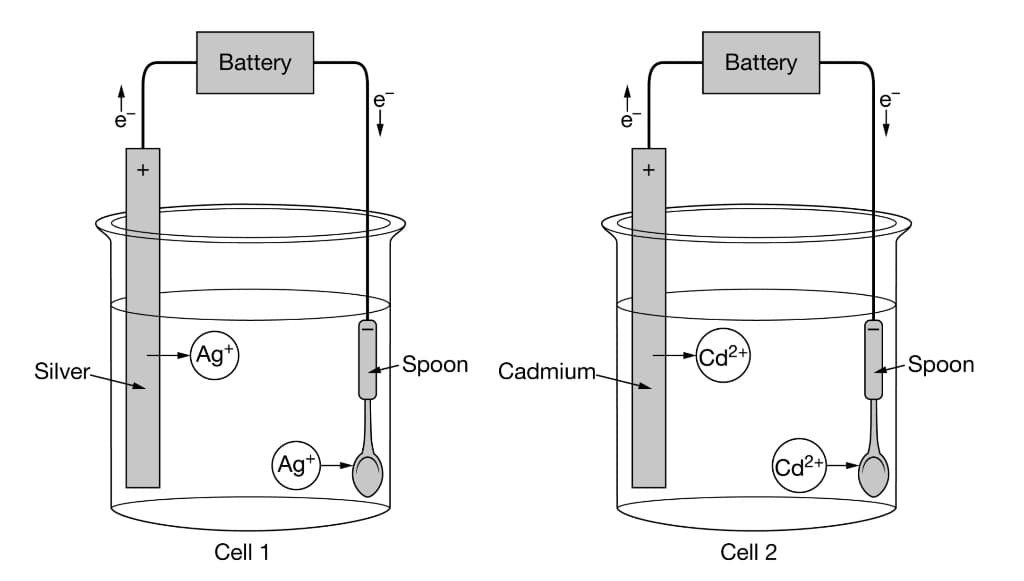
Two identical spoons were electroplated with Ag or Cd through the use of the electrolytic cells illustrated above. A current of 5.00A was supplied to each cell for 600. seconds, and the masses of the spoons before and after the electroplating were recorded. Which of the following mathematical equations can best be used to account for the much larger increase in mass of the spoon electroplated with Ag compared with the spoon electroplated with Cd? A. ΔG°= ΔH°−TΔS° B. ΔG°=−RT lnK C. E = E°−(RT/nF)lnQ D. I=q/t
Two identical spoons were electroplated with Ag or Cd through the use of the electrolytic cells illustrated above. A current of 5.00A was supplied to each cell for 600. seconds, and the masses of the spoons before and after the electroplating were recorded. Which of the following mathematical equations can best be used to account for the much larger increase in mass of the spoon electroplated with Ag compared with the spoon electroplated with Cd? A. ΔG°= ΔH°−TΔS° B. ΔG°=−RT lnK C. E = E°−(RT/nF)lnQ D. I=q/t
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 18.69QE: What is the voltage of a concentration cell of Fe2+ ions where the concentrations are 0.0025 and...
Related questions
Question
Two identical spoons were electroplated with Ag or Cd through the use of the electrolytic cells illustrated above. A current of 5.00A was supplied to each cell for 600. seconds, and the masses of the spoons before and after the electroplating were recorded. Which of the following mathematical equations can best be used to account for the much larger increase in mass of the spoon electroplated with Ag compared with the spoon electroplated with Cd?
A. ΔG°= ΔH°−TΔS°
B. ΔG°=−RT lnK
C. E = E°−(RT/nF)lnQ
D. I=q/t
Transcribed Image Text:Battery
Battery
(Ag*
Spoon
Cd2+
-Spoon
Silver-
Cadmium-
Ag*
Cd2+
Cell 1
Cell 2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning