Q: Consider the following molecules: A - BrF3 B - SCl2 C - CF4 D - SF6 E - BF3 Which one of…
A: The bonds are said to be polar if there is some electronegativity difference between the two atoms.
Q: 1. Acetone, C3H60(CH3COCH3) Lewis structure using dots to represent bonding electrons: Lewis…
A: To answer following questions , we would first draw lewis dot structure of acetone . Lewis dot…
Q: Molecular formula : CF4 Valence electrons: Bonds: Lone Pairs: Electron geometry: Hybridization…
A: Valence electrons can be calculated by adding bond pairs and lone pairs of a molecule. Electron…
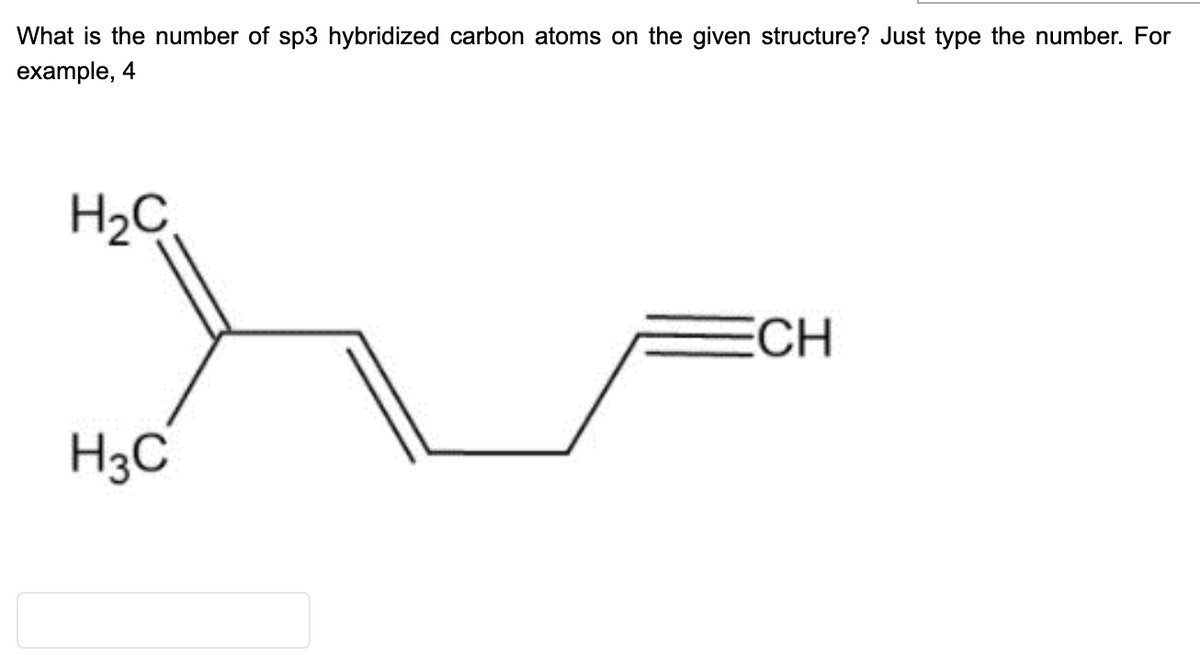
Q: Identify hybridization in each Carbon atom of given molecules.
A:
Q: From the following structure locate these examples of types of atoms or bonds: 1. a highly polarized…
A: In given structure we have to locate the following 1. a highly polarized single bond2. A highly…
Q: an sp³ hybridized carbon atom an sp² hybridized carbon atom the longest bond in the molecule E B the…
A:
Q: 30 - The hybridization of carbon atom in alkyne is. O A-30+1 π. OB - 40 O C-10+2π.
A:
Q: A single bond between two carbons with different hybridizations has a small dipole. What is the…
A:
Q: Draw the p orbitals on each sp² hybridized atom in the structures below Circle the atoms in each…
A: Topic-- Orbital diagram for the conjugated polyene and enone system
Q: The number of sp2 hybridized carbon atom * in Ascorbic acid is: HO HO () 2 О 1 О з 4 ОН ОН О
A: sp2 hybridized carbon must be attached with a double bond.
Q: Use the ball-and-stick model of vitamin Bg to answer each question. vitamin Be a. Draw a skeletal…
A: a).
Q: 1. a) Fill in the blanks regarding the carbon atoms that make up lovastatin, a drug used to treat…
A: Natural products chemistry
Q: Compound(central atom has more than more 4 electron pairs: BrF3 Valence electrons: Bonds: Lone…
A:
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. How many…
A: Citric acid is a weak organic acid. It has a sour taste. It is found abundantly in citric fruits…
Q: Oxygen atom: Total number of electron groups: Number of bonding groups/pairs: Number of lone pairs:…
A:
Q: Compound(central atom has more than more 4 electron pairs: XeF2 Valence electrons: Bonds: Lone…
A: Given : Compound XeF2 To find: Valence electrons,Bonds,Lone pairs,Electron geometry,Molecular…
Q: What are the hybridizations of the Carbons indicated by the arrows?
A:
Q: Draw a compound that contains only carbon atoms and hydrogen atoms that has 2sp3 and 2sp hybridised…
A:
Q: Compound(central atom has more than more 4 electron pairs: AsF3 Valence electrons: Bonds: Lone…
A: As is member of nitrogen family. Hence As have 5 valence electrons
Q: Which of the arrows points to an atom that is 30 (tertiary) and sp³ hybridized? с d a mamana www
A:
Q: State the orbital hybridization of each highlighted atom
A: It is given that a formic acid molecule is present and the hybridization of the highlighted carbon…
Q: Capsaicin is an active component of chili peppers, which are plan belonging to the genus Capsicum.…
A: The geometry around an atom in the molecule depends upon its hybridisation. For example, for sp3…
Q: Complete the following table: number of e = hybridization of = to make covalent bonds, atom uses:…
A: According to Valence Bond Theory, atomic orbitals are combined to form hybridized orbitals. By…
Q: 2. For each of the following make a labeled orbital diagram showing how different orbitals overlap…
A:
Q: 8. Draw a VBT (balloon-animal) representation of propene, C3H6. Draw only the large lobe of each…
A: In propene, CH2=CH-CH3, the first two C atoms from the left side are sp2 hybridized with trigonal…
Q: How many sp2 hybridized carbon atom exist in the following structure? HO. NH2 ZI
A: The sp2 hybridized carbon atoms are explained as those which have unsaturation and which contain a…
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. a. What…
A: Hey, since there are multiple subparts posted, we will answer first three subparts. If you want any…
Q: What kind of hybridization do you expect for each carbon atom in the molecule? a.1-Buten-3-yne
A:
Q: Compound(central atom has more than more 4 electron pairs: PF3 Valence electrons: Bonds: Lone…
A: For PF3 compound answer the following--
Q: Which of the following statements is TRUE about the structure benzene, C6H6? O All C atoms are sp²…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: which carbon is more electronegative : a sp3d hybridized carbon b sp2 hybridized carbon c sp…
A: The electronegativity depends upon the percentage of s-character. The more the percentage of…
Q: Но 4. Identify the functional groups present and hybridization of the labeled atoms. e но H a b H- H…
A: Functional group are collection of atoms which has their specific physical and chemical properties.…
Q: What is the hybridization at each (sp, sp2, sp3) also find molecular geometry (bent, trigonal…
A:
Q: sp' hybridized CH3 sp hybridized ECH, sp² hybridized → CH2 ČH, sp' hybridized ČH3
A: sp hybridization is observed when one s and one p orbital in the same main shell of an atom mix to…
Q: Predict the (1)bond angles around each carbon atom; (2) give the hybridization of each carbon atom;…
A: A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule.…
Q: Type of hybridization for between C=C_double bond in Alkynes is---- .A sp2 .B sp3 .C sp .D sp3d
A: Hybridization - It is the combination of atomic orbitals to form molecular orbitals which are lower…
Q: Based on the hybridization theory, which statement is INCORRECT about compound Z? H H H O Only one…
A: Since you are posted with multiple questions. As per the rule, I am answering the first question…
Q: 7 What valence orbitals (hybridized and/or nonhybridized) are available on an sphybridized carbon…
A: step 1) Introduction : Carbon has to form 2 covalent bonds which results in CH2, it is having 2…
Q: 8. Which of the following is nottrue of an sp hybridized carbon atom? a) One p orbital is present.…
A: Hybridization is the mixing of atomic orbitals having different energies to form hybrid orbitals of…
Q: Write a bond-line structure for the following compounds. Present correct geometry for sp3, sp2, and…
A: Given : Compound is given. To find : Bond line structure of the Compound with proper hybridization…
Q: Which of the following contains sp-hybridized carbon? O A. C4H8 О в. сзн6 C. C5H8 D. C6H12
A:
Q: 4. Describe the hybridization of each carbon atom in the following structure.
A: Given compound is : Describe the hybridization of each carbon atom in the following structure ?…
Q: Compound(central atom has more than more 4 electron pairs: PF5 Valence electrons: Bonds: Lone…
A:
Q: CH3+ and CH3– are two highly reactive carbon species. What is the predicted hybridization and…
A:
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. [3] [1].…
A:
Q: Circle the sp hybridized carbons that are present in 2,4-hexadiene. CH3–CH=CH–CH=CH–CH3
A: Given : molecule 2,4-hexadiene. CH3-CH=CH-CH=CH-CH3
Q: Use the ball-and-stick model to answer each question about carbidopa, a drug used in combination…
A: In ball and socket model, - White ball – Hydrogen Black ball – Carbon Read bass - Oxygen Blue ball –…
Q: Convert the given structure of caffeine into skeletal structure and identify the hybridization of…
A: Skeletal formula - this representation is also known as bond-line structure in which only bond lines…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Identify the number of sp3-hybridized carbon atoms in each of these isomers. 1. (CH3)2 CO 2. CH3CH2CHOwhich carbon is more electronegative : a sp3d hybridized carbon b sp2 hybridized carbon c sp hybridized carbonHow many unhybridized orbitals can you find in an sp2 carbon? How many hybridized orbitals can you find in an sp2 carbon?
- Which compound contains an sp2-hybridized carbon atom?In a tabular format, enumerate 5 molecules where the following atoms are found in: O (sp3-hybridized) Oxygen (sp2-hybridized) Nitrogen (sp2-hybridized) Carbon (sp2-hybridized)From the following structure locate these examples of types of atoms or bonds:1. a highly polarized single bond2. A highly polarized covalent double bond3. A nonpolar covalent bond4. A carbon atom with sp hybridization5. A carbon atom with sp2 hybridization6. A carbon atom with sp3 hybridization.7. A bond between atoms of different hybridization. Hello, I hope you can help me :)
- What is the hybridization of carbon in a singlet paired methylene and a triple paired methylene?A single bond between two carbons with different hybridizations has a small dipole. What is the direction of the dipole in the indicated bonds?a. 3 CHCH CH2 b. 3 CCH CHRamelteon is a hypnotic agent used in the treatment of insomnia. Anwser the following questions: 1. How many sp3-hybridized carbon atoms are present in this structure? 2. How many sp2-hybridized carbon atoms are present in this structure? 3. How many sp-hybridized carbon atoms are present in this structure? 4. How many lone pairs are present in this structure?

