Q: Antifreeze is composed mainly of ethylene glycol (C2H6O2, d = 1.11 g/cm3). It is added to a car…
A: The relationship between depression in freezing point (∆Tf) and molality of solition is ∆f = kf ×…
Q: The diagram below shows some isotherms of water. (a) Estimate values for the critical constants and…
A: To find out the value of the critical constant for water from the given isotherm. The three critical…
Q: At a certain temperature the following reactions have the constants shown: S(s) + O₂(g) = SO₂(g) Kc…
A:
Q: I: H₂S(aq) + Cl(aq) HCl(aq) + HS (aq) II: NH4 (aq) + HCO3(aq) H₂CO3(aq) + NH3(aq) A) Only I B) Only…
A: Given reactions are : I : H2S (aq) + Cl- (aq) <--------> HCl (aq) + HS- (aq) II. NH4+ (aq) +…
Q: H₂ + N₂ → NH3 CaCl2 + AgNO3 → Ca (NO3)2 + AgCl SnO2 + H2 → H₂O + Sn
A: 1.Balancing chemical equation means number of atom of each elements and charge involves in reactant…
Q: Part 4. Molecular View. Draw 3 molecules of water (liquid) interacting with 3 molecules of acetone…
A: Intermolecular forces are forces of attraction or repulsion that also occur between molecules of a…
Q: Fill compare a few different developing solvents and examine the effect of the polarity of the…
A: #Q.7: Since hexane is a symmetric hydrocarbon, it is nonpolar. Ethylacetate is polar due to the…
Q: Modify the given copy of the starting material to draw the major product(s). Use the single bond…
A:
Q: hat is the density of CO2 at 118.1 degF and 523.1 mmHg in grams per liter?
A: Density of a substance is calculated by dividing the given mass with volume Given temperature (T)…
Q: ) a) Using Fig. 1.9 calculate the compressibility factor Z for methane at 220 K and 46 bar. B/cm²…
A: Compressibility factor : It is the deviation in the behaviour of gas from its ideal behaviour.
Q: The molar composition of a gas is 10% H2, 10% O2, 30% CO2 and balance H2O. If 50% H20 condenses, the…
A: Solution is made up of two components salute and solvent the component which is present in major…
Q: A 13.7 kg sample of this new "compound" is separated into its component "elements" with the…
A: In this question, we have to find How many pennies are present in the 13.7 kg sample? How many…
Q: Choose the set of pressure intensities that are equivalent a. 4.33 psi, 10 ft. of water, 8.83 inches…
A: We use the below conversion factors to convert among psi, ft. water and inches of Hg. 1 psi = 2.307…
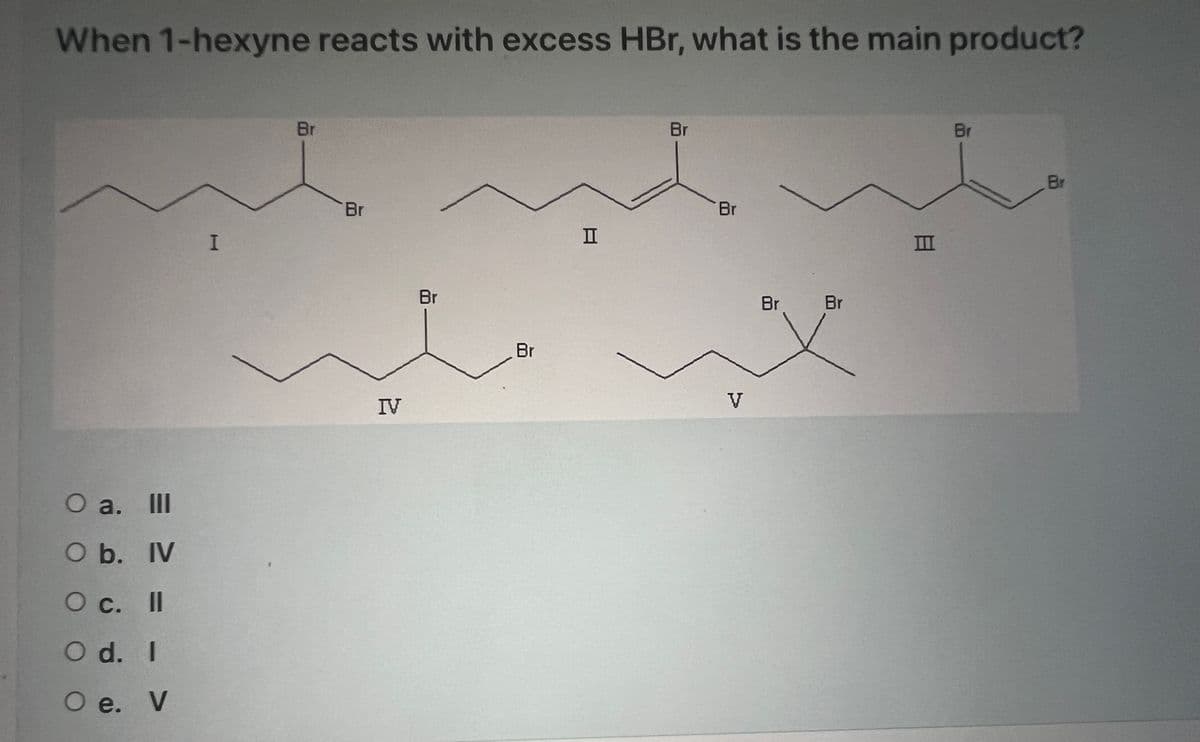
Q: Which alkyl bromide would be most reactive in an Sil reaction? Select one: Br Br Br Br
A:
Q: ΙΠΠ If two atoms of two different elements have the same number of valance electrons, 22 what does…
A: The electrons which are those present in outermost orbital of an atom are called as valence…
Q: Consider the following reaction, which takes place in a single elementary step: 2A + B A₂B k₁ k_1 If…
A: we have to calculate the value of kf for the reaction
Q: In a 0.080 M NH3 solution, what percent of the NH is present as NH?
A:
Q: <Homework #4 Exercise 8.66 MISSED THIS? Watch KCV 8.5, IWE 8.5; Read Section 8.6. You can click on…
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [N₂] [H₂]…
A:
Q: Use the periodic table to identify the elements with the following electron configurations.…
A:
Q: At 298 K. AH;131.28 kJ-mol for the reaction with Op.-8.53, 33.58, 29.12, and 28.82 J-K-mol for…
A: Given data :
Q: The index of hydrogen deficiency of the compound C12H15NO is: Select one: a. 5 b. 6 c. 8 d. 7 e.…
A: we have to calculate the hydrogen deficiency index for the given molecular formula
Q: Come up with a dilution scheme starting from 1000 μg/mL Mn stock solution so that the concentration…
A: Dilution scheme starting from 1000 μg/mL Mn stock solution so that the concentration of permanganate…
Q: For the synthesis of ammonia N₂(g) + 3H₂(g) = 2NH3(g) the equilibrium constant K. at 375°C is 1.2.…
A: Answer: In this question we have to decide whether given reaction will move in forward direction or…
Q: What is the most probable identity of the element X wb symbol is shown below?
A:
Q: Where would the r and d be mark on bcc?
A: In this question, we have to represent d and r in the bcc diagram in the preceding question.
Q: Use the periodic table to identify the elements with the following electron configurations. [Ne]3s1…
A: [Ne] 3s1We have to identify the element using periodic table and spell out the full name of the…
Q: Identify the following species as either a Lewis acid or a Lewis base, and briefly explain: HgSO4…
A: It is necessary to firstly know the Lewis acid base concept. Lewis acid - The species which is able…
Q: CH3 CH₂CI NaOH SN2
A: Organic reactions are those in which organic reactant react from organic product. SN2 reaction is…
Q: -. The mechanism for the reaction of nitrogen dioxide with carbon monoxide form nitric oxide and…
A: The balanced reaction and the intermediate are given below
Q: A. Predict the pressure of nitrogen gas at T =-98 K and v=0.00375 m³/kg on the basis of (a) the van…
A:
Q: A. Predict the pressure of nitrogen gas at T =-98 K and v=0.00375 m³/kg on the basis of (a) the van…
A:
Q: Part 3. Solubility. For the following molecules label whether you think it will be more soluble in…
A:
Q: How many protons appear in the balanced Vos lagi ag (in acid) *enter numeric value only. Answer:
A: Electrochemistry is the branch of chemistry in which both oxidation and reduction reaction occurs…
Q: The value of the gas constant in SI unit is 8.314 J mol-1 K-1 What is the unit in L atm mol-1 K-1 a.…
A: Given : The value of the gas constant ( R ) in SI unit is 8.314 J mol-1 K-1 .
Q: B. A 1.0-mol sample of an ideal gas is kept at 0 °C during an expansion from 0.003 m³ to 0.01 m³.…
A:
Q: Alanine (HA) is a diprotic amino acid with Kal = 4.53 x 10-3 and K₁2 = 1.36 x 10-10. Determine the…
A: a) A 0.241 M alanine hydrochloride (H2A+Cl-) solution- Now, the dissociation of H2A+Cl- in the…
Q: Ascorbic acid (H₂CH₂O) is a diprotic acid. The acid dissocation constants for H₂CH₂O are Kal = 8.00…
A:
Q: Compare the pH of a 0.040 M HCl solution with that of a 0.040 M H₂SO4 solution. (Hint: H₂SO4 is a…
A:
Q: Calculate the [H] and pH of a 4.91 x 10-4 M iodoacetic acid solution. The K₂ of iodoacetic acid is…
A:
Q: You time travel 100 years into the future and learn that se and D) have been discovered. Several…
A:
Q: 1.) i.)Using the infrared spectra provided assign peaks to the functional groups in (a)…
A: (a)3-nitroacetophenone; (b) 3-aminoacetophenone; (c) 1-(3-nitrophenyl)ethanol.
Q: A given mitochondria is about 1x107 m in diameter. An atom is around 1x10-10 m in diameter. How many…
A: Diameter of mitochondria = 1×10-7 m Diameter of an atom = 1×10-10 m
Q: For the reaction Ca(s)+Cl2(g)→CaCl2(s) calculate how many grams of the product form when 21.4 gg…
A: Given reaction is Ca(s)+Cl2(g)→CaCl2(s) We have to calculate how many grams of CaCl2 form when 21.4…
Q: 4. Draw the conjugate base for each of the following acids: a. b. c. NH3 d. H3O+ e. OH f. g.NH OH…
A: Conjugate base is formed when protan is abstracted from compound .
Q: 3 2 1 0 -1 -2 0.00086 0.00088 0.0009 Arrhenius Plot 0.00092 0.00094 0.00096 0.00098 0.001 0.00102 B.…
A: Below are the required data:
Q: 1. Ammonium Nitrate is an unstable explosive that also happens to be highly soluble in water. It is…
A:
Q: NH₂ CHO + ပုံ-ပုံ OH OC2H25 4-aminophenol Acetic Acid EtOH 4-(dodecyloxy)benzaldehyde PC12CHO…
A: We have to give the mechanism of the given reaction. Concept: Here in the given reaction,…
Q: name functional group
A: Since, Functional group represent the characteristics of the compound. Example- For methanol =>…
Q: isomeric relationship?
A:
3.7
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:(CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of theseWhich of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)
- Complete the following reaction: Pb+2 + SO4 -2 ---------------What are the products from the following reactions? Please help with part e,f,gFor SN2 Explain the order in which 1o (primary) alkyl halides reacted and explain why. The 1o primary alkyl halides are: (see picture below) 1-chlorobutane 1-bromobutane 1-chloro-2butene benzylchloride
- 2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd1. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)
- Chemistry 60. Give the principal product(s) expected, if any, when trans-1,3-pentadiene reacts under the following conditions. Assume one equivalent of each reagent reacts unless noted otherwise. (a) H2O, H3O+ (b) Na+EtO- in EtOH (c) Maleic anhydride heat2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :



