Use standard reduction potentials to calculate the standard free energy change in kJ for the reaction: Hg?*(aq) + Co(s)- Hg(1) + Co²*(aq) Answer: kJ K for this reaction would b than one. greater less Standard Reduction (Electrode) Potentials at 25 °C Half-Cell Reaction E° (volts) F2(g) + 2 e –→ 2 F(aq) 2.87 |Ce**(aq) + e¯ –→ Ce³*(aq) 4+ 1.61 2+ Mn04 (aq) + 8 H"(aq) + 5 e –→ Mn-"(aq) + 4 H2O(1) 1.51 Cl2(g) + 2 e–→ 2 Cl(aq) 1.36 | Cr2072 (aq) + 14 H*(aq) + 6 e → 2 Cr**(aq) + 7 H20(1) 3+ 1.33 02(g) + 4 H*(aq) + 4 e¯ → 2 H2O(1) 1.229 Br2(1) + 2 e–→ 2 Br (aq) 1.08 NO3 (aq) + 4 H"(aq)+3 e¯ → NO(g) + 2 H2O(1) 0.96 2+ 2 Hg"(aq) + 2 e Hg22"(aq) 0.920 Hg-"(aq) + 2 e → Hg(1) 2+ 0.855 Ag"(aq) + e¯ – Ag(s) 0.799 Hg22"(aq) + 2 e¯→2 Hg(1) 0.789 Fe*(aq) + e – Fe2+ (aq) 0.771 I2(s) + 2 e –→ 2 1(aq) 0.535 Fe(CN)6 (aq) + e¯→ Fe(CN)6* (aq) 0.48 Cu2*(aq) + 2 e →Cu(s) 0.337 Cu2"(aq) + e¯ → Cu*(aq) 0.153 S(s) + 2 H¨(aq) + 2 e¯ → H2S(aq) 0.14 |2н (aд) + 2 е — Н2(g) 0.0000 Pb2+, (aq) + 2 e → Pb(s) -0.126 Sn2+ (aq) + 2 e –→ Sn(s) -0.14 Ni2+(ag) + 2 e →Ni(s) -0.25 Co2+ *(aq) + 2 e→Co(s) -0.28 Ca2+ (aq) + 2 e → Cd(s) -0.403 |Cr*(aq) + e¯ C2*(aq) -0.41 Fe2*(aq) + 2 e →Fe(s) -0.44 Cr*(aq) + 3 e¯ → Cr(s) -0.74 Zn2*(aq) + 2 e → Zn(s) -0.763 2 H20(1) + 2 e →H2(g) +2 OH (aq) -0.83 Mn2+(ag) + 2 e¯→ Mn(s) -1.18 AlS*(aq) + 3 e →Al(s) -1.66 Mg"(aq) + 2 e →Mg(s) -2.37 Na (aq) + e –→ Na(s) -2.714 K*(aq) + e → K(s) -2.925 Li" (aq) + e –→ Li(s) -3.045
Use standard reduction potentials to calculate the standard free energy change in kJ for the reaction: Hg?*(aq) + Co(s)- Hg(1) + Co²*(aq) Answer: kJ K for this reaction would b than one. greater less Standard Reduction (Electrode) Potentials at 25 °C Half-Cell Reaction E° (volts) F2(g) + 2 e –→ 2 F(aq) 2.87 |Ce**(aq) + e¯ –→ Ce³*(aq) 4+ 1.61 2+ Mn04 (aq) + 8 H"(aq) + 5 e –→ Mn-"(aq) + 4 H2O(1) 1.51 Cl2(g) + 2 e–→ 2 Cl(aq) 1.36 | Cr2072 (aq) + 14 H*(aq) + 6 e → 2 Cr**(aq) + 7 H20(1) 3+ 1.33 02(g) + 4 H*(aq) + 4 e¯ → 2 H2O(1) 1.229 Br2(1) + 2 e–→ 2 Br (aq) 1.08 NO3 (aq) + 4 H"(aq)+3 e¯ → NO(g) + 2 H2O(1) 0.96 2+ 2 Hg"(aq) + 2 e Hg22"(aq) 0.920 Hg-"(aq) + 2 e → Hg(1) 2+ 0.855 Ag"(aq) + e¯ – Ag(s) 0.799 Hg22"(aq) + 2 e¯→2 Hg(1) 0.789 Fe*(aq) + e – Fe2+ (aq) 0.771 I2(s) + 2 e –→ 2 1(aq) 0.535 Fe(CN)6 (aq) + e¯→ Fe(CN)6* (aq) 0.48 Cu2*(aq) + 2 e →Cu(s) 0.337 Cu2"(aq) + e¯ → Cu*(aq) 0.153 S(s) + 2 H¨(aq) + 2 e¯ → H2S(aq) 0.14 |2н (aд) + 2 е — Н2(g) 0.0000 Pb2+, (aq) + 2 e → Pb(s) -0.126 Sn2+ (aq) + 2 e –→ Sn(s) -0.14 Ni2+(ag) + 2 e →Ni(s) -0.25 Co2+ *(aq) + 2 e→Co(s) -0.28 Ca2+ (aq) + 2 e → Cd(s) -0.403 |Cr*(aq) + e¯ C2*(aq) -0.41 Fe2*(aq) + 2 e →Fe(s) -0.44 Cr*(aq) + 3 e¯ → Cr(s) -0.74 Zn2*(aq) + 2 e → Zn(s) -0.763 2 H20(1) + 2 e →H2(g) +2 OH (aq) -0.83 Mn2+(ag) + 2 e¯→ Mn(s) -1.18 AlS*(aq) + 3 e →Al(s) -1.66 Mg"(aq) + 2 e →Mg(s) -2.37 Na (aq) + e –→ Na(s) -2.714 K*(aq) + e → K(s) -2.925 Li" (aq) + e –→ Li(s) -3.045
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 150CP: Given the following two standard reduction potentials, solve for the standard reduction potential of...
Related questions
Question

Transcribed Image Text:Use standard reduction potentials to calculate the standard free energy change in kJ for the reaction:
Hg?*(aq) + Co(s)-
Hg(1) + Co²*(aq)
Answer:
kJ
K for this reaction would b
than one.
greater
less
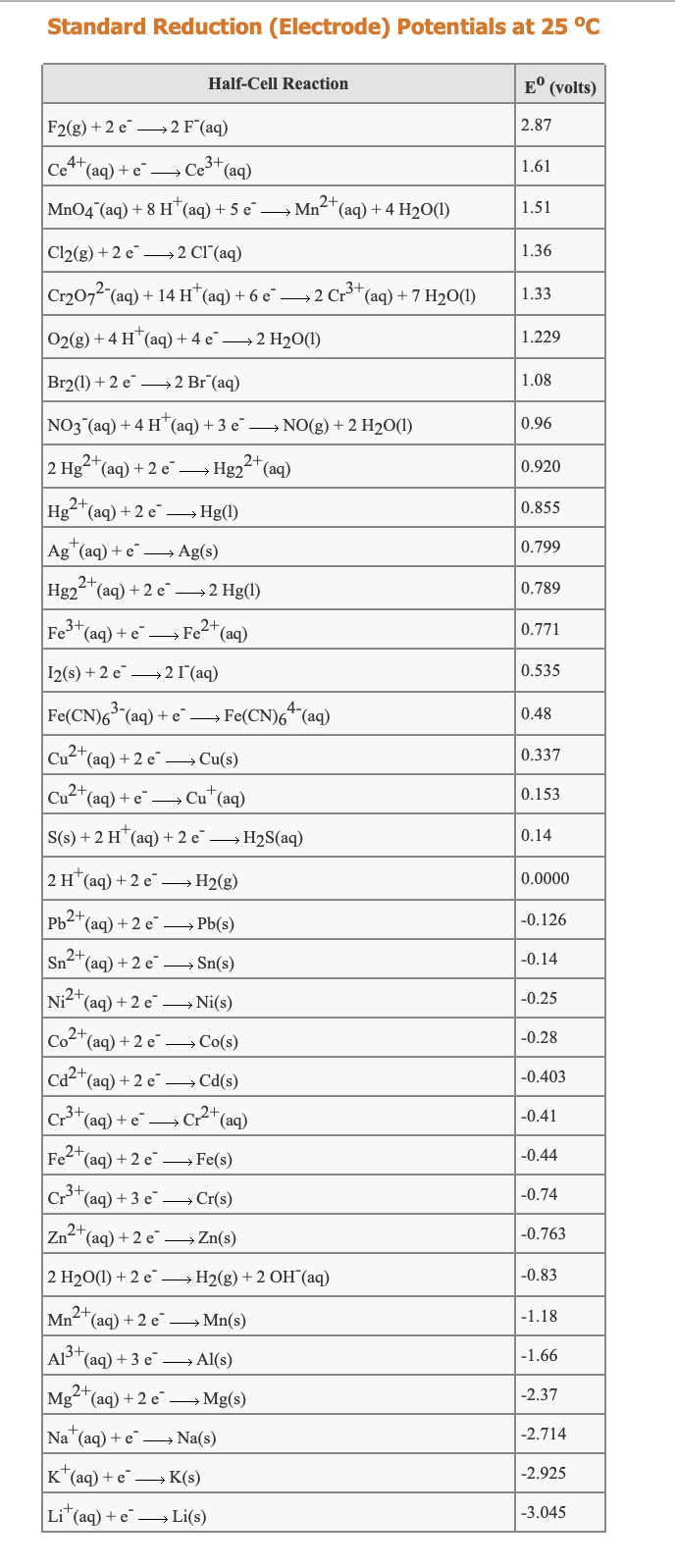
Transcribed Image Text:Standard Reduction (Electrode) Potentials at 25 °C
Half-Cell Reaction
E° (volts)
F2(g) + 2 e –→ 2 F(aq)
2.87
|Ce**(aq) + e¯ –→ Ce³*(aq)
4+
1.61
2+
Mn04 (aq) + 8 H"(aq) + 5 e –→ Mn-"(aq) + 4 H2O(1)
1.51
Cl2(g) + 2 e–→ 2 Cl(aq)
1.36
| Cr2072 (aq) + 14 H*(aq) + 6 e → 2 Cr**(aq) + 7 H20(1)
3+
1.33
02(g) + 4 H*(aq) + 4 e¯ → 2 H2O(1)
1.229
Br2(1) + 2 e–→ 2 Br (aq)
1.08
NO3 (aq) + 4 H"(aq)+3 e¯ → NO(g) + 2 H2O(1)
0.96
2+
2 Hg"(aq) + 2 e
Hg22"(aq)
0.920
Hg-"(aq) + 2 e → Hg(1)
2+
0.855
Ag"(aq) + e¯ – Ag(s)
0.799
Hg22"(aq) + 2 e¯→2 Hg(1)
0.789
Fe*(aq) + e –
Fe2+ (aq)
0.771
I2(s) + 2 e –→ 2 1(aq)
0.535
Fe(CN)6 (aq) + e¯→ Fe(CN)6* (aq)
0.48
Cu2*(aq) + 2 e →Cu(s)
0.337
Cu2"(aq) + e¯ → Cu*(aq)
0.153
S(s) + 2 H¨(aq) + 2 e¯ → H2S(aq)
0.14
|2н (aд) + 2 е — Н2(g)
0.0000
Pb2+,
(aq) + 2 e → Pb(s)
-0.126
Sn2+
(aq) + 2 e –→ Sn(s)
-0.14
Ni2+(ag) + 2 e →Ni(s)
-0.25
Co2+
*(aq) + 2 e→Co(s)
-0.28
Ca2+
(aq) + 2 e → Cd(s)
-0.403
|Cr*(aq) + e¯
C2*(aq)
-0.41
Fe2*(aq) + 2 e →Fe(s)
-0.44
Cr*(aq) + 3 e¯ → Cr(s)
-0.74
Zn2*(aq) + 2 e → Zn(s)
-0.763
2 H20(1) + 2 e →H2(g) +2 OH (aq)
-0.83
Mn2+(ag) + 2 e¯→ Mn(s)
-1.18
AlS*(aq) + 3 e →Al(s)
-1.66
Mg"(aq) + 2 e →Mg(s)
-2.37
Na (aq) + e –→ Na(s)
-2.714
K*(aq) + e → K(s)
-2.925
Li" (aq) + e –→ Li(s)
-3.045
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning