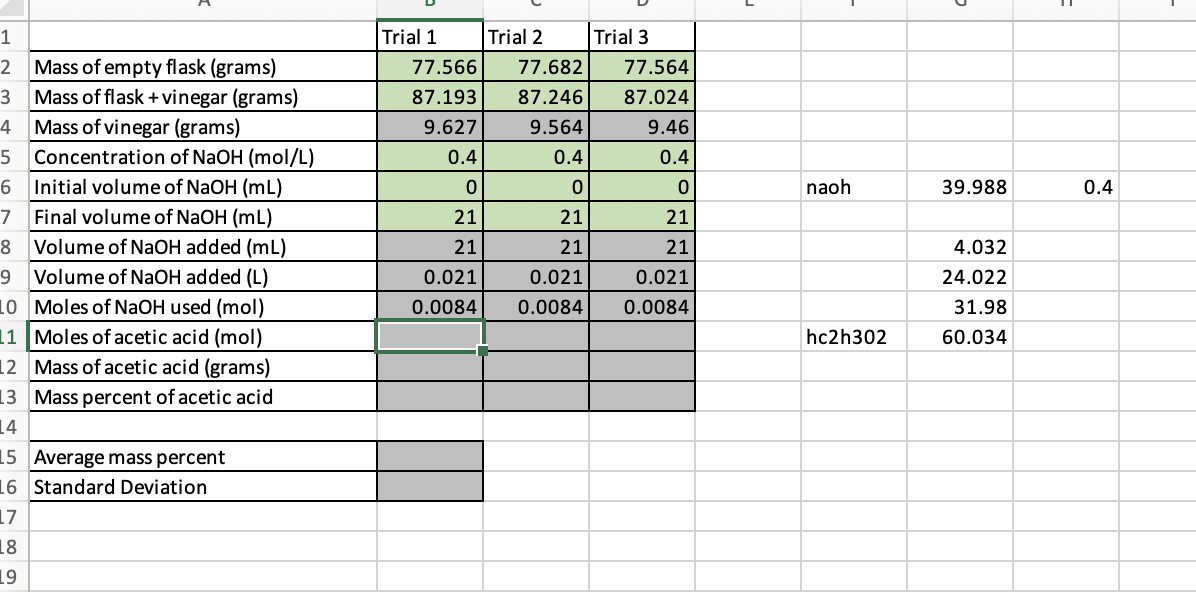
Trial 1 Trial 2 Trial 3 Mass of empty flask (grams) 77.566 77.682 77.564 Mass of flask + vinegar (grams) Mass of vinegar (grams) Concentration of NaOH (mol/L) 87.193 87.246 87.024 9.627 9.564 9.46 0.4 0.4 0.4 Initial volume of NaOH (mL) Final volume of NaOH (mL) 21 21 21 Volume of NaOH added (mL) 21 21 21 Volume of NaOH added (L) 0.021 0.021 0.021 Moles of NaOH used (mol) 0.0084 0.0084 0.0084 Moles of acetic acid (mol) Mass of acetic acid (grams) Mass percent of acetic acid Average mass percent Standard Deviation
Trial 1 Trial 2 Trial 3 Mass of empty flask (grams) 77.566 77.682 77.564 Mass of flask + vinegar (grams) Mass of vinegar (grams) Concentration of NaOH (mol/L) 87.193 87.246 87.024 9.627 9.564 9.46 0.4 0.4 0.4 Initial volume of NaOH (mL) Final volume of NaOH (mL) 21 21 21 Volume of NaOH added (mL) 21 21 21 Volume of NaOH added (L) 0.021 0.021 0.021 Moles of NaOH used (mol) 0.0084 0.0084 0.0084 Moles of acetic acid (mol) Mass of acetic acid (grams) Mass percent of acetic acid Average mass percent Standard Deviation
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 27QAP
Related questions
Question
Transcribed Image Text:1
Trial 1
Trial 2
Trial 3
Mass of empty flask (grams)
Mass of flask + vinegar (grams)
Mass of vinegar (grams)
Concentration of NaOH (mol/L)
77.566
77.682
77.564
3
87.193
87.246
87.024
9.627
9.564
9.46
5
0.4
0.4
0.4
Initial volume of NaOH (mL)
Final volume of NaOH (mL)
naoh
39.988
0.4
7
21
21
21
Volume of NaOH added (mL)
21
21
21
4.032
9.
Volume of NaOH added (L)
0.021
0.021
0.021
24.022
10 Moles of NAOH used (mol)
11 Moles of acetic acid (mol)
12 Mass of acetic acid (grams)
0.0084
0.0084
0.0084
31.98
hc2h302
60.034
13 Mass percent of acetic acid
14
15 Average mass percent
16 Standard Deviation
17
18
19
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax