V = a (2r)3 %3D 8(1.69 Å)³ %3D - Part B face-centered cubic Express the volume in cubic angstroms to three significant digits. ? V = Submit Previous Answers Request Answer
V = a (2r)3 %3D 8(1.69 Å)³ %3D - Part B face-centered cubic Express the volume in cubic angstroms to three significant digits. ? V = Submit Previous Answers Request Answer
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter9: Liquids, Solids, And Materials
Section: Chapter Questions
Problem 74QRT
Related questions
Question
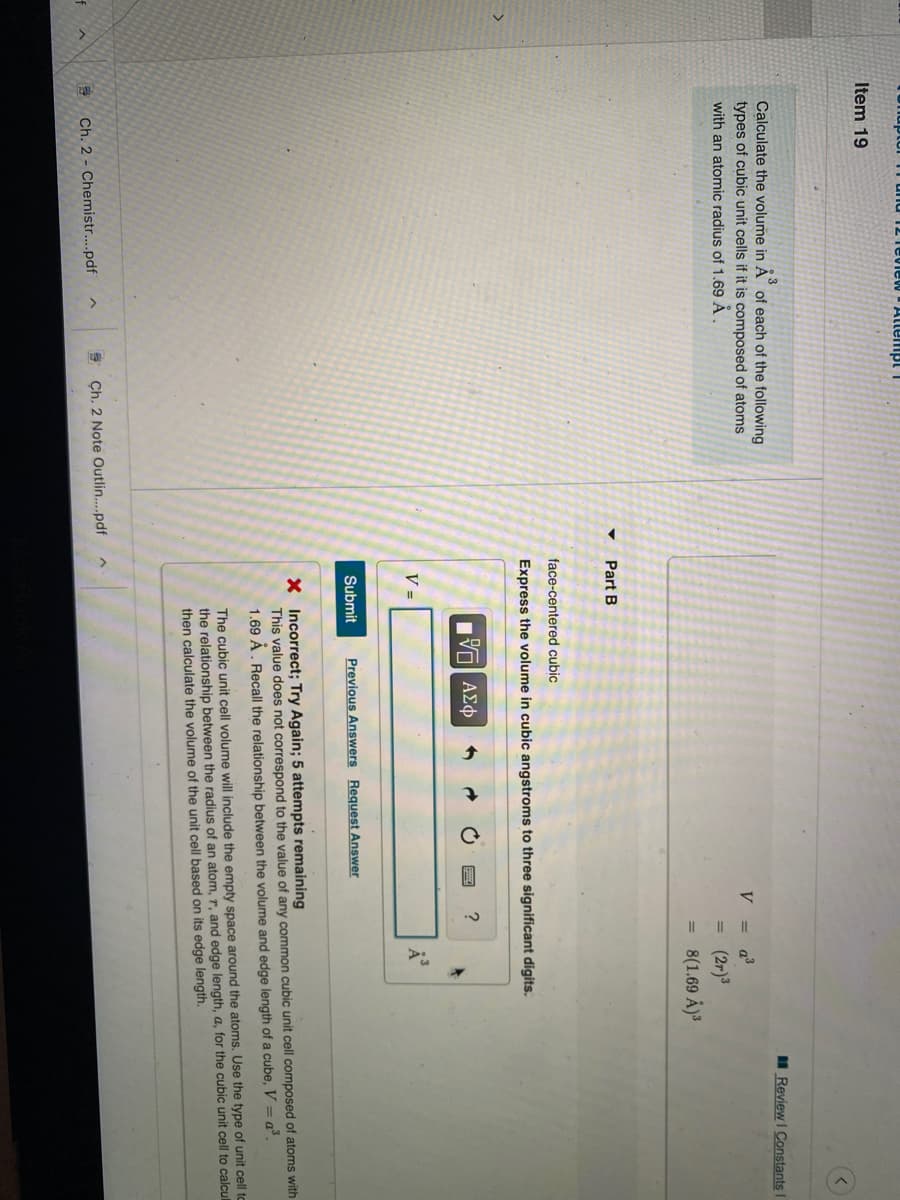
Transcribed Image Text:Item 19
II Review I Constants
Calculate the volume in A of each of the following
types of cubic unit cells if it is composed of atoms
V = a³
with an atomic radius of 1.69 Ả .
(2r)3
= 8(1.69 Å)³
Part B
face-centered cubic
Express the volume in cubic angstroms to three significant digits.
V =
Submit
Previous Answers Request Answer
X Incorrect; Try Again; 5 attempts remaining
This value does not correspond to the value of any common cubic unit cell composed of atoms with
1.69 Á. Recall the relationship between the volume and edge length of a cube, V = a°.
The cubic unit cell volume will include the empty space around the atoms. Use the type of unit cell to
the relationship between the radius of an atom, r, and edge length, a, for the cubic unit cell to calcul
then calculate the volume of the unit cell based on its edge length.
S Çh. 2 Note Outlin..pdf
Ch. 2 - Chemistr..pdf
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning