Q: Which is the stronger acid? a. HCl or HBr b. CH3OH or CH3SH c. HF or HCl d. H2s or H2O
A: Since your question has multiple sub-parts, we will solve first three sub-part for you. If you want…
Q: Rank the following compounds in order of decreasing acidity, putting the most acidic first. CH4 NH3…
A: Acidity is the character of the molecule to lose its proton in the aqueous solutions. Multiple…
Q: H2C CH2 H2C-
A: The methylene group attached to the electron-withdrawing carbony group is called the active…
Q: Which of the following is a diprotic acid? HCl H2SO4 CH3COOH H3PO4
A: Which of the following is a diprotic acid? HCl H2SO4 CH3COOH H3PO4
Q: Indicate whether the phenol on the left of each pair is more or less acidic than the one on the righ…
A:
Q: Arrange the following carboxylic acids in order of acidity. COOH соон COOH COOH The strongest acid…
A: We have to consider two factors: If an electron withdrawing atom is attached to the carbon chain,…
Q: HN-CH3 CH2 NO2 H2C-CI COOH COOH ČOOH II III
A: In this question, we will arrange all these three Compounds in Increasing order of Acidity. You can…
Q: Which of the following bases can remove a proton from acetic acid in a reaction that favors…
A: Given bases:
Q: Both acetic acid and phenol will react with aqueous NaOH to yield a salt, but only acetic acid will…
A: The acid is the one that donates the proton and accepts the pair of electrons while the base is the…
Q: Place the following compounds in order of their acidity with #1 being the most acidic and #5 being…
A: The substance which gives hydrogen ions, known as acid. The strength of an acid depends on the…
Q: Choose the stronger acid: H2S or H2Se O H2Se O H2S Why is this the stronger acid? O It has more lone…
A: In such cases acidity is explained in term of bond strength. Weaker the bond stronger the acid.
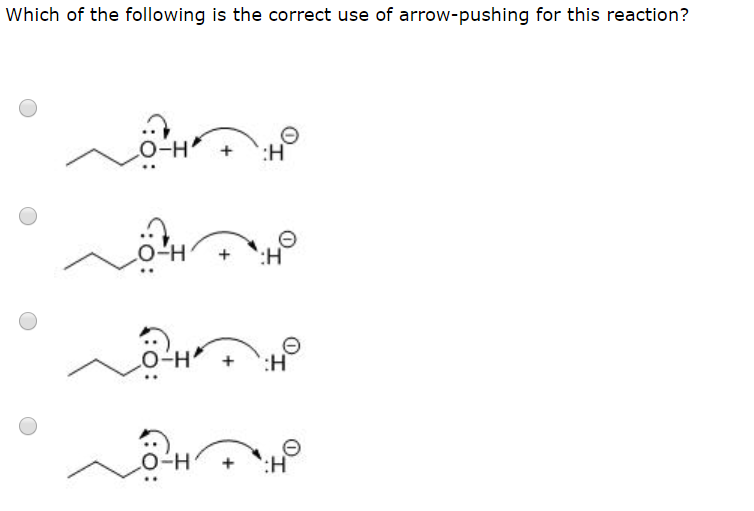
Q: For each reaction, label the Lewis acid and base. Use curved arrow notation to show the movement of…
A: Lewis acid is that compound which accepts a lone pair of electrons And Lewis base is that compound…
Q: Label each reactant and product in this reaction as a Brønsted acid or base. CH, OH + OH- CH,O¯ +…
A: According to Bronsted concept: An acid is defined as a substance which has a tendency to donate a…
Q: (CHINH + CH,CH mwo e acids that appeared in the preceding ans according to decreasing acidity. most…
A: In acid base reaction,reaction takes place in the direction where weak acid/base if formed.
Q: Which hydrogen in pseudoephedrine, the nasal decongestant in the commercial medication Sudafed, is…
A:
Q: complete the equation for the reaction between each Lewis acid-base pair. In each equation, label…
A: Concept introduction: Lewis acid: These are substance that donates proton and accepts a pair of…
Q: Rank each set of compounds in order of increasing basicity.(a) NaOH, NH3, CH3NH2, Ph¬NH2 (
A: The increasing order of basicity of given compounds is as follows:
Q: a) Which of the following sulfonic acids is the strongest? H3C-S-OH 3C-S-OH -OH A B
A: According to the Bronsted lowry concept : Acids are those substances which possess the ability to…
Q: Vhich is the most acidic proton in the following compound? IV H. CN II H- H. -II H.
A: Most acidic proton is the proton which can be abstracted easliy. After the removal of proton, the…
Q: Rank the following compounds from strongest acid to weakest acid:
A: When the H+ (proton) is donated by compound 3 we will have a -ve charge on O which is in conjugation…
Q: N. DN H-N F E OF
A: When loan pair of nitrogen not involved in delocalization (resonance) then it shows more basic…
Q: | Rank the following compounds from strongest acid to weakest acid. `OH F3C *OH A D
A: Carboxylic acids and ketones both belong to the class of carbonyl group. But carboxylic acids are…
Q: Brønsted acidities
A:
Q: Write a stepwise reaction sequence using proton transfer reactions toshow how the following reaction…
A: The reaction involves proton transfer reactions. The first step in the reaction is the extraction of…
Q: H A В
A: Carbonyl compounds are those functional groups that contains (C=O) bond. The carbon adjacent to…
Q: The following reaction runs as written therefore... 1HClO2 +1NH3 ==>1ClO2- +1NH4+ (a)HClO2 is a…
A: The reaction given is, => HClO2 + NH3 -------> ClO2- + NH4+ Given : The reaction runs in the…
Q: strongest acid?
A: In general, acids are those which are capable of donating protons (H+). These generic acids also…
Q: Rank the compound of the attached group from strongest acid to weakest acid:
A: The acidity depends on the stability of conjugate base formed after release of proton. Higher the…
Q: Complete the following acid-base reactions.
A: The given reaction is,
Q: List the following compounds in order from strongest acid to weakest acid: CH4 CH3COOH CH3OH CHC,OH
A: An acid is a compound that loses proton or accepts an electron. However, a base will be just…
Q: Rank the following conjugate bases in order of decreasing basicity, putting the most basic first.…
A: The basic strength of any molecule is directly proportional to the tendency to donate the lone pair…
Q: Rank the following compounds from least acidic to most. Rank from least acidic to most acidic. To…
A: We know that terminal alkynes are most acidic and middle alkynes are least acidic and acidity of…
Q: In each reaction, Label the reactants as a Lewis base (nucleophile) or a Lewis acid (electrophile).…
A: the given reactant is:-
Q: Predict the stronger acid in each pair: (a) HCl or HF;(b) H3PO4 or H3AsO4; (c) HBrO3 or HBrO2; (d)…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: What is the conjugate acid of HAsO4-2? H3O+ H2AsO4- H3AsO4 H3As AsO4-3
A: A conjugate acid contains one more hydrogen atom and one more positive charge than the base that…
Q: Which compound of each of the following pairs is the STRONGER BASE? * (A) (B) O Option A O Option B…
A:
Q: Arrange the ff in increasing acidity: water, ethanol, t-butyl alcohol, phenol
A: Acidity is defined as the ease with which a molecule can donate protons. if it can donate PROTONS…
Q: Rank the labeled protons (Ha–Hc) in mandelic acid, a naturally occurring carboxylic acid in plums…
A: The stability of a conjugate base formed after the removal of a proton decides the acidic strength…
Q: Complete the folowing reactions. If no reaction occurs, write "No reaction" a) CH -Ç H-CH, -COOH…
A: Acids possess an ability to liberate H+ ions in solution. When they are treated with strong metals,…
Q: From the following list, please choose the conjugate acid of NH3' - CH2 - O NH3* - CHR – COOH O NH3*…
A: Acids are substances that release H+ ions. Bases are substances that accepts H+ ions. Note: Since…
Q: Which of the following species is the strongest base? Multiple Choice HO H2N" CH3COO cr
A: A solution is said to be strong base , if it easily accepts H+ (protons ) in its solution.
Q: Label each reactant and product in this reaction as a Bronsted acid or base. CH3OH + OH- ---->…
A: In the neutralization reaction, the species donates a proton H+ is called Bronsted acid and the…
Q: 5) H₂SO4 + OH → HSO₂ + H₂O 6) H₂SO4 + H₂O → H₂0² + HSO4
A:
Q: What would be a conjugate acid-base pair in the following reaction? HS^- + H2PO4^-2 --> HPO4^-2 +…
A:
Q: In each reaction, Label the reactants as a Lewis base (nucleophile) or a Lewis acid (electrophile).…
A: Lewis acid is one which which accept lone pair and Lewis base is one which donate lone pair of…
Q: Which anion (A or B) is the stronger base?
A: A base is a substance which is capable of accepting H+ ions or donating electron pairs. The ease…
Q: For each attached reaction, label the Lewis acid and base. Use curved arrow notation to show the…
A: Lewis acid bases theory defines acids and bases as: Acids: Electron pair acceptors Bases: Electron…
Q: "OH CH3 CH3
A: Basicity of these anions depends upon the electron density on oxygen atom.
Q: Rank the following species in order of increasing acidity. Explain your reasons for ordering them as…
A: Species in order of increasing acidity : NH3 < H2O < CH3OH < CH3COOH < HF <…
Q: (a) (b) (c) NH2 NH2 OH of
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Answer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.As we shall see in Chapter 19, hydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone.Complete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reaction. (a) (b) (c) (d)
- (E) Label each of the following as strong acid, strong base, or neither.In the following acid-base reaction, which is the acid, which is the base, which is the conjugate acid and which is the conjugate base: (C2H)- + C3H6O = C2H2 + (C3H6O)-I) The following reaction runs as written therefore... 1HClO2 +1NH3 ==>1ClO2- +1NH4+ (a)HClO2 is a weaker Bronsted base than NH4+(b)ClO2- is a stronger Bronsted base than NH3(c)HClO2 is a stronger Bronsted Acid than NH4+(d)None of those is correct.
- Of the following, which is the strongest acid? H2SO3 HSO4- HSO3- H2SeO4 H2SO4In each of the following acid-base reactions, identify the Bronsted acid and base on the left, and theirconjugate partners on the right: (a) HAsO32- + CH3CH2CO2H → H2AsO3- + CH3CH2CO2- (b) HPO32- + HSeO4- → SeO42- + H2PO3- (c) HC6H5O72- + HS- → H2S + C6H5O73-Rank the compounds or ions in order of increasing acidity (or decreasing p?a).
- Arrange the following compounds or anions in each group in order of decreasing basicity: (a) H3C−, H2N−, HO−, F− (b) H2O, NH3, HO−, H2N−For each equilibrium, label the stronger acid, stronger base, weaker acid and weaker base. For each reaction(s), does the position of equilibrium lie toward the right or left? H3PO4 + OH- ⇌ H2PO4- + H2O H2O + Cl- ⇌ HCl + OHLabel each reactant and product in this reaction as a Bronsted acid or base. CH3OH + OH- ----> CH3O- + H2O <----





