Water potential is the tendency of water to move from one place to another. Pumpkin cores were placed in sucrose solutions at 22°C and normal atmospheric pressure. The following data were gathered. Complete the above table. Graph the percentage change in mass. Label both the x and y axes. Using the graph, determine the molar concentration of the pumpkin cores and explain how it is related to pressure potential.
Water potential is the tendency of water to move from one place to another. Pumpkin cores were placed in sucrose solutions at 22°C and normal atmospheric pressure. The following data were gathered. Complete the above table. Graph the percentage change in mass. Label both the x and y axes. Using the graph, determine the molar concentration of the pumpkin cores and explain how it is related to pressure potential.
Chapter9: Energy For Today
Section: Chapter Questions
Problem 9E
Related questions
Question
100%
- Water potential is the tendency of water to move from one place to another. Pumpkin cores were placed in sucrose solutions at 22°C and normal atmospheric pressure. The following data were gathered.
- Complete the above table.
- Graph the percentage change in mass. Label both the x and y axes.
- Using the graph, determine the molar concentration of the pumpkin cores and explain how it is related to pressure potential.
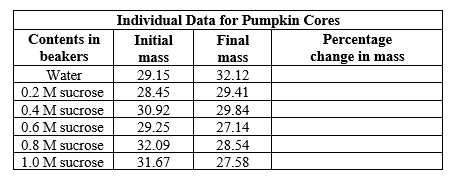
Transcribed Image Text:Individual Data for Pumpkin Cores
Contents in
Initial
Percentage
change in mass
Final
beakers
mass
mass
Water
29.15
32.12
0.2 M sucrose
28.45
29.41
0.4 M sucrose
30.92
29.84
0.6 M sucrose
29.25
27.14
0.8 M sucrose
32.09
28.54
1.0 M sucrose
31.67
27.58
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning