Chapter12: Gravimetric Methods Of Analysis
Section: Chapter Questions
Problem 12.20QAP
Related questions
Question
Transcribed Image Text:WLVZ | Online teaching and lea
New tab
Ô https://east.cengagenow.com/ilrn/takeAssignment/takeCXPCompliantActivity.do?locator=Dassignment-take
CHAPTER 2 - ATOMS, MOLECULES, AND IONS
OPrevious Page 1 of 2
Next O
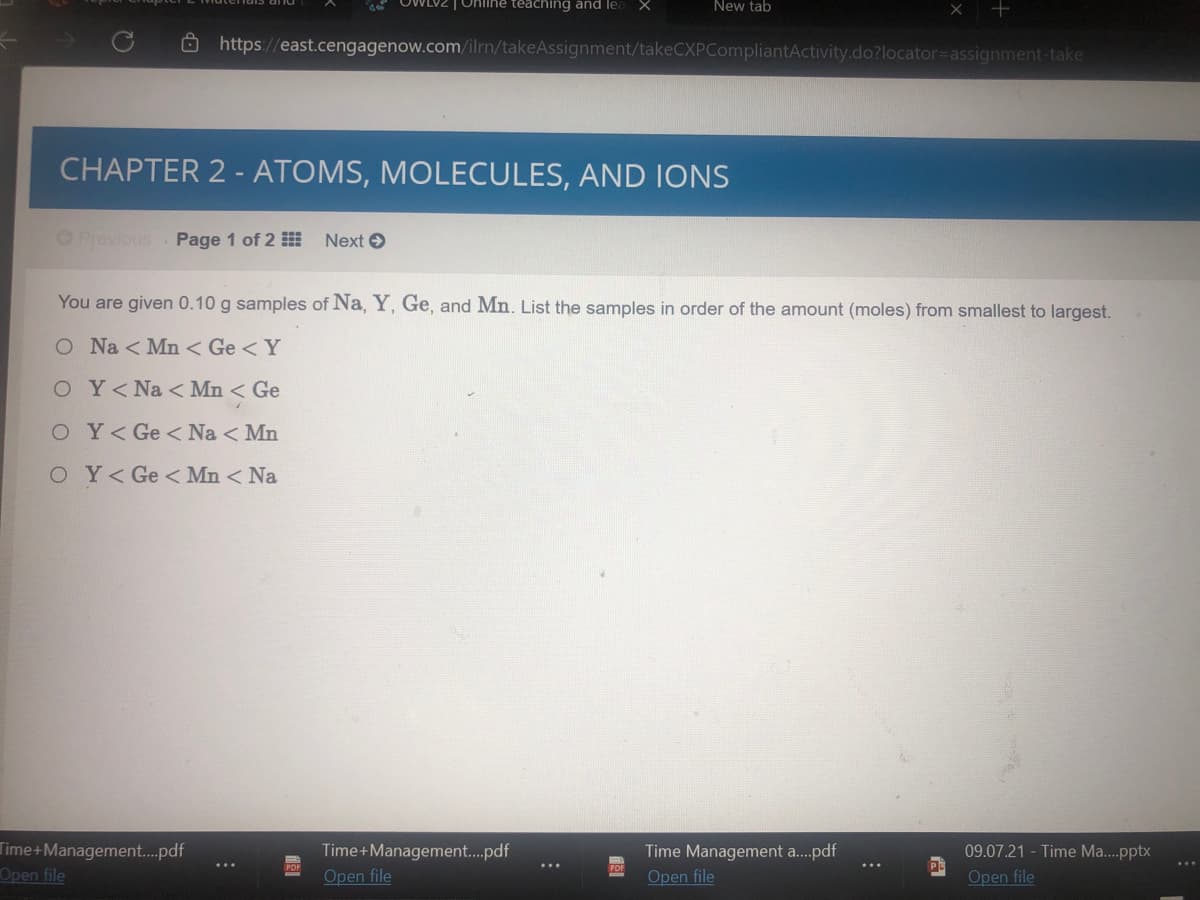
You are given 0.10 g samples of Na, Y, Ge, and Mn. List the samples in order of the amount (moles) from smallest to largest.
O Na < Mn < Ge <Y
OY<Na < Mn < Ge
O Y<Ge < Na < Mn
OY< Ge < Mn < Na
Time+Management.pdf
Open file
09.07.21 - Time Ma..pptx
Time+Management.pdf
Open file
Time Management a.pdf
Open file
Open file
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning