Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter13: The Chemistry Of Solutes And Solutions
Section: Chapter Questions
Problem 83QRT
Related questions
Question
Which of the five dyes tested in this experiment has the strongest ability to absorb light? How do you know?
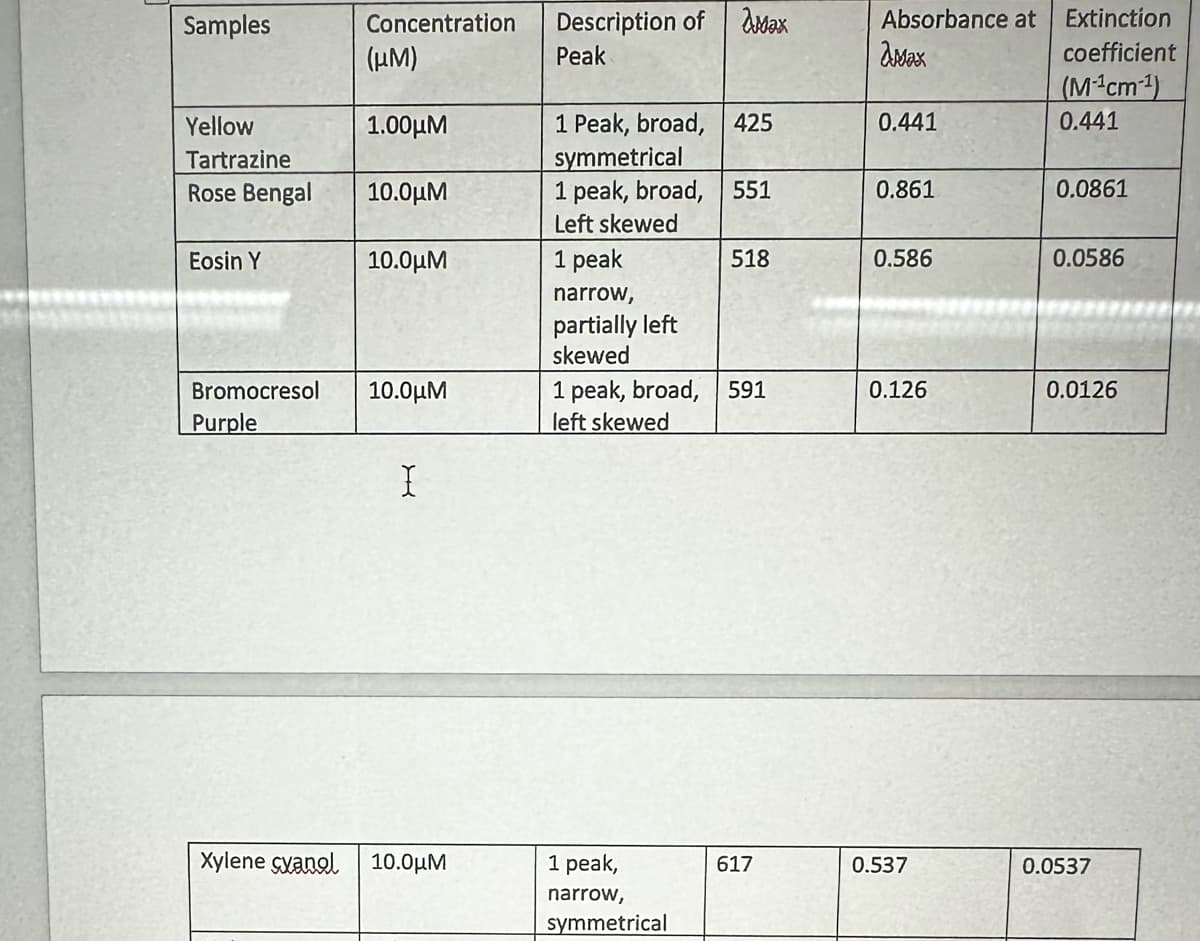
Transcribed Image Text:Samples
Yellow
Tartrazine
Rose Bengal
Eosin Y
Concentration
(μM)
1.00μ.
10.0μΜ
10.0μΜ
Bromocresol 10.0μΜ
Purple
X
Xylene cyanol 10.0μΜ
Description of
Peak
1 Peak, broad, 425
symmetrical
1 peak, broad, 551
Left skewed
1 peak
narrow,
partially left
skewed
Max
1 peak,
narrow,
symmetrical
518
1 peak, broad, 591
left skewed
617
Absorbance at
amax
0.441
0.861
0.586
0.126
0.537
Extinction
coefficient
(M¹cm¹)
0.441
0.0861
0.0586
0.0126
0.0537
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
