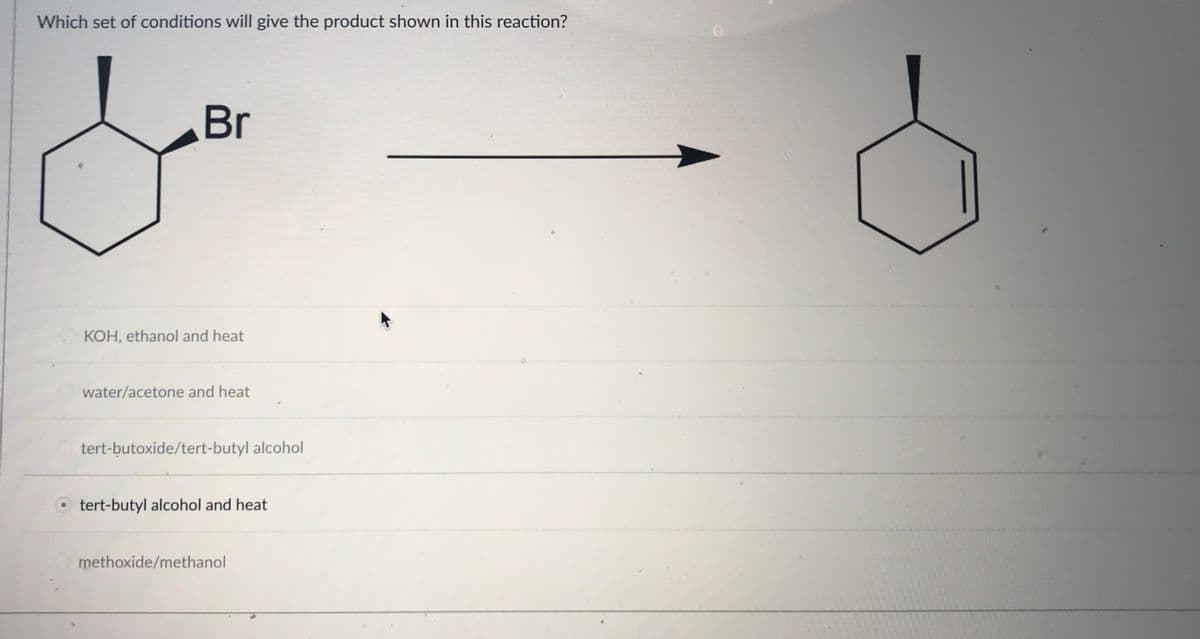
Which set of conditions will give the product shown in this reaction? Br
Q: What are the three reactions catalyzed by PLP? Just name them.
A: Pyridoxal phosphate (PLP) refers to a coenzyme present in a variety of reactions and it is the…
Q: Describe several general ways that Ab-Ag reactions are detected.
A: Antigens are the protein molecules capable of inducing an immune response. Antibodies…
Q: what is the Km for the following enzyme michael menten
A: Michaelis menten constant, Km is the substrate concentration required to produce half maximum…
Q: What is an endergo reaction?
A: An endergonic reaction is a chemical reaction which requires external force of energy to carry out…
Q: Using the half reactions from the given table, what is the reaction of this reduction-oxidation?
A: Tricarboxylic acid cycle is the final common oxidative pathway for oxidation of carbs, fats and…
Q: Given the following reaction, identify the class of the enzyme in CH20-PO, 2- CH,OH он O. OH OH OH…
A: Enzymes are biocatalysts that speed the reactions by decreasing the activation energy of the…
Q: What is the product rule?
A: Probabilities are generally mathematical measures and are a way of determining how likely something…
Q: What is the enzyme's turnover number?
A: Given, Mr of enzyme = 50,000 Concentration of enzyme = 10 μg Turnover number indicates maximum…
Q: Under standard conditions, will the following reaction proceed spontaneously as written? Fumarate +…
A: A spontaneous processes is one that occurs without the addition of external energy. A spontaneous…
Q: Write the reaction, occurring in the liver, that causes theoxidation of ethanol. What is the product…
A: The liver is an organ involved in detoxification reactions in the human body.
Q: What is an alternate name for the Krebs Cycle?
A: Krebs cycle is a series of chemical reaction which was used to generate the energy through the…
Q: What is the rate-limiting reaction?
A: A Rate-limiting reaction is one of the slowest reactions in any series of multistep reactions. This…
Q: Give a reaction equation for the qualitative and quantitative determination of I2.
A: Iodine: Iodine (I) is a lustrous element; atomic number is 53. It is the heaviest of the stable…
Q: Why are two equivalents of base required to prepare the active thiamine catalyst?
A: Catalyst is a substance that can be added to a reaction to increase the reaction rate without…
Q: What is the definition of hydrolosis reaction?
A: In human body, digestion and glucose metabolism are examples of hydrolysis reaction. Solubilization…
Q: In the reaction shown above, Acetaldehyde is being. to form Ethanol. (note the direction of the…
A: The fermentation process is an anaerobic process of generating ATP(adenosine triphosphate); a unit…
Q: What three types of reactions don’t fit in the simple Michaelis-Menten model
A: Enzymes are proteins that accelerate the biochemical reactions. They are temperature sensitive.…
Q: hat is the condensed formula of the products obtained from complete hydrolysis with the picture?
A: Background information for lipids Lipids are a group of compounds that are oily or greasy in…
Q: Is It Possible to Design an Enzyme to CatalyzeAny Desired Reaction?
A: Enzymes catalyze thermodynamically favorable reactions causing them to proceed at extraordinarily…
Q: Draw the product that is obtained from the reaction of cholesterol with the H2O, H+
A: Cholesterol is the major sterol in animal tissues. It consists of steroid nucleus called…
Q: From the active site diagram below, please indicate the cofactor from the indicated components. HO 4…
A: Cofactors are the non protein molecules that help the enzyme to carry out catalysis. Enzymes without…
Q: What are the two properties of enzymes that make them especially useful catalysts?
A: In the human body, many chemical reactions are taking place and every chemical reaction needs a…
Q: What is the mechanism of an enzyme reaction involving a coenzyme for Redox Rxns?
A: Enzymes are a class of proteins that performs various activities in the living body. They are also…
Q: The hydrolysis of ATP to yield AMP and PPi yields_____________ kJ/mol
A: ATP is considered an energy caring molecule that available in the living cell. ATP is made of three…
Q: For the reaction below: CH3CH2I DMF
A: It is an williamson ether synthesis
Q: Which curve represents the spontaneous reaction, and which the nonspontaneous?
A: Gibbs Free energy is the energy that was available to do work under constant temperature and…
Q: In casein hydrolysis the substrate for the enzyme is?
A: The substance in which enzyme acts is known as substrate. Enzyme is a protein which catalyse a…
Q: Why is it unlikely that nonenzymatic catalysts operate by preferentially binding the transition…
A: Both enzymes and catalysts control the speed of a reaction. The distinction between enzymes and…
Q: What are allosteric enzymes and what graph is obtained from the reaction of this enzyme?
A: Enzymes are the proteinaceous substances which are capable of catalyzing chemical reactions of…
Q: Given the following reactions, identify the class and subclass of the enzyme involved
A: The enzyme commission divided enzymes into six different classes. This classification if carried out…
Q: What is meant by general acid catalysis with respect to enzyme mechanisms?
A: An enzyme draws substrates to its active site, catalyzes the chemical reaction that creates…
Q: Under standard conditions, will the following reaction proceed spontaneously as written?
A: A chemical process in which one substance changes into another is known as a chemical reaction. A…
Q: How does the rate of the forward reaction compare to the rate of the reverse reaction for an…
A: LeChatelier's principle, predicts the change in the conditions in chemical reactions that affects…
Q: What properties of transition metals make them useful as enzyme cofactors?
A: Cofactor is a compound that is not made up of protein but rather than is a metal ion. They serves as…
Q: What products will result from the hydrolysis of 1-arseno-3- phosphoglycerate?
A: Introduction: In the glycolysis pathway, the enzyme glyceraldehyde 3-phosphate dehydrogenase…
Q: What will be the products? Which enzyme will catalyzes this reaction?
A: Proteases are synthesized as inactive proteins called zymogens. They are cleaved by other proteases…
Q: Mevalonate is a component of which biochemical pathway?
A: The carboxylate anion of mevalonic acid is known as mevalonate is of major pharmaceutical…
Q: What is the name of reagent used? (b) What is your observation if any unreacted starting material is…
A: Ans. Aspirin is a common drug, used as an anti-inflammatory and in the diagnosis of minor aches,…
Q: What are the 8 compounds formed in the Krebs cycle?
A: Kreb's cycle: A series of chemical reactions used by all aerobic organisms to generate…
Q: Write out the structures for the cofactors involved in the following biochemical reactions:
A: a). Methylation - It means addition of methyl group to a substrate. b). Amino acid decarboxylation -…
Q: The following is an example of a _____________________reaction.CH3CH2OH CH2 => CH2 + H2O
A: Ethanol (CH3CH2OH) is alcohol, which exhibits hydroxyl group as the functional group.
Q: Is the reaction of 2-phosphoglycerate to 2-phosphoenolpyuruvate is a redox reaction?
A: This occurs in glycolysis.
Q: Explain the process of polyacrylamide reaction, and what are the two reagents are crucial for this…
A: Polyacrylamide is defined as a mesh like matrix that is suitable for the separation of proteins of…
Q: How can phosphorylation drive an endergonic reaction?
A: Phosphorylation is the addition of a phosphate group to a substrate. An endergonic reaction requires…
Q: H2N NH3 HO, H,O H2N. NH2 СООН COOH glutamic acid glutamine why does the reaction not proceed?
A: Amino acids are units that synthesize proteins. Amino acids are synthesized by ribosomes. The…
Q: Minor product(s): Мео
A: Elimination reaction
Q: Name the enzyme whose substrate is(a) Urea (b) Cellulose
A: Introduction: Enzymes are proteins that catalyze the reaction. Their main function is to accelerate…
Q: Please name the missing product marked x in this reaction H-Q АТР + C--0 - X + ADP
A: In such reaction ATP molecules is reacts with carbonate molecule which gives ADP and carboxyl…
Q: Identify the intermediate in the phosphoglucomutase reaction.
A: Phosphoglucomutase is an enzyme that helps in the interconversion of glucose 6 (G6P) phosphate to…
Step by step
Solved in 2 steps with 1 images

- Which of the following reagents can be used in the reaction in the photo? A.) pyridinium chlorochromate B.) potassium permanganate C.) sodium dichromate with sulfuric acid D.) potassium chromate with sulfuric acidWhich of the following correctly identifies the products and reactants labeled in the image above?Which of the following classifications apply to the formation of 3-hydroxybutyrate from acetoacetate?(a) Condensation (b) Hydrolysis(c) Oxidation (d) Reduction
- Under standard conditions, will the following reaction proceed spontaneously as written? Fumarate + NADH + H+⇌ succinate + NAD+Which of the following best describes this reaction ? Choose one: O A. redox O B. condensation C. hydrolysis Identify the following electron transport intermediate. H : O : O : H the superoxide free radical B. hydrogen peroxide the hydroxyl free radical water hydroxide ion
- Define the following terms:a. allyl groupb. epoxidec. SAM d. PAPSe. phase I reactionDetermine whether or not reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. HBr(aq) + KOH(aq)--------> H2O(l) + KBr(aq)Use the data from the table below to answer the questions on an ezyme obtained from potato extract. The greater the absorbance value, the more product generated for each chemical reaction Table 1: Summary Data for Enzyme Catalyzed Reaction Product Formation Measured as Absorbance Tube Distilled Water Catechol PTU (Inhibitor) Potato Extract Results (Absorbance) 1 5.5 ml 0.5 ml -- --- 0.02 2 5 ml 0.5 ml --- 0.5 ml 0.68 3 4.5 ml 0.5 ml 0.5 ml 0.5 ml 0.04 4 4.5 ml 0.5 ml 1.0 ml 1.26 5 4.5 ml 1.0 ml 0.5 ml 1.22 a) Explain with respect to enzyme activity why test tube 1 produced very little absorbance: b) Explain with respect to enzyme activity why test tube 3 produced very little absorbance: c) Explain how to tubes 4 and 5 differ with respect to reaction conditions. d) Explain with respect to enzyme activity why tubes 4 and 5 have similar absorbance and why both have…
- The following is an example of a _____________________reaction.CH3CH2OH CH2 => CH2 + H2OIdentify the following electron transport intermediate. : O : O : A. the superoxide free radical B. the hydroxide ionC. dihydrogen oxideD. hydrogen peroxideE. the hydroxyl free radicalHow do you account for the difference in reaction between fresh and rancid oil?

