When 362. g of alanine (C3H-NO₂) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 8.20 °C less than the freezi point of pure X. Calculate the mass of iron(III) chloride that must be dissolved in the same mass of X to produce the same depression in freezing point. The van't Hoff fact i = 3.56 for iron (III) chloride in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 3 significant digits. 0x10 X
When 362. g of alanine (C3H-NO₂) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 8.20 °C less than the freezi point of pure X. Calculate the mass of iron(III) chloride that must be dissolved in the same mass of X to produce the same depression in freezing point. The van't Hoff fact i = 3.56 for iron (III) chloride in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 3 significant digits. 0x10 X
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter10: Properties Of Solutions
Section: Chapter Questions
Problem 124CP
Related questions
Question
100%
Please let me know if there are any units associated with it and keep in mind the sig figs its asking for. Thank you so much I'll give you a thumbs up??.
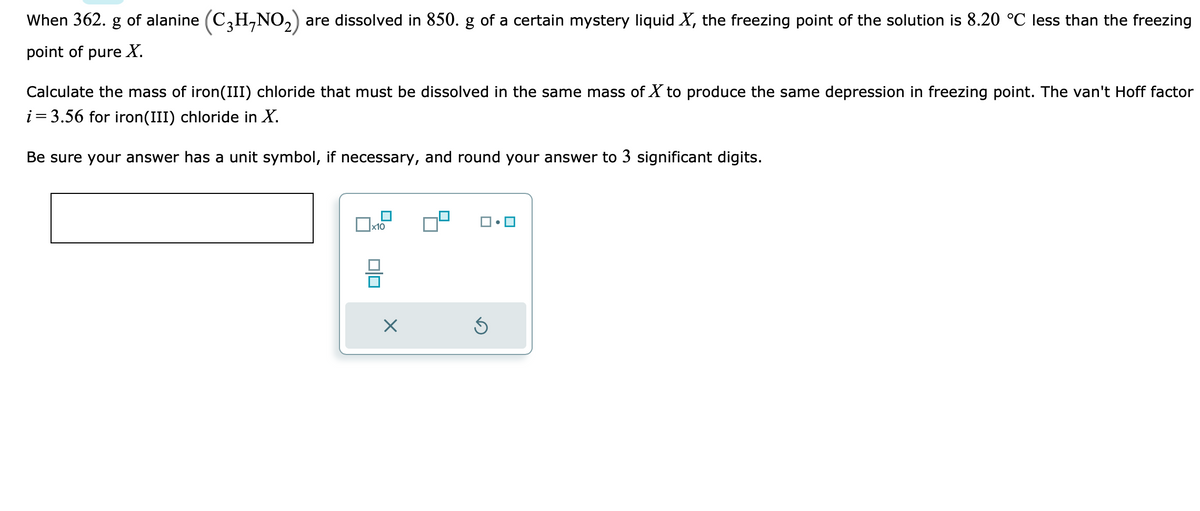
Transcribed Image Text:When 362. g of alanine (C₂H-NO₂) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 8.20 °C less than the freezing
point of pure X.
Calculate the mass of iron(III) chloride that must be dissolved in the same mass of X to produce the same depression in freezing point. The van't Hoff factor
i = 3.56 for iron(III) chloride in X.
Be sure your answer has a unit symbol, if necessary, and round your answer to 3 significant digits.
x10
00
×
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning