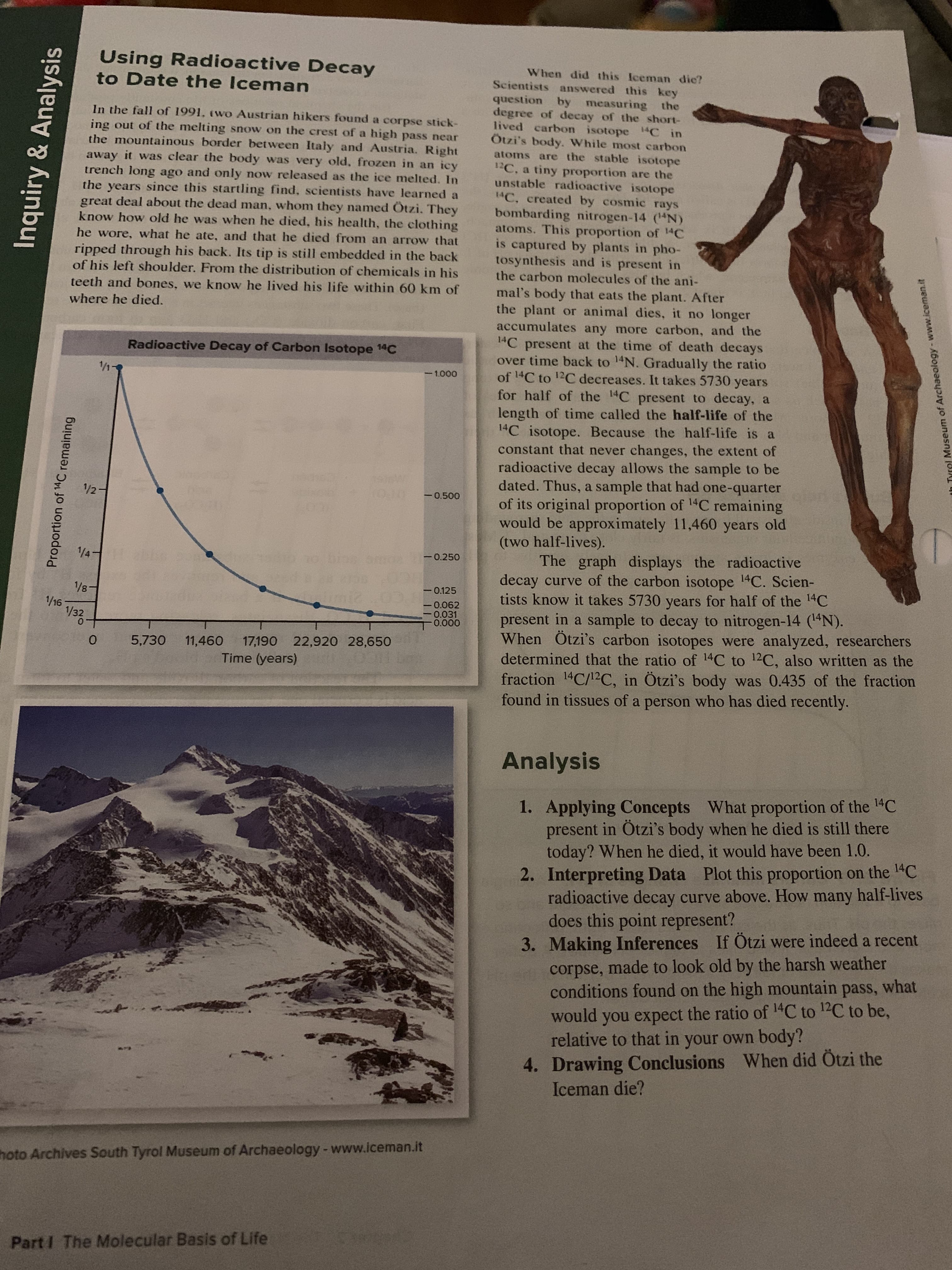
Using Radioactive Decay to Date the Iceman When did this Iceman dic? Scientists answered this key question by measuring the degree of decay of the short- lived carbon isotope 1C in Ötzi's body. While most carbon atoms are the stable isotope 12C. unstable radioactive isotope 14C. created by cosmic rays bombarding nitrogen-14 (4N) atoms. This proportion of 14C is captured by plants in pho- tosynthesis and is present in In the fall of 1991. (wo Austrian hikers found a corpse stick- ing out of the melting snow on the crest of a high pass near the mountainous border between Italy and Austria. Right away it was clear the body was very old, frozen in an icy now released as the ice melted. In tiny proportion are the a trench long ago and only the years since this startling find, scientists have learned a great deal about the dead man, whom they named Ötzi. They know how old he was when he died, his health, the clothing he wore, what he ate, and that he died from an arrow that ripped through his back. Its tip is still embedded in the back of his left shoulder. From the distribution of chemicals in his the carbon molecules of the ani- teeth and bones, we know he lived his life within 60 km of mal's body that eats the plant. After the plant or animal dies, it no longer accumulates any more carbon, and the 1C present at the time of death decays over time back to 14N. Gradually the ratio of 14C to 12C decreases. It takes 5730 years for half of the 14C present to decay, a length of time called the half-life of the 1C isotope. Because the half-life is a constant that never changes, the extent of radioactive decay allows the sample to be dated. Thus, a sample that had one-quarter of its original proportion of 14C remaining would be approximately 11,460 years old (two half-lives). The graph displays the radioactive decay curve of the carbon isotope 14C. Scien- tists know it takes 5730 years for half of the 14C present in a sample to decay to nitrogen-14 (14N) When Ötzi's carbon isotopes were analyzed, researchers determined that the ratio of 14C to 12C, also written as the fraction 14C/12C, in Ötzi's body was 0.435 of the fraction found in tissues of a person who has died recently. where he died. Radioactive Decay of Carbon Isotope 14C 1- -1.000 1/21 -0.500 1/4 10 OruC 0.250 0.125 1/8-1 1/16 1/32 0.062 0.031 0.000 17,190 22,920 28,650 11,460 5,730 Time (years) Analysis of the 14C Applying Concepts What proportion present in Otzi's body when he died is still there today? When he died, it would have been 1.0. 2. Interpreting Data Plot this proportion on the 4C radioactive decay curve above. How many half-lives does this point represent? 3. Making Inferences If Otzi were indeed a recent corpse, made to look old by the harsh weather conditions found on the high mountain pass, what would you expect the ratio of 1C to 12C to be, relative to that in your own 1. body? 4. Drawing Conclusions When did Ötzi the Iceman die? hoto Archives South Tyrol Museum of Archaeology -www.iceman.it Part I The Molecular Basis of Life Inquiry & Analysis Proportion of 14C remaining th Tyrel Museum of Archaeology-www.iceman.it
Using Radioactive Decay to Date the Iceman When did this Iceman dic? Scientists answered this key question by measuring the degree of decay of the short- lived carbon isotope 1C in Ötzi's body. While most carbon atoms are the stable isotope 12C. unstable radioactive isotope 14C. created by cosmic rays bombarding nitrogen-14 (4N) atoms. This proportion of 14C is captured by plants in pho- tosynthesis and is present in In the fall of 1991. (wo Austrian hikers found a corpse stick- ing out of the melting snow on the crest of a high pass near the mountainous border between Italy and Austria. Right away it was clear the body was very old, frozen in an icy now released as the ice melted. In tiny proportion are the a trench long ago and only the years since this startling find, scientists have learned a great deal about the dead man, whom they named Ötzi. They know how old he was when he died, his health, the clothing he wore, what he ate, and that he died from an arrow that ripped through his back. Its tip is still embedded in the back of his left shoulder. From the distribution of chemicals in his the carbon molecules of the ani- teeth and bones, we know he lived his life within 60 km of mal's body that eats the plant. After the plant or animal dies, it no longer accumulates any more carbon, and the 1C present at the time of death decays over time back to 14N. Gradually the ratio of 14C to 12C decreases. It takes 5730 years for half of the 14C present to decay, a length of time called the half-life of the 1C isotope. Because the half-life is a constant that never changes, the extent of radioactive decay allows the sample to be dated. Thus, a sample that had one-quarter of its original proportion of 14C remaining would be approximately 11,460 years old (two half-lives). The graph displays the radioactive decay curve of the carbon isotope 14C. Scien- tists know it takes 5730 years for half of the 14C present in a sample to decay to nitrogen-14 (14N) When Ötzi's carbon isotopes were analyzed, researchers determined that the ratio of 14C to 12C, also written as the fraction 14C/12C, in Ötzi's body was 0.435 of the fraction found in tissues of a person who has died recently. where he died. Radioactive Decay of Carbon Isotope 14C 1- -1.000 1/21 -0.500 1/4 10 OruC 0.250 0.125 1/8-1 1/16 1/32 0.062 0.031 0.000 17,190 22,920 28,650 11,460 5,730 Time (years) Analysis of the 14C Applying Concepts What proportion present in Otzi's body when he died is still there today? When he died, it would have been 1.0. 2. Interpreting Data Plot this proportion on the 4C radioactive decay curve above. How many half-lives does this point represent? 3. Making Inferences If Otzi were indeed a recent corpse, made to look old by the harsh weather conditions found on the high mountain pass, what would you expect the ratio of 1C to 12C to be, relative to that in your own 1. body? 4. Drawing Conclusions When did Ötzi the Iceman die? hoto Archives South Tyrol Museum of Archaeology -www.iceman.it Part I The Molecular Basis of Life Inquiry & Analysis Proportion of 14C remaining th Tyrel Museum of Archaeology-www.iceman.it
Biology: The Unity and Diversity of Life (MindTap Course List)
14th Edition
ISBN:9781305073951
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Chapter19: Life's Origin And Early Evolution
Section: Chapter Questions
Problem 3DAA
Related questions
Question
#2
Transcribed Image Text:Using Radioactive Decay
to Date the Iceman
When did this Iceman dic?
Scientists answered this key
question by measuring the
degree of decay of the short-
lived carbon isotope 1C in
Ötzi's body. While most carbon
atoms are the stable isotope
12C.
unstable radioactive isotope
14C. created by cosmic rays
bombarding nitrogen-14 (4N)
atoms. This proportion of 14C
is captured by plants in pho-
tosynthesis and is present in
In the fall of 1991. (wo Austrian hikers found a corpse stick-
ing out of the melting snow on the crest of a
high pass near
the mountainous border between Italy and Austria. Right
away it was clear the body was very old, frozen in an
icy
now released as the ice melted. In
tiny proportion are the
a
trench long ago and only
the years since this startling find, scientists have learned a
great deal about the dead man, whom they named Ötzi. They
know how old he was when he died, his health, the clothing
he wore, what he ate, and that he died from an arrow that
ripped through his back. Its tip is still embedded in the back
of his left shoulder. From the distribution of chemicals in his
the carbon molecules of the ani-
teeth and bones, we know he lived his life within 60 km of
mal's body that eats the plant. After
the plant or animal dies, it no longer
accumulates any more carbon, and the
1C present at the time of death decays
over time back to 14N. Gradually the ratio
of 14C to 12C decreases. It takes 5730 years
for half of the 14C present to decay, a
length of time called the half-life of the
1C isotope. Because the half-life is a
constant that never changes, the extent of
radioactive decay allows the sample to be
dated. Thus, a sample that had one-quarter
of its original proportion of 14C remaining
would be approximately 11,460 years old
(two half-lives).
The graph displays the radioactive
decay curve of the carbon isotope 14C. Scien-
tists know it takes 5730 years for half of the 14C
present in a sample to decay to nitrogen-14 (14N)
When Ötzi's carbon isotopes were analyzed, researchers
determined that the ratio of 14C to 12C, also written as the
fraction 14C/12C, in Ötzi's body was 0.435 of the fraction
found in tissues of a person who has died recently.
where he died.
Radioactive Decay of Carbon Isotope 14C
1-
-1.000
1/21
-0.500
1/4
10
OruC 0.250
0.125
1/8-1
1/16
1/32
0.062
0.031
0.000
17,190 22,920 28,650
11,460
5,730
Time (years)
Analysis
of the 14C
Applying Concepts What proportion
present in Otzi's body when he died is still there
today? When he died, it would have been 1.0.
2. Interpreting Data Plot this proportion on the 4C
radioactive decay curve above. How many half-lives
does this point represent?
3. Making Inferences If Otzi were indeed a recent
corpse, made to look old by the harsh weather
conditions found on the high mountain pass, what
would you expect the ratio of 1C to 12C to be,
relative to that in your own
1.
body?
4. Drawing Conclusions When did Ötzi the
Iceman die?
hoto Archives South Tyrol Museum of Archaeology -www.iceman.it
Part I The Molecular Basis of Life
Inquiry & Analysis
Proportion of 14C remaining
th Tyrel Museum of Archaeology-www.iceman.it
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning


Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning